Exhibit 99.1
 | Promising Therapies Inspired by Urgent Unmet Patient Needs |
 | Forward Looking Statements This presentation contains forward-looking statements that involve substantial risks and uncertainties. "Forward-looking statements," as that term is defined in the Private Securities Litigation Reform Act of 1995, are statements that are not historical facts and involve a number of risks and uncertainties. Words herein such as "may," "will," "should," "could," "would," "expects," "plans," "anticipates," "believes," "estimates," "projects," "predicts," "intends," "potential," "continues," and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) may identify forward-looking statements. The forward-looking statements in this presentation are based upon the Company's current expectations and beliefs, and involve known and unknown risks, uncertainties and other factors, which may cause the Company's actual results, performance and achievements and the timing of certain events to differ materially from the results, performance, achievements or timing discussed, projected, anticipated or indicated in any forward-looking statements. Such risks, uncertainties and other factors include, among others, the following: the risk that brensocatib does not prove to be effective or safe for patients in ongoing and future clinical studies, including the ASPEN and STOP-COVID19 studies; business or economic disruptions due to catastrophes or other events, including natural disasters or public health crises; impact of the novel coronavirus (COVID-19) pandemic and efforts to reduce its spread on our business, employees, including key personnel, patients, partners and suppliers; failure to successfully commercialize in the United States or European Union (amikacin liposome inhalation suspension and Liposomal 590 mg Nebuliser Dispersion, respectively), or to maintain U.S. or EU approval for ARIKAYCE®, the Company's only approved product; uncertainties in the degree of market acceptance of ARIKAYCE by physicians, patients, third-party payors and others in the healthcare community; the Company's inability to obtain full approval of ARIKAYCE from the FDA, including the risk that the Company will not timely and successfully complete the study to validate a PRO tool and the confirmatory post-marketing clinical trial required for full approval of ARIKAYCE; inability of the Company, PARI Pharma GmbH (PARI) or the Company's.other third-party manufacturers to comply with regulatory requirements related to ARIKAYCE or the Lamira® Nebulizer System; the Company's inability to obtain adequate reimbursement from government or third-party payors for ARIKAYCE or acceptable prices for ARIKAYCE; development of unexpected safety or efficacy concerns related to ARIKAYCE or the Company’s product candidates; inaccuracies in the Company's estimates of the size of the potential markets for ARIKAYCE or its product candidates or in data the Company has used to identify physicians, expected rates of patient uptake, duration of expected treatment, or expected patient adherence or discontinuation rates; the Company's inability to create an effective direct sales and marketing infrastructure or to partner with third parties that offer such an infrastructure for distribution of ARIKAYCE or any of the company’s product candidates that are approved in the future; failure to obtain regulatory approval to expand ARIKAYCE's indication to a broader patient population; failure to successfully conduct future clinical trials for ARIKAYCE, brensocatib, trepostinil palmitil inhalation powder (TPIP) and the Company's other product candidates, due to the Company's limited experience in conducting preclinical development activities and clinical trials necessary for regulatory approval and the Company's potential inability to enroll or retain sufficient patients to conduct and complete the trials or generate data necessary for regulatory approval, among other things; risks that the Company's clinical studies will be delayed or that serious side effects will be identified during drug development; failure to obtain, or delays in obtaining, regulatory approvals for ARIKAYCE outside the U.S.or European Union or for the Company's product candidates in the U.S., Europe, Japan or other markets, including the United Kingdom as a result of its recent exit from the European Union; failure of third parties on which the Company is dependent to manufacture sufficient quantities of