UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
☒Annual Report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934:
For the fiscal year ended: December 31, 2019
☐Transition report pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934:
For the transition period from:
| XG SCIENCES, INC. | ||||
| (Exact name of registrant as specified in its charter) |
| Michigan | 333-209131 | 20-4998896 | ||
| (State or other jurisdiction of incorporation or organization) | (Commission File No.) | (I.R.S. Employer Identification No.) |
3101 Grand Oak Drive
Lansing, MI 48911
(Address of principal executive offices) (zip code)
(517) 703-1110
(Issuer Telephone number)
Securities registered under Section 12(b) of the Act: None.
Securities registered under Section 12(g) of the Act: None.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Exchange Act. Yes☐ No☒
Indicate by check mark whether the registrant (1) filed all reports required to be filed by Section 13 or 15(d) of the Exchange Act during the past 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes☒ No☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T during the preceding 12 months (or for such shorter period that the registrant was required to submit and post such files). Yes☒ No☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act): Yes☐ No☒
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or a smaller reporting company, and whether the registrant is an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
| Large accelerated filer ☐ | Accelerated filer ☐ |
| Non-accelerated filer ☐ | Smaller reporting company ☒ |
Emerging growth company☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.☒
As of December 31, 2019, the aggregate market value of the registrant’s common stock held by non-affiliates of the registrant was approximately $25,359,328, based on the price at which the common equity was last sold (i.e., $8.00 per share).
The number of shares outstanding of the registrant’s common stock, no par value per share, as of April 29, 2020 was 4,024,443.
TABLE OF CONTENTS
Note Regarding Forward-Looking Statements:
The information in this Annual Report on Form 10-K contains “forward-looking statements” and information within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”) relating to XG Sciences, Inc., a Michigan corporation and its subsidiary, XG Sciences IP, LLC, a Michigan corporation (collectively referred to as “we”, “us”, “our”, “XG Sciences”, “XGS”, or the “Company”), which are subject to the “safe harbor” created by those sections. These forward-looking statements include, but are not limited to, statements concerning our strategy, future operations, future financial position, future revenues, projected costs, prospects and plans and objectives of management. The words “anticipates,” “believes,” “estimates,” “expects,” “intends,” “may,” “plans,” “projects,” “will,” “would” and similar expressions are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. We may not actually achieve the plans, intentions or expectations disclosed in our forward-looking statements and you should not place undue reliance on our forward-looking statements.
These forward-looking statements involve known and unknown risks and uncertainties that could cause our actual results, performance or achievements to differ materially from those expressed or implied by the forward-looking statements, including, without limitation, the risks set forth beginning on page 13 under the section entitled “Risk Factors” herein.
XG Sciences was formed in May 2006 for the purpose of commercializing certain technology to produce graphene nanoplatelets and integrated, value-added products containing graphene nanoplatelets. First isolated and characterized in 2004, graphene is a single layer of carbon atoms configured in an atomic-scale honeycomb lattice. Among many noted properties, monolayer graphene is harder than diamonds, lighter than steel but significantly stronger, and conducts electricity better than copper. Graphene nanoplatelets are particles consisting of multiple layers of graphene. Graphene nanoplatelets have unique capabilities for energy storage, thermal conductivity, electrical conductivity, barrier properties, lubricity and the ability to impart physical property improvements when incorporated into plastics, metals or other matrices.
We believe the unique properties of graphene and graphene nanoplatelets will enable numerous new product applications and the market for such products will quickly grow to be a significant market opportunity. Our business model is to design, manufacture and sell advanced materials we call xGnP®graphene nanoplatelets and value-added products incorporating xGnP® graphene nanoplatelets. We currently have hundreds of customers trialing our products for numerous applications, including, but not limited to lithium ion batteries, lead acid batteries, thermally conductive adhesives, composites, thermal management and heat transfer, inks and coatings, printed electronics, construction materials, cement, 3-D printing and in a range of other industrial uses. We believe our proprietary processes have enabled us to be a low-cost producer of high-quality, graphene nanoplatelets and value-added integrated products containing graphene nanoplatelets and that we are well positioned to address a wide range of end-use applications.
1
Our Customers
We sell products to customers around the world and have sold materials to over 1,300 customers in 47 countries since 2008. Some of these customers are research organizations and some are commercial organizations. Our customers have included well-known automotive and OEM suppliers around the world (Ford, Johnson Controls, Magna, Honda Engineering), world-scale lithium ion battery manufacturers in the US, South Korea and China (Samsung SDI, LG Chemical, Lishen, A123) , diverse specialty material companies (3M, BASF, Henkel, Dow Chemical, DuPont) and leading research centers such as Lawrence Livermore National Laboratory and Oakridge National Laboratory. The majority of our customers are still ordering in small quantities consistent with their development and qualification work, although others have started to order in 10’s to 100’s of kilograms and some at the metric ton level.