ARIKAYCE or the Company's product candidates for commercial or clinical needs, to conduct the Company's clinical trials, or to comply with the Company’s agreements or laws and regulations that impact the Company's business or agreements with the Company; the Company's inability to attract and retain key personnel or to effectively manage the Company's growth; the Company's inability to adapt to its highly competitive and changing environment; the Company's inability to adequately protect its intellectual property rights or prevent disclosure of its trade secrets and other proprietary information and costs associated with litigation or other proceedings related to such matters; restrictions or other obligations imposed on the Company by its agreements related to ARIKAYCE or the Company's product candidates, including its license agreements with PARI and AstraZeneca AB, and failure of the Company to comply with its obligations under such agreements; the cost and potential reputational damage resulting from litigation to which the Company is or may become a party, including product liability claims; the Company's limited experience operating internationally; changes in laws and regulations applicable to the Company's business, including any pricing reform, and failure to comply with such laws and regulations; inability to repay the Company's existing indebtedness and uncertainties with respect to the Company's ability to access future capital; and delays in the execution of plans to build out an additional third-party manufacturing facility approved by the FDA and unexpected expenses associated with those plans. The Company may not actually achieve the results, plans, intentions or expectations indicated by the Company's forward-looking statements because, by their nature, forward-looking statements involve risks and uncertainties because they relate to events and depend on circumstances that may or may not occur in the future. For additional information about the risks and uncertainties that may affect the Company's business, please see the factors discussed in Item 1A, "Risk Factors," in the Company's Annual Report on Form 10-K for the year ended December 31, 2019, Quarterly Reports on Form 10-Q for the quarters ended March 31, 2020, June 30, 2020 and September 30, 2020, and any subsequent Company filings with the SEC. The Company cautions readers not to place undue reliance on any such forward-looking statements, which speak only as of the date of this presentation. The Company disclaims any obligation, except as specifically required by law and the rules of the SEC, to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements. |
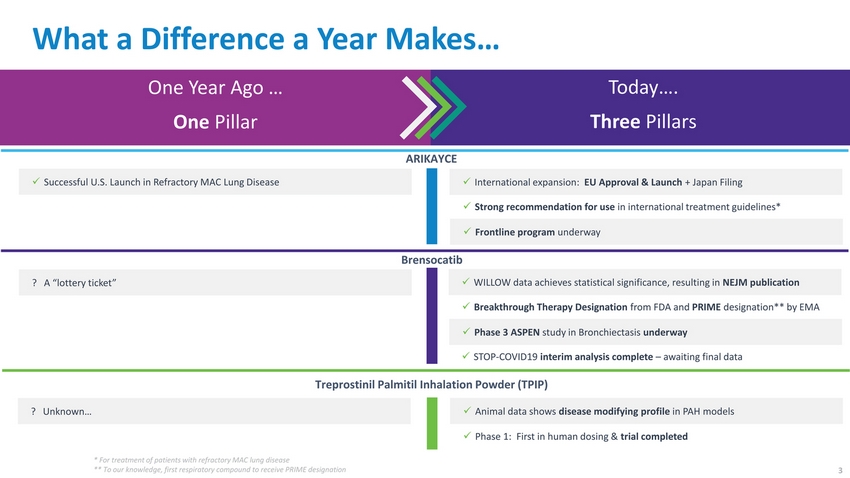 | What a Difference a Year Makes… One Year Ago … One Pillar Today…. Three Pillars Successful U.S. Launch in Refractory MAC Lung Disease ARIKAYCE International expansion: EU Approval & Launch + Japan Filing Strong recommendation for use in international treatment guidelines* Frontline program underway ? A “lottery ticket” Brensocatib WILLOW data achieves statistical significance, resulting in NEJM publication Breakthrough Therapy Designation from FDA and PRIME designation** by EMA Phase 3 ASPEN study in Bronchiectasis underway STOP-COVID19 interim analysis complete – awaiting final data Treprostinil Palmitil Inhalation Powder (TPIP) ? Unknown…✓ Animal data shows disease modifying profile in PAH models Phase 1: First in human dosing & trial completed * For treatment of patients with refractory MAC lung disease ** To our knowledge, first respiratory compound to receive PRIME designation3 |
 | Our Mission To transform the lives of patients with serious and rare diseases Our Vision To be a globally-recognized leading biotech company that empowers great people to deliver, with a profound sense of urgency and compassion, life-altering therapies to small patient populations experiencing big health problems |
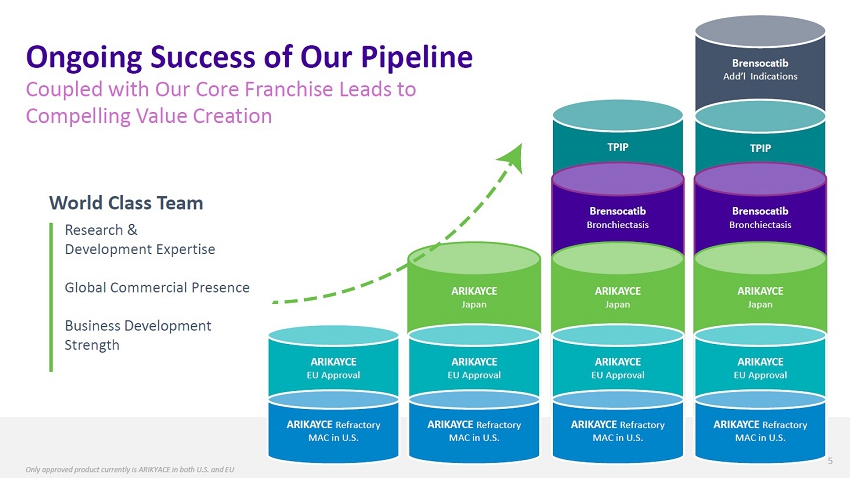 | Ongoing Success of Our Pipeline Coupled with Our Core Franchise Leads to Compelling Value Creation Brensocatib Add’l Indications TPIP TPIP World Class Team Research & Development Expertise Brensocatib Bronchiectasis Brensocatib Bronchiectasis Global Commercial Presence Business Development Strength ARIKAYCE EU Approval ARIKAYCE Japan Front Line ARIKAYCE EU Approval ARIKAYCE Japan Front Line ARIKAYCE EU Approval ARIKAYCE Japan Front Line ARIKAYCE EU Approval ARIKAYCE Refractory MAC in U.S. ARIKAYCE Refractory MAC in U.S. ARIKAYCE Refractory MAC in U.S. ARIKAYCE Refractory MAC in U.S. 5 Only approved product currently is ARIKYACE in both U.S. and EU |
 | Three Pillars of Value Creation PRECLINICALPHASE 1PHASE 2PHASE 3APPROVED ARIKAYCE® (Amikacin Liposome Inhalation Suspension)* Refractory NTM: M. avium complex (MAC) Front Line Label Expansion Brensocatib: DPP1 Inhibitor Bronchiectasis Cystic Fibrosis (IIR) COVID19 Treprostinil Palmitil Inhalation Powder (TPIP) Pulmonary Arterial Hypertension (PAH) Pulmonary Hypertension associated with Interstitial Lung Disease (PH-ILD) Idiopathic Pulmonary Fibrosis (IPF) In the U.S., as a condition of accelerated approval, Insmed is conducting an additional clinical study to support full approval. Full approved has been granted by the European Commission. 6 |
 | Treprostinil Palmitil Inhalation Powder (TPIP) 7 |
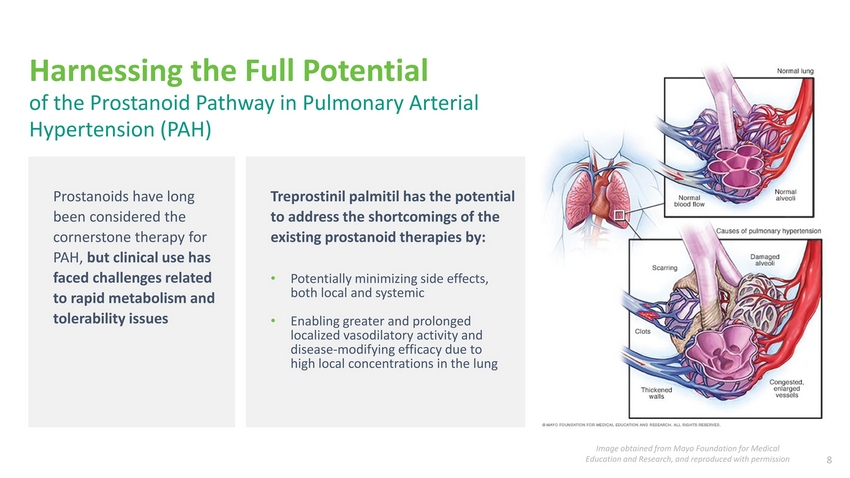 | Harnessing the Full Potential of the Prostanoid Pathway in Pulmonary Arterial Hypertension (PAH) Prostanoids have long been considered the cornerstone therapy for PAH, but clinical use has faced challenges related to rapid metabolism and tolerability issues Treprostinil palmitil has the potential to address the shortcomings of the existing prostanoid therapies by: Potentially minimizing side effects, both local and systemic Enabling greater and prolonged localized vasodilatory activity and disease-modifying efficacy due to high local concentrations in the lung Image obtained from Mayo Foundation for Medical Education and Research, and reproduced with permission 8 |
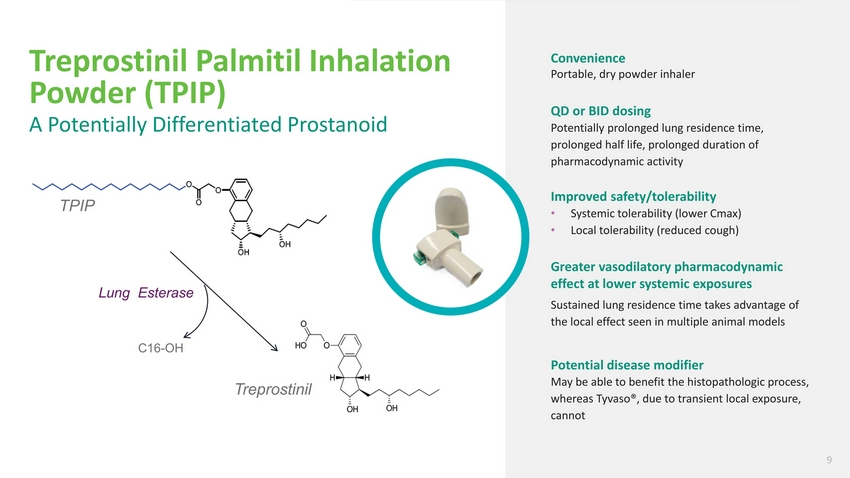 | Treprostinil Palmitil Inhalation Powder (TPIP) A Potentially Differentiated Prostanoid TPIP Lung Esterase Convenience Portable, dry powder inhaler QD or BID dosing Potentially prolonged lung residence time, prolonged half life, prolonged duration of pharmacodynamic activity Improved safety/tolerability Systemic tolerability (lower Cmax) Local tolerability (reduced cough) Greater vasodilatory pharmacodynamic effect at lower systemic exposures Sustained lung residence time takes advantage of the local effect seen in multiple animal models C16-OH Treprostinil Potential disease modifier May be able to benefit the histopathologic process, whereas Tyvaso®, due to transient local exposure, cannot 9 |
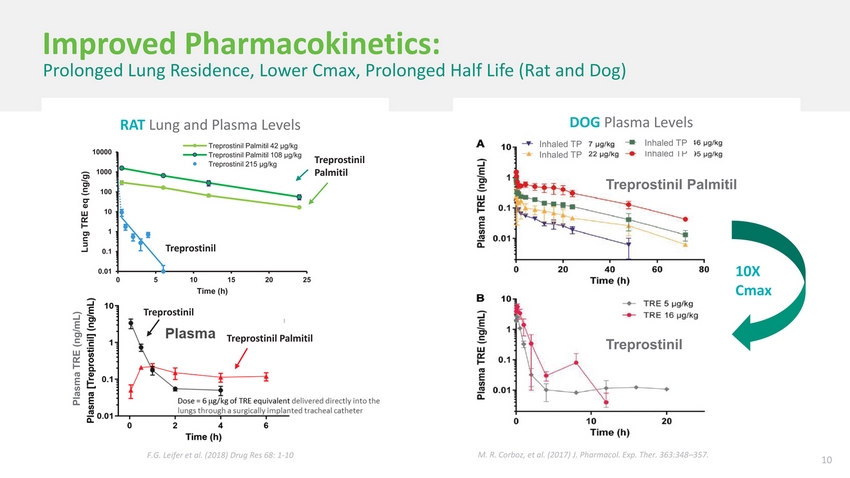 | Improved Pharmacokinetics: Prolonged Lung Residence, Lower Cmax, Prolonged Half Life (Rat and Dog) RAT Lung and Plasma Levels DOG Plasma Levels 10000 Lung TRE eq (ng/g) 100 Treprostinil Palmitil 42 μg/kg Treprostinil Palmitil 108 μg/kg Treprostinil 215 μg/kg Treprostinil Palmitil Inhaled TP Inhaled TP Inhaled TP Inhaled TP Treprostinil Palmitil 10 1 0.1 0.01 Treprostinil 0510152025 Time (h) 10X Cmax Plasma TRE (ng/mL) Treprostinil Palmitil Treprostinil F.G. Leifer et al. (2018) Drug Res 68: 1-10 M. R. Corboz, et al. (2017) J. Pharmacol. Exp. Ther. 363:348–357. 10 |
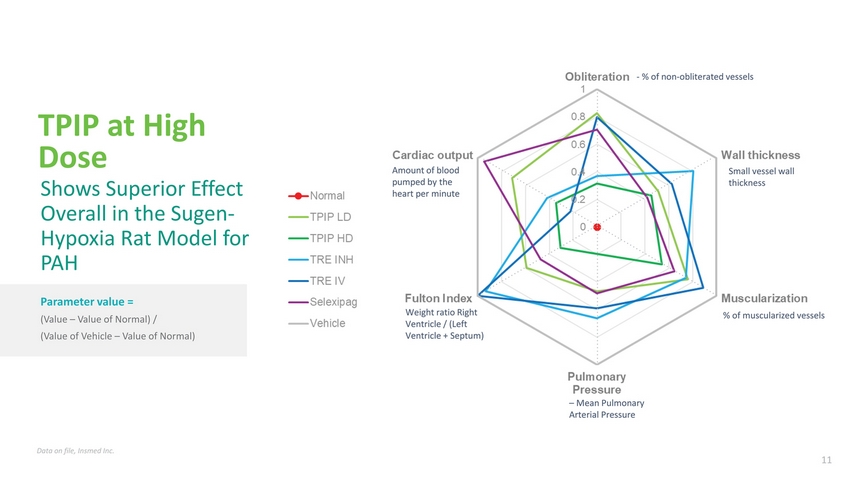 | TPIP at High Dose Shows Superior Effect Overall in the Sugen-Hypoxia Rat Model for PAH Parameter value = (Value – Value of Normal) / (Value of Vehicle – Value of Normal) Amount of blood pumped by the heart per minute Weight ratio Right Ventricle / (Left Ventricle + Septum) % of non-obliterated vessels Small vessel wall thickness % of muscularized vessels – Mean Pulmonary Arterial Pressure Data on file, Insmed Inc. |
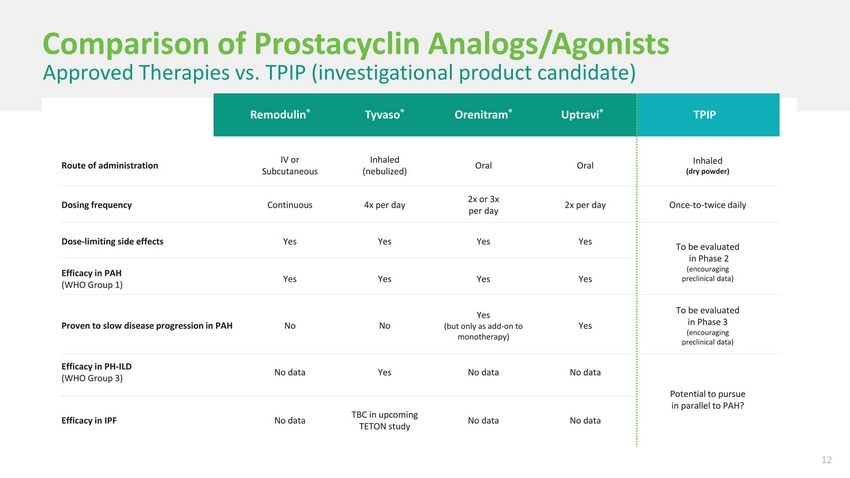 | Comparison of Prostacyclin Analogs/Agonists Approved Therapies vs. TPIP (investigational product candidate) Remodulin®Tyvaso®Orenitram®Uptravi®TPIP Route of administrationIV or Subcutaneous Inhaled (nebulized) OralOralInhaled (dry powder) Dosing frequencyContinuous4x per day2x or 3x per day 2x per dayOnce-to-twice daily Dose-limiting side effectsYesYesYesYesTo be evaluated in Phase 2 (encouraging Efficacy in PAH (WHO Group 1)YesYesYesYes preclinical data) Proven to slow disease progression in PAHNoNo Yes (but only as add-on to monotherapy) Yes To be evaluated in Phase 3 (encouraging preclinical data) Efficacy in PH-ILD (WHO Group 3)No dataYesNo dataNo data Potential to pursue in parallel to PAH? Efficacy in IPFNo dataTBC in upcoming TETON study No dataNo data |