We have also licensed some of our base manufacturing technology to other companies. Our licensees include POSCO, the fifth largest steel manufacturer in the world by 2018 tonnage output, and Cabot Corporation (“Cabot”), a leading global specialty chemicals and performance materials company. These licensees further extend our technology through their customer networks. Ultimately, we believe we may benefit in terms of royalties on sales of xGnP® graphene nanoplatelets produced and sold by our licensees.
Our Products
XG Sciences is a manufacturer of graphene nanoplatelets and value-added products that contain graphene nanoplatelets. The term “graphene” is used widely in the literature and the popular press to cover a variety of specific forms of the material. We generally think about two broad classes of graphene materials:
| 1. | One-atom thick films of carbon commonly referred to as monolayer graphene, manufactured typically from gases by assembling molecules to form relatively large, transparent sheets of material. These materials have been characterized by their performance attributes that differentiate them from other advanced materials and that may include: 200 times stronger than steel, flexible and able to stretch up to 25% of its original length, optically transparent, more electrically conductive than copper, more thermally conductive than any other known material and atomic-level barrier properties. XG Sciences does not manufacture these films and does not participate in the markets for these films and believes that, in general, the markets for these films do not compete with those for graphene nanoplatelets. |
| 2. | Ultra-thin particles of carbon that consist of layers of graphene sheets ranging in thickness from a few layers to many layers – that are commonly referred to as graphene nanoplatelets (“GNP” or “GNPs”). Because GNPs are thin and can be manufactured in a range of diameters, they are useful for a wide variety of applications. XG Sciences manufactures GNPs that range in thickness from a few nanometers and up to 10-20 nanometers and with diameters ranging from less than 1 micron and up to 100 microns. The manufacture of these graphene particles is our main area of expertise, and their use in practical applications is the focus of our sales, marketing and development activities. |
2
The well-publicized isolation and characterization of graphene in 2004 at the University of Manchester, has spawned a new class of 2D materials based on layers of carbon atoms arranged in a hexagonal array and each carbon having lone pair electrons. The unique characterization and related performance of this new class of materials is derived from their two-dimensional nature and their composition of sp2carbon atoms arranged in a hexagonal array. The ability of any new material to be exploited in industrial applications will depend on its fit-for-performance. In the case of graphene nanoplatelets, the fit-for-performance is very much related to their aspect ratio (among other factors) in that the diameter is typically significantly greater than the thickness. This is what differentiates the material from bulk graphite of high crystallinity and purity. We classify nanoplatelets consisting of largely basal planes of carbon atoms packed in a hexagonal array (i.e., graphene) as graphene nanoplatelets so long as their aspect ratio may be classified as two-dimension and are thus in the form of platelets. Such a definition implies that the thickness is nanoscale – GNPs having a thickness in the range from generally 0.6 nanometers and up to many 10’s of nanometers. We have chosen to utilize the definitions as set out by the Carbon Journal editorial team (Carbon, volume 65, pp.1-6) and Fullerex (Bulk Graphene Pricing Report, 2019) which provides classification for the various material types which provide meaningful descriptions of commercially available graphene.
Graphene Product Definitions Based on Layer Thickness
| Number of Layers | Product Description | |
| 1 | Graphene (monolayer) | |
| 1-3 | Very Few Layer Graphene (vFLG) | |
| 2-5 | Few Layer Graphene (FLG) | |
| 2-10 | Multilayer Graphene (MLG) | |
| >10 | Graphene Nanoplatelets (GNPs) |
Bulk Materials.