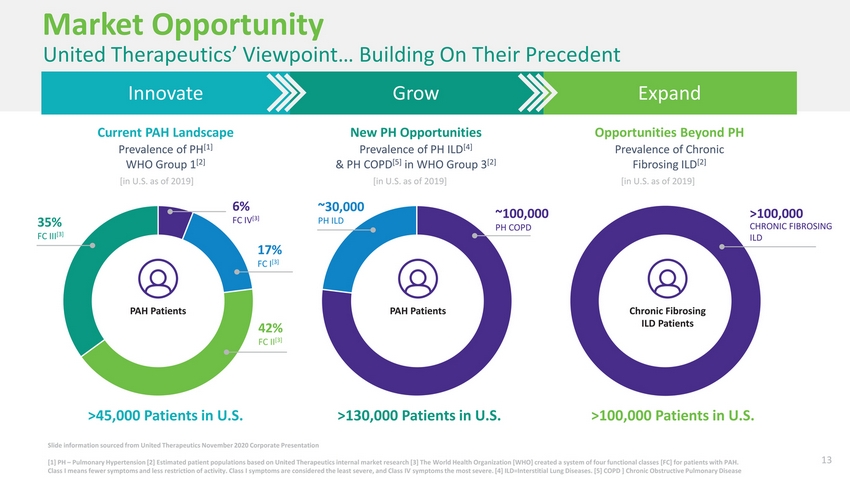 | Market Opportunity United Therapeutics’ Viewpoint… Building On Their Precedent InnovateGrowExpand Current PAH LandscapeNew PH OpportunitiesOpportunities Beyond PH Prevalence of PH[1] WHO Group 1[2] Prevalence of PH ILD[4] & PH COPD[5] in WHO Group 3[2] Prevalence of Chronic Fibrosing ILD[2] [in U.S. as of 2019][in U.S. as of 2019][in U.S. as of 2019] 35% FC III[3] 6% FC IV[3] 17% FC I[3] ~30,000 PH ILD ~100,000 PH COPD >100,000 CHRONIC FIBROSING ILD PAH PatientsPAH PatientsChronic Fibrosing 42% FC II[3] ILD Patients >45,000 Patients in U.S.>130,000 Patients in U.S.>100,000 Patients in U.S. Slide information sourced from United Therapeutics November 2020 Corporate Presentation [1] PH – Pulmonary Hypertension [2] Estimated patient populations based on United Therapeutics internal market research [3] The World Health Organization [WHO] created a system of four functional classes [FC] for patients with PAH. 13 Class I means fewer symptoms and less restriction of activity. Class I symptoms are considered the least severe, and Class IV symptoms the most severe. [4] ILD=Interstitial Lung Diseases. [5] COPD ] Chronic Obstructive Pulmonary Disease |
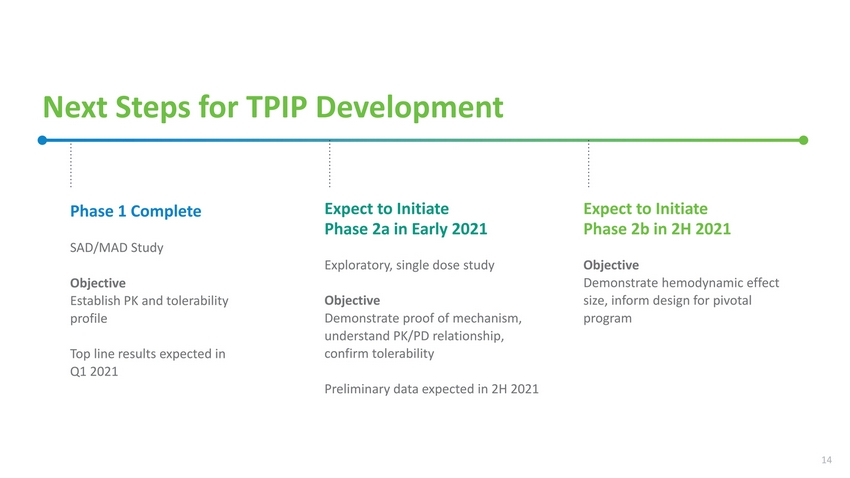 | Next Steps for TPIP Development Phase 1 Complete SAD/MAD Study Objective Establish PK and tolerability profile Top line results expected in Q1 2021 Expect to Initiate Phase 2a in Early 2021 Exploratory, single dose study Objective Demonstrate proof of mechanism, understand PK/PD relationship, confirm tolerability Preliminary data expected in 2H 2021 Expect to Initiate Phase 2b in 2H 2021 Objective Demonstrate hemodynamic effect size, inform design for pivotal program 14 |
 | Brensocatib 15 |
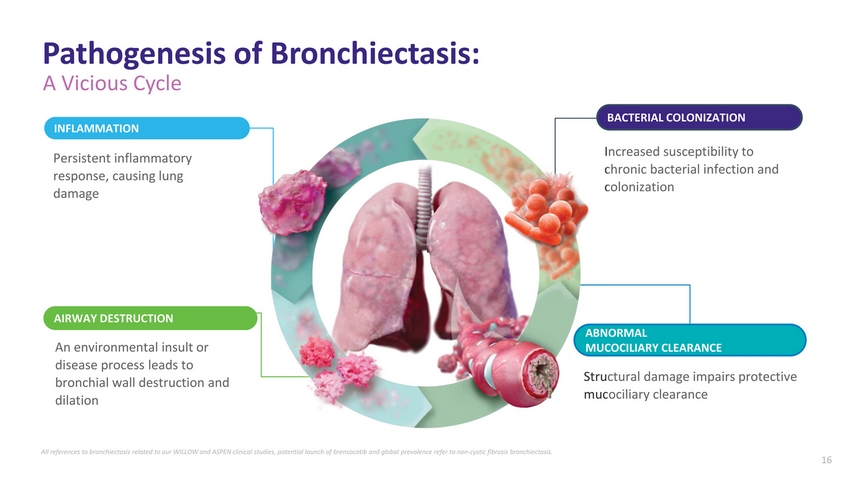 | Pathogenesis of Bronchiectasis: A Vicious Cycle INFLAMMATION Persistent inflammatory response, causing lung damage BACTERIAL COLONIZATION Increased susceptibility to chronic bacterial infection and colonization AIRWAY DESTRUCTION An environmental insult or disease process leads to bronchial wall destruction and dilation ABNORMAL MUCOCILIARY CLEARANCE Structural damage impairs protective mucociliary clearance All references to bronchiectasis related to our WILLOW and ASPEN clinical studies, potential launch of brensocatib and global prevalence refer to non-cystic fibrosis bronchiectasis. 16 |
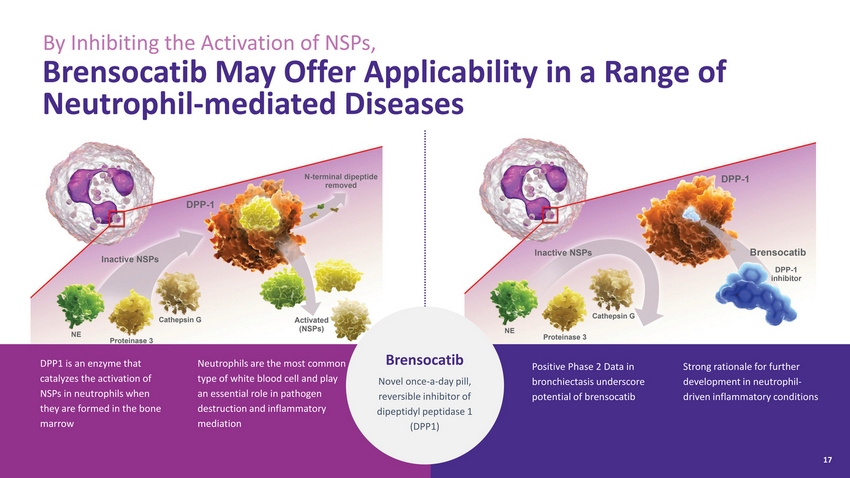 | By Inhibiting the Activation of NSPs, Brensocatib May Offer Applicability in a Range of Neutrophil-mediated Diseases DPP1 is an enzyme that catalyzes the activation of NSPs in neutrophils when they are formed in the bone marrow Neutrophils are the most common type of white blood cell and play an essential role in pathogen destruction and inflammatory mediation Brensocatib Novel once-a-day pill, reversible inhibitor of dipeptidyl peptidase 1 (DPP1) Positive Phase 2 Data in bronchiectasis underscore potential of brensocatib Strong rationale for further development in neutrophil-driven inflammatory conditions 17 |
 | WILLOW Study Success Recognized Worldwide Both 10 mg & 25 mg group met primary endpoint with statistical significance Exacerbation-related hospitalization rates was higher & duration of hospitalization was longer in the placebo arm Risk of exacerbation over six months reduced by 42% for 10 mg group, 38% for 25 mg group 18 |
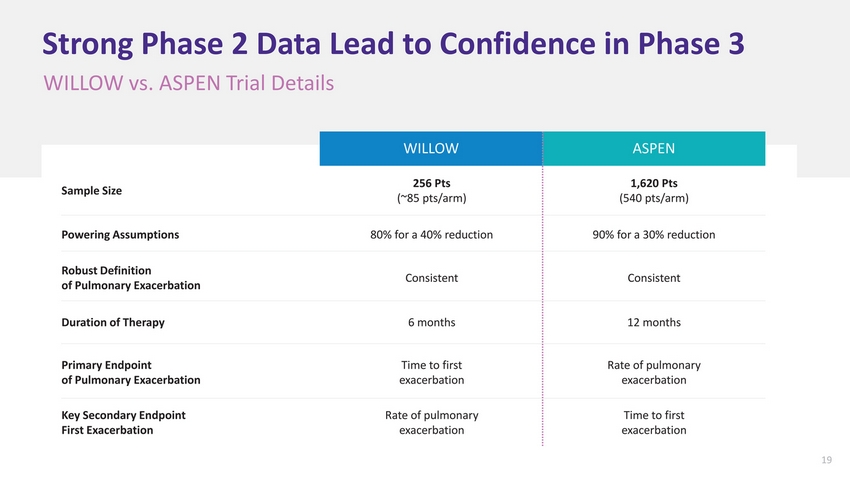 | Strong Phase 2 Data Lead to Confidence in Phase 3 WILLOW vs. ASPEN Trial Details WILLOWASPEN Sample Size256 Pts (~85 pts/arm) 1,620 Pts (540 pts/arm) Powering Assumptions80% for a 40% reduction90% for a 30% reduction Robust Definition of Pulmonary ExacerbationConsistentConsistent Duration of Therapy6 months12 months Primary Endpoint of Pulmonary Exacerbation Time to first exacerbation Rate of pulmonary exacerbation Key Secondary Endpoint First Exacerbation Rate of pulmonary exacerbation Time to first exacerbation 19 |
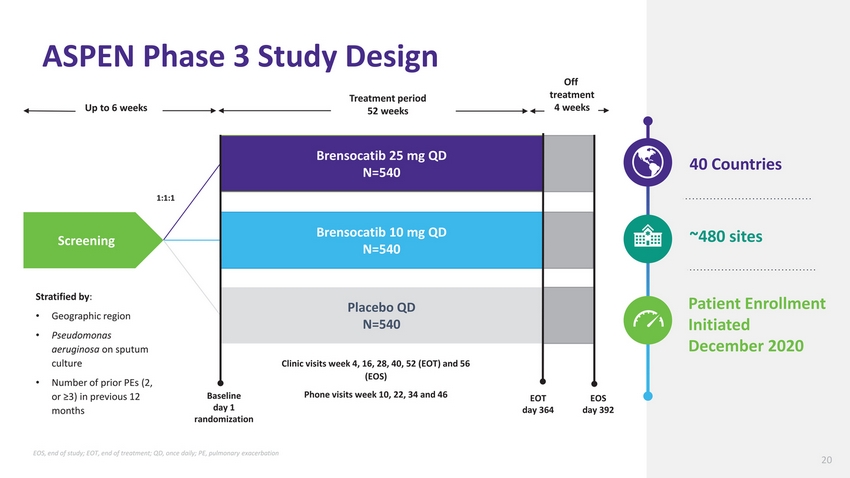 | ASPEN Phase 3 Study Design Treatment period Off treatment Up to 6 weeks 52 weeks 4 weeks Brensocatib 25 mg QD N=540 40 Countries 1:1:1 Screening Brensocatib 10 mg QD N=540 ~480 sites Stratified by: Geographic region Pseudomonas aeruginosa on sputum culture Number of prior PEs (2, or ≥3) in previous 12 months Baseline day 1 randomization Placebo QD N=540 Clinic visits week 4, 16, 28, 40, 52 (EOT) and 56 (EOS) Phone visits week 10, 22, 34 and 46 EOT day 364 EOS day 392 Patient Enrollment Initiated December 2020 EOS, end of study; EOT, end of treatment; QD, once daily; PE, pulmonary exacerbation 20 |