We sell bulk materials under the trademarked brand name of xGnP®graphene nanoplatelets. These materials are produced in various grades, which are analogous to average platelet thickness, and average platelet diameters. There are three commercial grades (Grades H, M & R), each of which is offered in three standard platelet diameters and a fourth, C Grade, which is offered in three standard surface areas. We also have access to other development grades (Grade T, for example), but which are not yet made available commercially and have been used internally for those products containing graphene nanoplatelets. These bulk materials, which normally ship in the form of a dry powder, are especially applicable for use as additives in polymeric or metallic composites, or in coatings or other formulations where particular electrical, thermal or barrier performance are desired by our customers. We also offer our material in the form of dispersions of nanoplatelets in liquids such as water, alcohol, or organic solvents, or mixed into resins or polymers such as thermoplastics or thermosets. We use two different commercial processes to produce these bulk materials:
| Grade H/M/R/T materials are produced through chemical intercalation of natural graphite followed by thermal exfoliation using a proprietary process developed by us. The “grade” designates the thickness and surface characteristics of the material, and each grade is available in various average particle/platelet diameters. Surface area, calculated by the Brunauer, Emmet, and Teller (BET) Method, is used as a convenient proxy for thickness, so each grade of products produced through chemical intercalation is designated by its average surface area, which ranges from 50 to 150 m2/gram of material. We are able to extend the surface area higher (250 m2/gram for T Grade) but are not yet producing these materials in metric-ton quantities. As the market need emerges for this class of materials, we anticipate scaling them. For example, we introduced a new Grade of xGnP® powders, R-Grade, with improved electrical conductivity targeting use in applications for electrically and thermally conductive ink and composites and have scaled R-Grade to metric-ton quantities. |
| Grade C materials are produced through a high-shear mechanical exfoliation using a proprietary process and equipment that we invented, designed, constructed and patented. The Grade C materials are smaller platelet diameters than those grades produced through chemical exfoliation, and Grade C materials are designated by their BET surface area, which ranges from 300 to 800 m2/gram. We are able to produce C-Grade materials having other surface areas and may make those available commercially as needed by our customers. |
The following graphic depicts xGnP®graphene nanoplatelets as a function of both layer thickness and aspect ratio (thickness by diameter), two key parameters which will influence their performance in a range of industrial applications. The performance of graphene nanoplatelets in a specific application is primarily a function of the platelet’s diameter, thickness, planarity (or more broadly – morphology), and, to some extent, the nature and concentration of any chemical groups on the platelets. There are other factors that may impact performance, such as optimized dispersion, product form delivered to the customer (powder vs. slurry), and so on. However, for the most part, performance in an application is related to the physical characteristics of the nanoplatelets. XG Sciences is skilled in the design and manufacture of graphene nanoplatelets and our two proprietary manufacturing processes allow for the production of a broad product portfolio varying across a broad range of both thickness and diameter, the two variables most likely related to performance in a range of end-use applications.
3
XG Sciences’ Graphene Nanoplatelet Product Portfolio and Versus Graphite
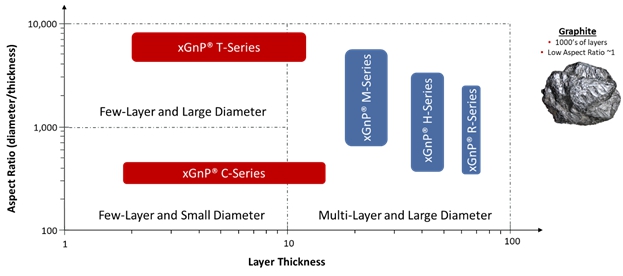
We believe we are a “platform play” in advanced materials, because our proprietary processes allow us to produce varying grades of graphene nanoplatelets that may be mapped to a variety of applications and in many end-use market segments. While we supply bulk materials consisting of various forms of xGnP® graphene nanoplatelets and in the form of dispersions of nanoplatelets in liquids such as water, alcohol or organic solvents, we also produce products that contain graphene nanoplatelets which we refer to as integrated products. We have done this to further differentiate our products from those potential suppliers offering only graphene nanoplatelets in powder form.
Composites. Our composites, integrated-products portfolio includes resins derived from a range of thermoplastics and thermosets into which our xGnP® graphene nanoplatelets are compounded. These products are supplied as master batches of resins with graphene nanoplatelets pre-dispersed such that their performance has been optimized for use in various end-use applications that may include industrial, automotive and sporting goods and which have demonstrated efficacy in standard injection molding, compression molding, blow molding and 3-D processes, to name but a few. Our current product portfolio of polymer resins containing various forms of our xGnP®graphene nanoplatelets and in varying concentration includes polyurethane (XGPU), polypropylene (XGPP), polyethylene terephthalate (XGPET), vinyl ester (XGVE), polyetherimide (XGPEI) and high-density polyethylene (XGHDPE). Other polymers may be added over time depending on the end-market and customer needs. In addition, we offer various bulk materials with demonstrated efficacy in plastic composites to impart improved physical performance to such matrices, which may be supplied as dry powders or as aqueous-based or solvent-based dispersions or cakes, as described previously. We have also targeted use of our graphene nanoplatelets as an additive in cement mixtures which we believe results in improved barrier resistance, durability, toughness and corrosion protection. Our GNP Concrete Additive product promotes the formation of more uniform and smaller-grain cement structure, improving both flexural and compressive strength. In addition, the embedded graphene nanoplatelets will stop cracks from forming and retard crack propagation, should any cracks form – the combination of which will further improve cement lifetime and durability.