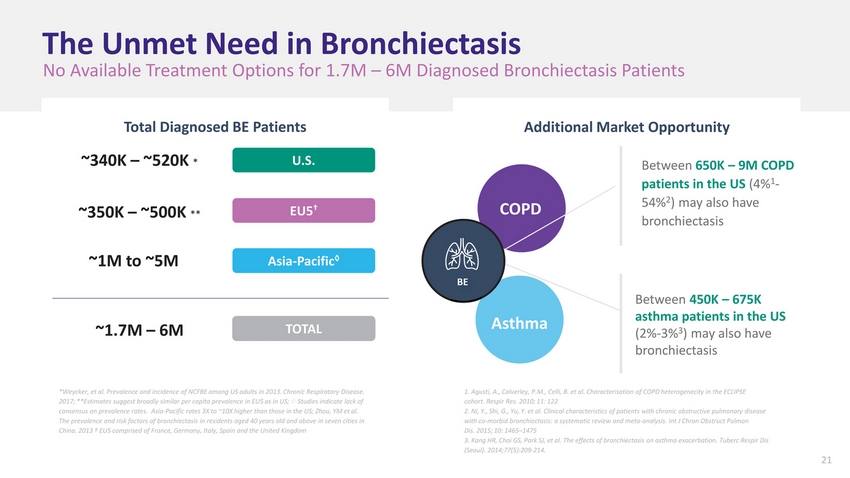 | The Unmet Need in Bronchiectasis No Available Treatment Options for 1.7M – 6M Diagnosed Bronchiectasis Patients Total Diagnosed BE PatientsAdditional Market Opportunity ~340K – ~520K * U.S. Between 650K – 9M COPD patients in the US (4%1-2 ~350K – ~500K ** EU5† COPD 54% ) may also have bronchiectasis ~1M to ~5M ~1.7M – 6M Asia-Pacific◊ TOTAL BE Asthma Between 450K – 675K asthma patients in the US (2%-3%3) may also have bronchiectasis *Weycker, et al. Prevalence and incidence of NCFBE among US adults in 2013. Chronic Respiratory Disease. 2017; **Estimates suggest broadly similar per capita prevalence in EU5 as in US; ◊ Studies indicate lack of consensus on prevalence rates. Asia-Pacific rates 3X to ~10X higher than those in the US; Zhou, YM et al. The prevalence and risk factors of bronchiectasis in residents aged 40 years old and above in seven cities in China. 2013 † EU5 comprised of France, Germany, Italy, Spain and the United Kingdom Agusti, A., Calverley, P.M., Celli, B. et al. Characterisation of COPD heterogenecity in the ECLIPSE cohort. Respir Res. 2010; 11: 122 Ni, Y., Shi, G., Yu, Y. et al. Clinical characteristics of patients with chronic obstructive pulmonary disease with co-morbid bronchiectasis: a systematic review and meta-analysis. Int J Chron Obstruct Pulmon Dis. 2015; 10: 1465–1475 Kang HR, Choi GS, Park SJ, et al. The effects of bronchiectasis on asthma exacerbation. Tuberc Respir Dis (Seoul). 2014;77(5):209-214. |
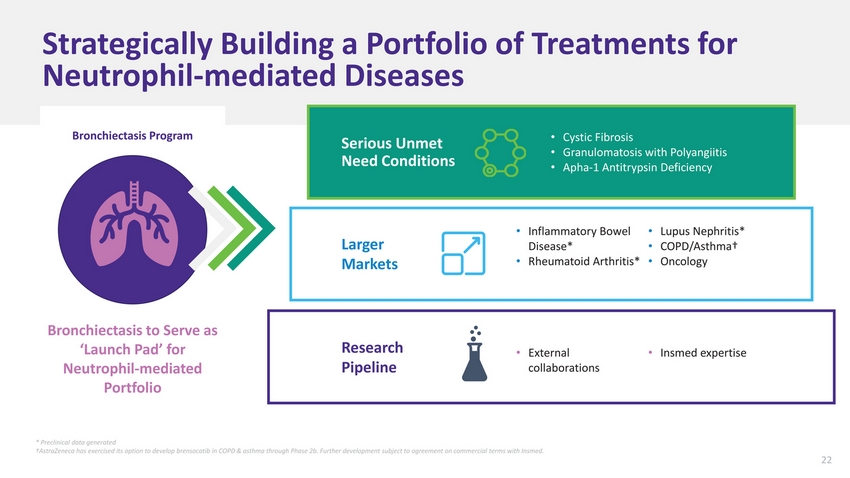 | Strategically Building a Portfolio of Treatments for Neutrophil-mediated Diseases Bronchiectasis Program Serious Unmet Need Conditions Cystic Fibrosis Granulomatosis with Polyangiitis Apha-1 Antitrypsin Deficiency Larger Markets Inflammatory Bowel Disease* Rheumatoid Arthritis* Lupus Nephritis* COPD/Asthma† Oncology Bronchiectasis to Serve as ‘Launch Pad’ for Neutrophil-mediated Portfolio Research Pipeline External collaborations Insmed expertise * Preclinical data generated †AstraZeneca has exercised its option to develop brensocatib in COPD & asthma through Phase 2b. Further development subject to agreement on commercial terms with Insmed. |
 | ARIKAYCE 23 Not for promotional use |
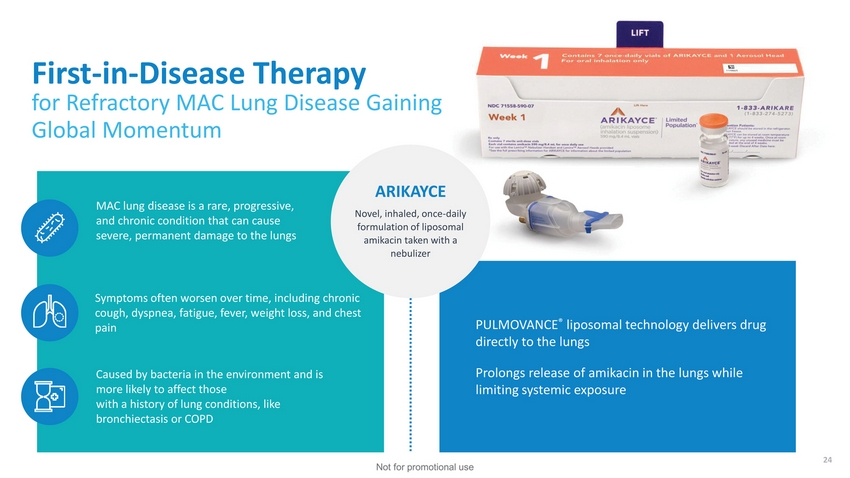 | First-in-Disease Therapy for Refractory MAC Lung Disease Gaining Global Momentum MAC lung disease is a rare, progressive, and chronic condition that can cause severe, permanent damage to the lungs ARIKAYCE Novel, inhaled, once-daily formulation of liposomal amikacin taken with a nebulizer Symptoms often worsen over time, including chronic cough, dyspnea, fatigue, fever, weight loss, and chest pain Caused by bacteria in the environment and is more likely to affect those with a history of lung conditions, like bronchiectasis or COPD PULMOVANCE® liposomal technology delivers drug directly to the lungs Prolongs release of amikacin in the lungs while limiting systemic exposure 24 Not for promotional use |
 | Building on the Success of the Launch with Recent Advancements International guideline inclusion with a strong recommendation for use (1) U.S. sNDA approval adding Culture Conversion Data Beyond 12 Months to Label Approved in Europe in late 2020 Planned expansion into Japan by mid-2021 European Launch is Underway Reimbursement achieved in Germany Netherlands Healthcare Assessment recommends reimbursement ARIKAYCE was one of the U.S. Top 10 most successful non-oncology rare disease launches (1) For adult patients with limited or no treatment options after six months of failed treatment. 25 Not for promotional use |
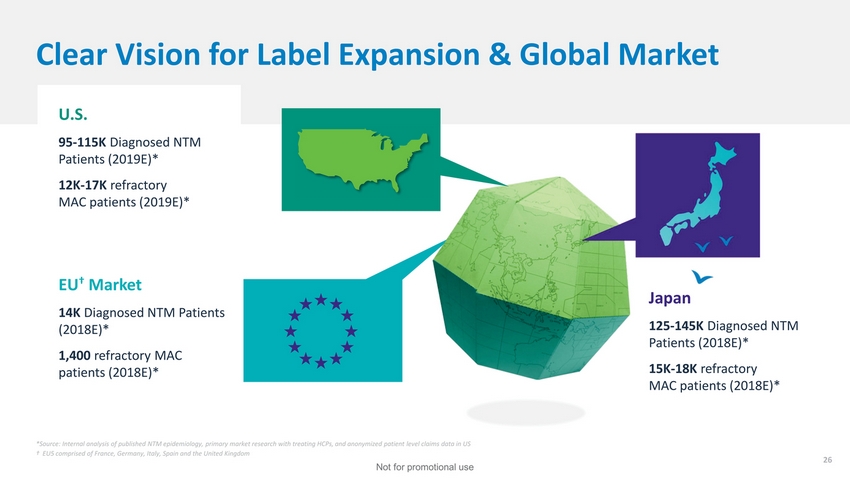 | Clear Vision for Label Expansion & Global Market U.S. 95-115K Diagnosed NTM Patients (2019E)* 12K-17K refractory MAC patients (2019E)* EU† Market 14K Diagnosed NTM Patients (2018E)* 1,400 refractory MAC patients (2018E)* Japan 125-145K Diagnosed NTM Patients (2018E)* 15K-18K refractory MAC patients (2018E)* *Source: Internal analysis of published NTM epidemiology, primary market research with treating HCPs, and anonymized patient level claims data in US † EU5 comprised of France, Germany, Italy, Spain and the United Kingdom 26 Not for promotional use |
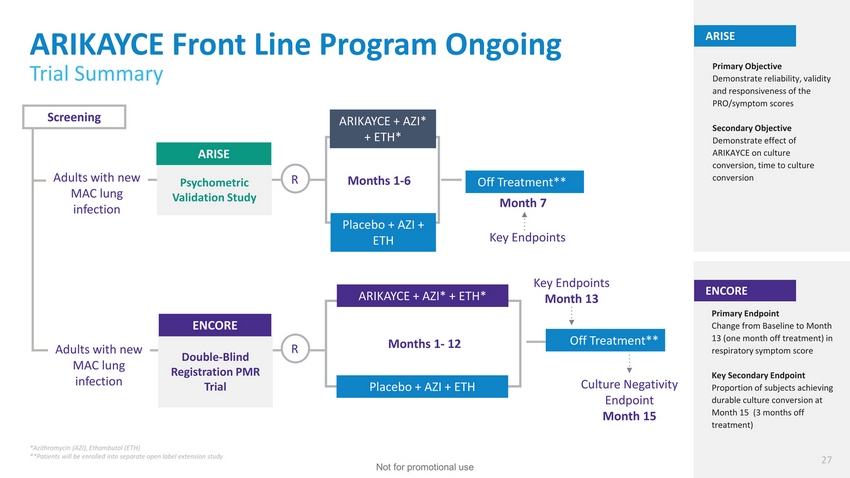 | ARIKAYCE Front Line Program Ongoing Trial Summary ARISE Primary Objective Demonstrate reliability, validity and responsiveness of the PRO/symptom scores Screening Adults with new ARISE Psychometric ARIKAYCE + AZI* + ETH* RMonths 1-6 Off Treatment** Secondary Objective Demonstrate effect of ARIKAYCE on culture conversion, time to culture conversion MAC lung infection Validation StudyMonth 7 Placebo + AZI + ETH Key Endpoints Adults with new MAC lung infection ENCORE Double-BlindR Registration PMR Trial ARIKAYCE + AZI* + ETH* Months 1-12 Placebo + AZI + ETH Key Endpoints Month 13 Off Treatment** Culture Negativity Endpoint Month 15 ENCORE Primary Endpoint Change from Baseline to Month 13 (one month off treatment) in respiratory symptom score Key Secondary Endpoint Proportion of subjects achieving durable culture conversion at Month 15 (3 months off treatment) *Azithromycin (AZI), Ethambutol (ETH) **Patients will be enrolled into separate open label extension study 27 Not for promotional use |
 | Financial Strength to Achieve Our Objectives $588.8M as of 9/30/20 102M* as of 9/30/20 Cash and Cash Equivalents Common Shares Outstanding *Excludes stock options, unvested RSUs and shares underlying outstanding convertible notes 28 |
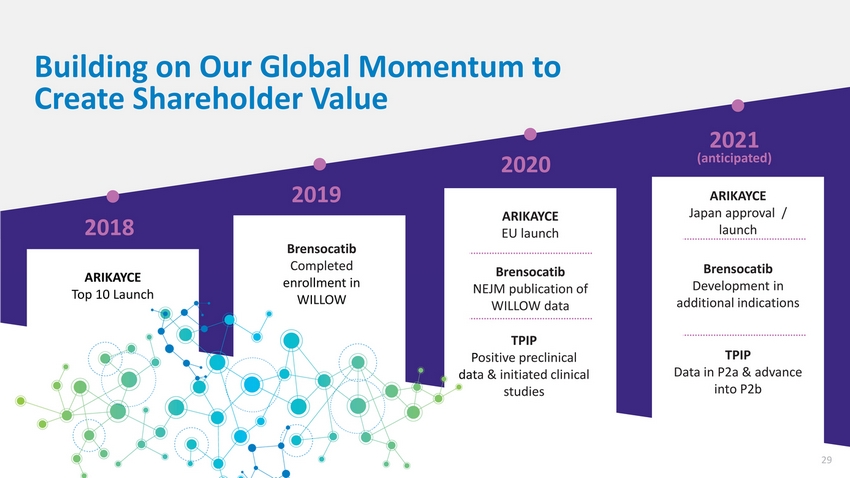 | Building on Our Global Momentum to Create Shareholder Value 2020 2019 ARIKAYCE 2021 (anticipated) ARIKAYCE Japan approval / 2018 ARIKAYCE Top 10 Launch Brensocatib Completed enrollment in WILLOW EU launch Brensocatib NEJM publication of WILLOW data TPIP Positive preclinical data & initiated clinical studies launch Brensocatib Development in additional indications TPIP Data in P2a & advance into P2b 29 |
 | Anticipated Near-Term Events Support All Three Pillars of Value Creation Strategy ARIKAYCE: Commercial Expansion And Label Extension EU Approval And Launch Frontline Program Initiation (ARISE / ENCORE) Japan Approval Japan Launch Brensocatib: Unlocking Options For Neutrophil-mediated Diseases ASPEN Trial Initiation (P3 Bronchiectasis) STOP-COVID19 Data Cystic Fibrosis PK Study Initiation 4Q 2020✓ YE 2020✓ 1Q 2021 Mid-2021 YE 2020✓ 1H 2021 Mid-2021 TPIP: Changing The Prostacyclin Paradigm Phase 1 Data (PK and safety) Phase 2a Trial Initiation Phase 2a Preliminary Data 1Q 2021 1Q 2021 2H 2021 0 30 |
 | We are committed to bringing forth technologies and medicines in therapeutic areas with the greatest potential to make a difference in patients’ lives. Investor Relations investor.relations@insmed.com 646-351-0954 Powered by Purpose 3131 |
 | Appendix 32 |
 | Robust Development Cycle Capabilities with Clear Commercialization Path Pipeline Highlight: Populations with Urgent Unmet Patient Needs Drug Discovery Research ARIKAYCE® Treprostinil Palmitil Inhalation Powder (TPIP) Clinical Development Management of the Regulatory Pathway Global Commercialization Business Development (Licensing and Acquisition) Brensocatib An advanced research and development pipeline has potential to address the unmet needs of patients with serious and rare diseases. Successfully brought a first-in-disease therapy independently from concept to market and has expertise in every stage of the development cycle. 33 |
 | Insmed Differentiated With ARIKAYCE, Brensocatib and TPIP, we have developed three value drivers, plus Robust Pipeline and Development Capabilities: Clinical Development Drug Discovery Research Business Development Management of the Regulatory Pathway Global Commercialization Strong expertise and potential across the development cycle. Many companies succeed at part of this equation. We do it all, well. 34 |
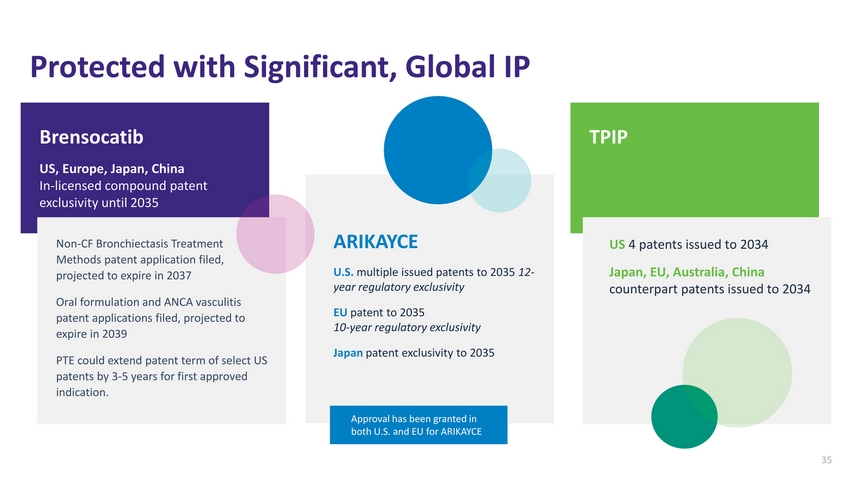 | Protected with Significant, Global IP Brensocatib US, Europe, Japan, China In-licensed compound patent exclusivity until 2035 TPIP Non-CF Bronchiectasis Treatment Methods patent application filed, projected to expire in 2037 Oral formulation and ANCA vasculitis patent applications filed, projected to expire in 2039 PTE could extend patent term of select US patents by 3-5 years for first approved indication. ARIKAYCE U.S. multiple issued patents to 2035 12-year regulatory exclusivity EU patent to 2035 10-year regulatory exclusivity Japan patent exclusivity to 2035 US 4 patents issued to 2034 Japan, EU, Australia, China counterpart patents issued to 2034 Approval has been granted in both U.S. and EU for ARIKAYCE 35 |
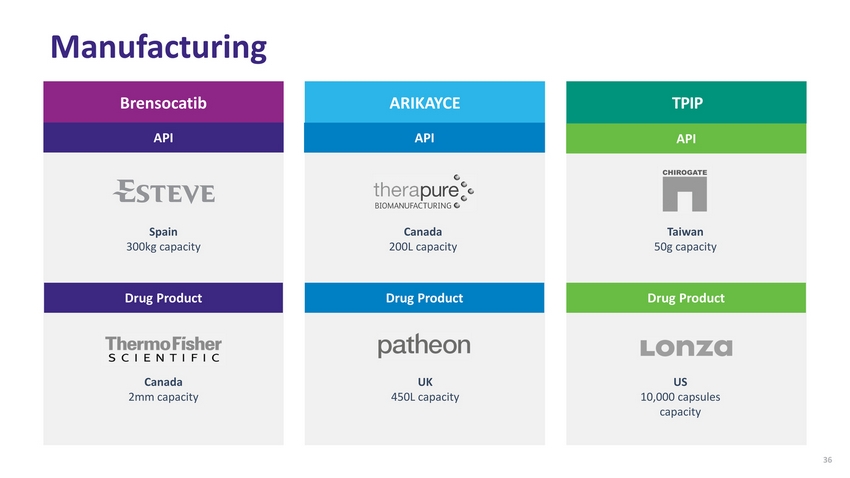 | Manufacturing Brensocatib ARIKAYCE TPIP API API API Spain 300kg capacity Canada 200L capacity Taiwan 50g capacity Drug Product Drug Product Drug Product Canada 2mm capacity UK 450L capacity US 10,000 capsules capacity 36 |
 | W I L L O W S T U D Y• Data Demonstrate Potential for First-in-Class Bronchiectasis Treatment Randomized, double-blind, Phase 2 study; tested 10 mg, 25 mg and placebo CT scan-confirmed NCFBE; ≥2 pulmonary exacerbations in past 12 months Risk of exacerbation over six months reduced by 42% for 10 mg group, 38% for 25 mg group Primary and secondary endpoints met with statistical significance Data demonstrate positive correlation between neutrophil elastase reduction and risk of exacerbation in 10mg group ASPEN Phase 3 program initiated in late 2020 AstraZeneca Exercises First Option in License Agreement Validating Potential for COPD and Asthma 2020: PRIME Designation (EMA) & Breakthrough Therapy Designation (FDA) Granted |
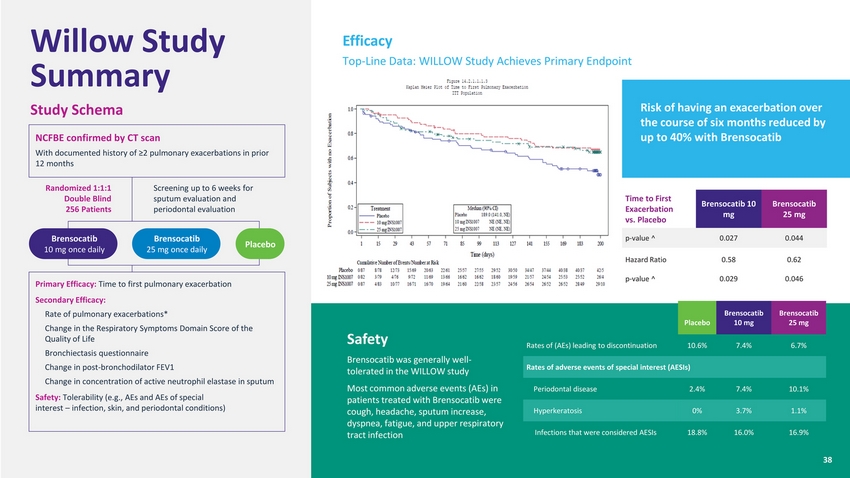 | Willow Study Summary Study Schema NCFBE confirmed by CT scan With documented history of ≥2 pulmonary exacerbations in prior 12 months Efficacy Top-Line Data: WILLOW Study Achieves Primary Endpoint Risk of having an exacerbation over the course of six months reduced by up to 40% with Brensocatib Randomized 1:1:1 Double Blind 256 Patients Screening up to 6 weeks for sputum evaluation and periodontal evaluation Time to First Exacerbation vs. Placebo Brensocatib 10 mg Brensocatib 25 mg Brensocatib 10 mg once daily Brensocatib 25 mg once dailyPlacebo p-value ^0.0270.044 Primary Efficacy: Time to first pulmonary exacerbation Secondary Efficacy: Rate of pulmonary exacerbations* Change in the Respiratory Symptoms Domain Score of the Quality of Life Bronchiectasis questionnaire Change in post-bronchodilator FEV1 Change in concentration of active neutrophil elastase in sputum Safety: Tolerability (e.g., AEs and AEs of special interest – infection, skin, and periodontal conditions) Safety Brensocatib was generally well-tolerated in the WILLOW study Most common adverse events (AEs) in patients treated with Brensocatib were cough, headache, sputum increase, dyspnea, fatigue, and upper respiratory tract infection Hazard Ratio Placebo Brensocatib 10 mg Brensocatib 25 mg Rates of (AEs) leading to discontinuation 10.6% 7.4% 6.7% Rates of adverse events of special interest (AESIs) Periodontal disease 2.4% 7.4% 10.1% Hyperkeratosis 0% 3.7% 1.1% Infections that were considered AESIs 18.8% 16.0% 16.9% 0.58 0.029 0.62 0.046 |
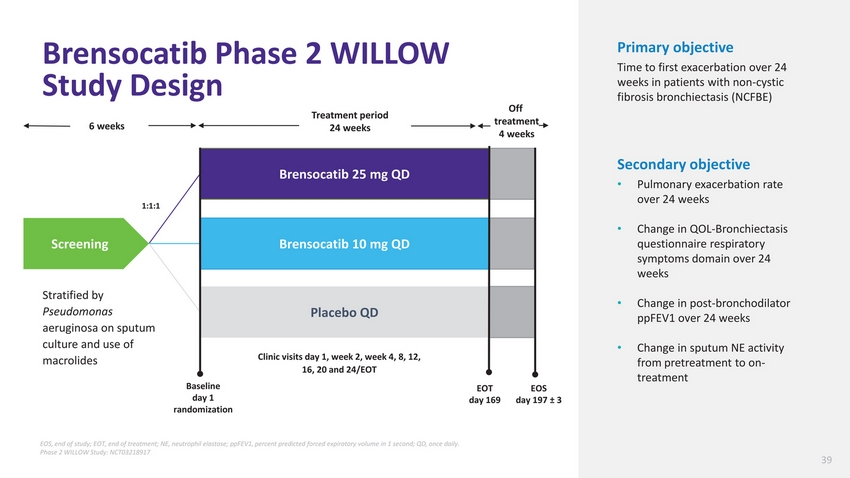 | Brensocatib Phase 2 WILLOW Study Design Treatment period Off treatment Primary objective Time to first exacerbation over 24 weeks in patients with non-cystic fibrosis bronchiectasis (NCFBE) 6 weeks 24 weeks 4 weeks Screening Stratified by Pseudomonas 1:1:1 Brensocatib 25 mg QD Brensocatib 10 mg QD Placebo QD Secondary objective Pulmonary exacerbation rate over 24 weeks Change in QOL-Bronchiectasis questionnaire respiratory symptoms domain over 24 weeks Change in post-bronchodilator ppFEV1 over 24 weeks aeruginosa on sputum culture and use of macrolides Baseline day 1 randomization Clinic visits day 1, week 2, week 4, 8, 12, 16, 20 and 24/EOT EOT day 169 EOS day 197 ± 3 Change in sputum NE activity from pretreatment to on-treatment EOS, end of study; EOT, end of treatment; NE, neutrophil elastase; ppFEV1, percent predicted forced expiratory volume in 1 second; QD, once daily. Phase 2 WILLOW Study: NCT03218917 39 |
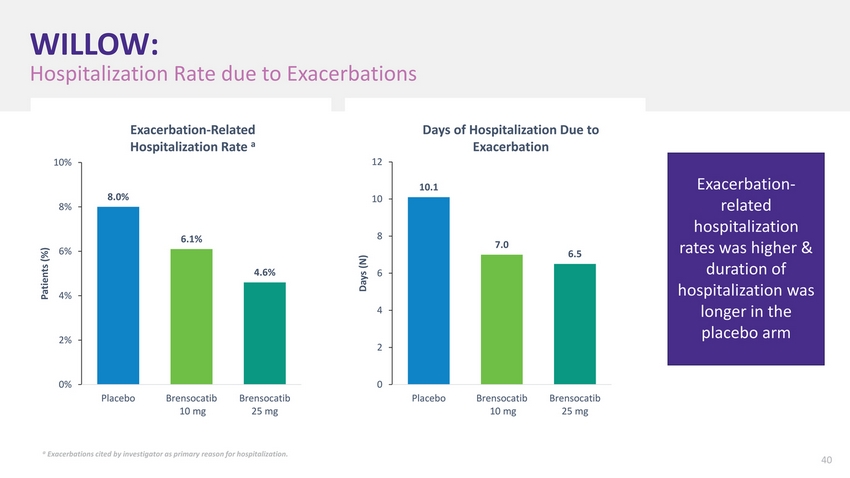 | WILLOW: Hospitalization Rate due to Exacerbations 10% 8% Patients (%) 4% 2% 8.0% Exacerbation-Related Hospitalization Rate a 6.1% 4.6% Days of Hospitalization Due to Exacerbation 12 10.1 10 8 7.0 Days (N) 6 4 2 Exacerbation-related hospitalization rates was higher & duration of hospitalization was longer in the placebo arm 0% PlaceboBrensocatib 10 mg Brensocatib 25 mg 0 PlaceboBrensocatib 10 mg Brensocatib 25 mg a Exacerbations cited by investigator as primary reason for hospitalization. 40 |
 | 25 mg brensocatib vs. placebo Rate of Exacerbations by Patient Subgroups 10 mg brensocatib vs. placebo Chalmers et al, ERS International Congress 2020, 7-9 September. RCT4135 BSI, bronchiectasis severity index; FEV1, forced expiratory volume in 1 second aBulgaria/Poland 41 |
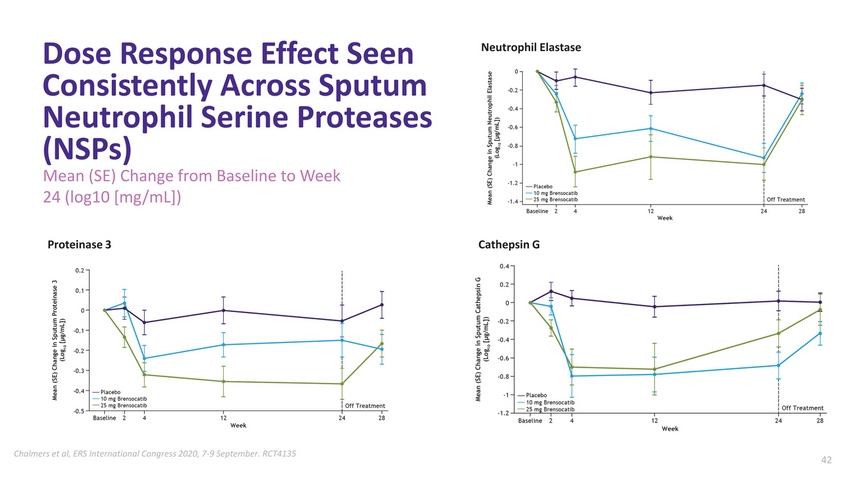 | Dose Response Effect Seen Consistently Across Sputum Neutrophil Serine Proteases (NSPs) Mean (SE) Change from Baseline to Week 24 (log10 [mg/mL]) Neutrophil Elastase Proteinase 3Cathepsin G Chalmers et al, ERS International Congress 2020, 7-9 September. RCT413542 |
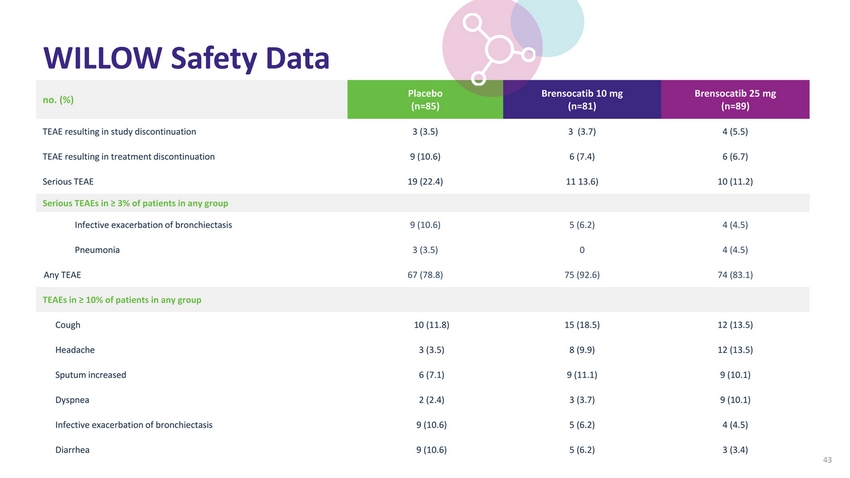 | WILLOW Safety Data no. (%)Placebo (n=85) Brensocatib 10 mg (n=81) Brensocatib 25 mg (n=89) TEAE resulting in study discontinuation3 (3.5)3 (3.7)4 (5.5) TEAE resulting in treatment discontinuation9 (10.6)6 (7.4)6 (6.7) Serious TEAE19 (22.4)11 13.6)10 (11.2) Serious TEAEs in ≥ 3% of patients in any group |
 | ASPEN Study: Phase 3 Pivotal Program Ongoing Single Trial Designed to Support Global Filing Breakthrough Therapy Designation & PRIME designation granted for brensocatib in non-cystic fibrosis bronchiectasis (NCFBE) Robust efficacy endpoints, supported by WILLOW results Integrates feedback from Health Authorities 44 |
 | ASPEN Timeline Considerations 40 Countries ~480 sites 6 Fast Track Countries Targeting WILLOW study sites 45 |
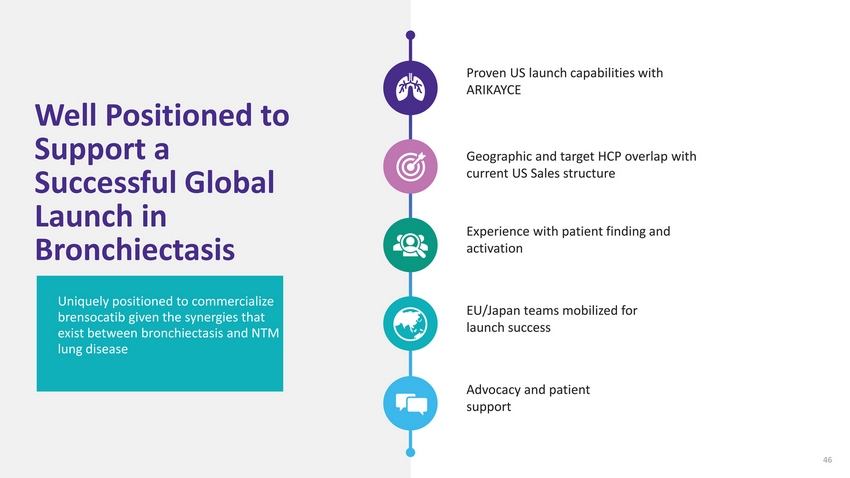 | Well Positioned to Support a Successful Global Launch in Bronchiectasis Uniquely positioned to commercialize brensocatib given the synergies that exist between bronchiectasis and NTM lung disease Proven US launch capabilities with ARIKAYCE Geographic and target HCP overlap with current US Sales structure Experience with patient finding and activation EU/Japan teams mobilized for launch success Advocacy and patient support 46 |
 | ARIKAYCE Treats Refractory MAC Lung Disease Potential Progression Toward Use as Front-line and/or Maintenance Therapy 12-17 K refractory US: 100K patients Approved Treatment in MAC Lung Disease First and only therapy approved in the United States as part of a combination antibacterial drug regimen for adult patients with limited or no alternative treatment options Received both breakthrough therapy & orphan drug designations Label Expansion in Progress Action Front-line Phase 3 underway Opportunity Successful data and subsequent regulatory approval could result in a 5-fold increase in the addressable market 47 |
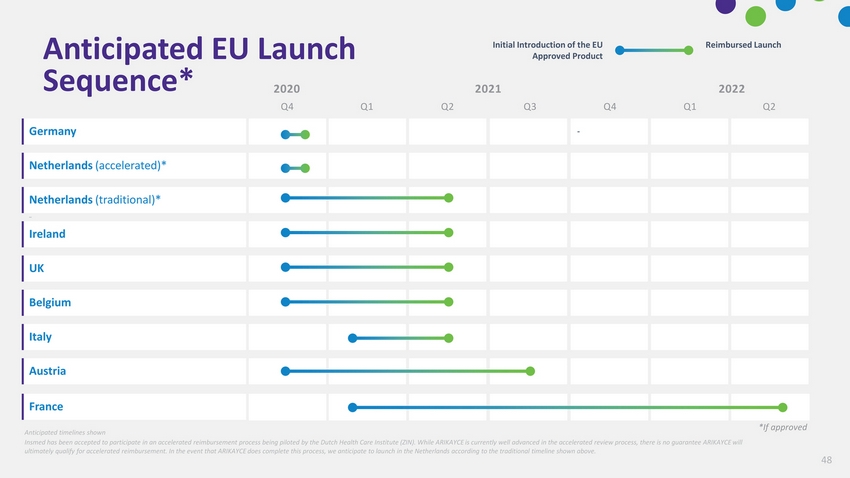 | Germany Ireland Anticipated timelines shown Insmed has been accepted to participate in an accelerated reimbursement process being piloted by the Dutch Health Care Institute (ZIN). While ARIKAYCE is currently well advanced in the accelerated review process, there is no guarantee ARIKAYCE will ultimately qualify for accelerated reimbursement. In the event that ARIKAYCE does complete this process, we anticipate to launch in the Netherlands according to the traditional timeline shown above. *If approved |
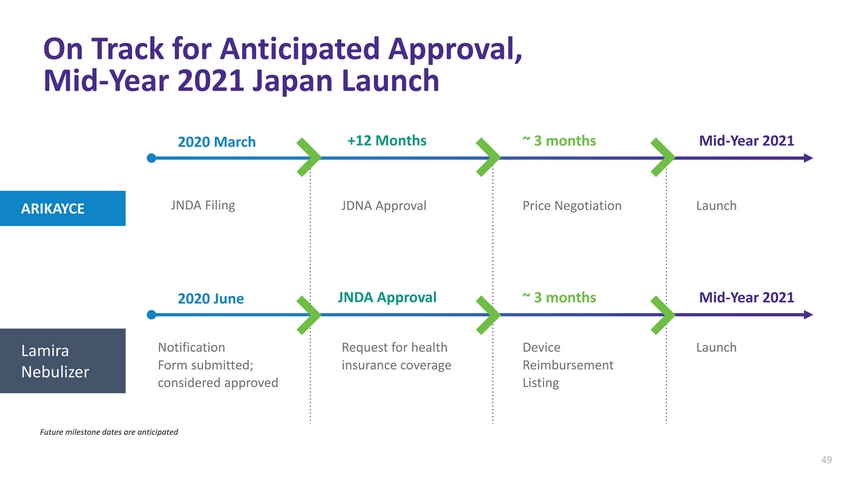 | On Track for Anticipated Approval, Mid-Year 2021 Japan Launch 2020 March+12 Months~ 3 monthsMid-Year 2021 ARIKAYCE JNDA Filing JDNA ApprovalPrice NegotiationLaunch 2020 JuneJNDA Approval~ 3 monthsMid-Year 2021 Lamira Nebulizer Notification Form submitted; considered approved Request for health insurance coverage Device Reimbursement Listing Launch Future milestone dates are anticipated |
 | Pulmonary 3. 1. Hypoxemia Hypertension: Diagnostic classification Idiopathic PAH (IPAH) Heritable BMPR2 ALK1, ENG, SMAD9, CAV1, KCNK3 1.2.3. Unknown Drug-and toxin-induced Associated with Connective tissue diseases HIV infection Portal hypertension Congenital heart diseases Schistosomiasis 1’ Pulmonary veno-occlusive disease and / or pulmonary capillary hemangiomatosis 1’ Persistent pulmonary hypertension of the newborn 2. left heart disease Left ventricular systolic dysfunction Left ventricular diastolic dysfunction Valvular disease Congenital/acquired left heart inflow/ outflow tract obstruction and congenital cardiomyopathies Chronic obstructive pulmonary disease Interstitial lung disease Other pulmonary diseases with mixed restrictive and obstructive pattern Sleep-disordered breathing Alveolar hypoventilation disorders Chronic exposure to high altitude Developmental abnormalities 4. hypertension (CTEPH) 5. Hematologic disorders: chronic hemolytic anemia, myeloproliferative disorders, splenectomy Systemic disorders: sarcoidosis, pulmonary histiocytosis: lymphangioleiomyomatosis Metabolic disorders: glycogen storage disease, Gaucher disease, thyroid disorders Others: tumoral obstruction, fibrosing mediastinitis, chronic renal failure, segmental PH |
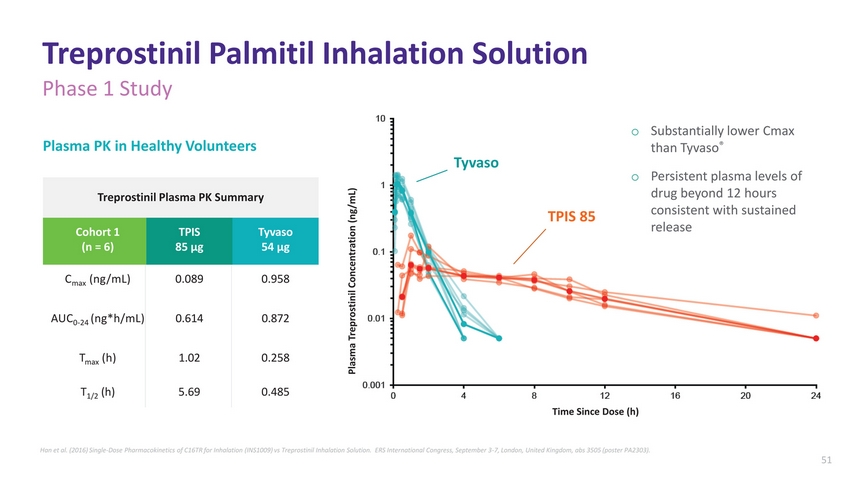 | Treprostinil Palmitil Inhalation Solution Phase 1 Study Plasma PK in Healthy Volunteers Plasma Treprostinil Concentration (ng/mL) TPIS 85 Substantially lower Cmax than Tyvaso® Persistent plasma levels of drug beyond 12 hours consistent with sustained release Treprostinil Plasma PK Summary Cohort 1 (n = 6) TPIS 85 µg Tyvaso 54 µg Cmax (ng/mL) 0.089 0.958 AUC0-24 (ng*h/mL) 0.614 0.872 Tmax (h) 1.02 0.258 T1/2 (h) 5.69 0.485 Han et al. (2016) Single-Dose Pharmacokinetics of C16TR for Inhalation (INS1009) vs Treprostinil Inhalation Solution. ERS International Congress, September 3-7, London, United Kingdom, abs 3505 (poster PA2303). |