Inks and Coatings. These consist of specially formulated dispersions of xGnP®together with solvents, binders, and other additives to make electrically or thermally conductive products designed for printing or coating and which are showing promise in diverse customer applications such as advanced packaging, electrostatic dissipation and thermal management. We also offer a set of standardized ink formulations suitable for printing. These inks offer the capability to print electrical circuits or antennas and may be suitable for other electrical or thermal applications and can be further customized for specific customer requirements.
Energy Storage Materials. These consist of specialty advanced materials that have been formulated for specific applications in the energy storage segment. We offer various bulk materials for use as conductive additives for cathodes and anodes in lithium-ion batteries, as additives to anode slurries for lead-carbon batteries, as a component in coatings for current collectors used in lithium-ion batteries and we are investigating the use of our materials as part of other energy-storage components.
4
Thermal Management Materials. These consist mainly of various thermal interface materials (“TIM’s”) in the form of custom greases or pastes. Our custom thermal interface materials are also designed to be used in various high temperature environments. Additionally, we offer various bulk materials for use as active components in adhesives, liquids, coatings and plastic composites to impart improved thermal management performance to such matrices.
Our Focus Areas
We prioritize our efforts in specific areas and with specific customers that we believe represent opportunities for either relatively near-term revenue or especially large and attractive markets. At this time, we are focused on four key vertical markets: Automotive, Sporting Goods, Packaging and Industrial. The following graphic provides examples of target applications within each of the four key verticals where XG Sciences has either commercial sales or is in development with one or more customers.
XGS Market/Application Focus Areas

Addressable Markets
The markets for our materials are large and growing. As one example, the 2019 North American packaging market for plastic bottles and containers was estimated to be more than $34 billion (Mordor Intelligence). Further, Mordor estimated the 2019 global market for PET water bottles at 543.8 billion units. XG Sciences is engaged with customers in North America targeting the use of xGnP® graphene nanoplatelets in a range of packaging applications, including water bottles, and we are expanding our market activities into other geographies. If each water bottle produced in 2019 were to incorporate just 1 milligram of xGnP®graphene nanoplates, the total revenue available to XG Sciences may range from $200 to $300 million, depending on product form. A second example is for use in tires where graphene-based products may bring significant advancements. A typical tire is a composite of several parts: sidewalls, inner liner and the tire tread - which itself is a rubber component comprising a steel chord reinforcement of a particulate-reinforced rubber network. If one considers that the tire tread is the only part of the vehicle’s contact with the road, it becomes apparent that all sources of energy dissipation are via the tread material and will have a major influence on total performance such as handling, fuel efficiency and braking performance. Data are starting to show that incorporation of graphene nanoplatelets into the production of a tire tread can significantly impact the rolling resistance and abrasion resistance, both of which will result in improved fuel efficiency and tire lifetime. These performance attributes also seem to come with improvements in such properties as tear strength and tensile performance, allowing formulators to target a wide range of passenger vehicle, truck and off-road applications. Based on data from the European Rubber Journal (January 9, 2019 and according the International Rubber Study Group), the 2019 global rubber market used in tires is estimated at 30 million metric tons. If graphene is used in only 20% of tires and adopted at 1 weight percent based on the rubber content, then the demand could easily exceed 60,000 metric tons for graphene-related materials. In a third example, there is an industry-wide need to continue to drive light weighting in vehicles. The primary objective is to save on fuel consumption for cars still using internal combustion engines, but also to extend drive time for emerging electrical vehicles. Data from the Department of Energy (“Materials Technologies: Goals, Strategies, and Top Accomplishments”, Department of Energy, Vehicle Technologies Program, August 2010) indicate that in 1977, conventional steel comprised 75% of the materials used in the manufacture of an auto. By 2010, that number was reduced to about 65% with a significant increase in high-strength steel, aluminum and polymer composites. In order to meet the industry light-weighting targets, it is estimated that by 2035, polymer composite usage will increase to 20% of all materials. Improvements to today’s composite materials are needed for this to occur and graphene nanoplatelets are a good candidate to enable the broad adoption of polymer composites for use in automotive manufacture. In 2018, 71 million cars were produced globally (Estimated Worldwide automobile production, Statista.com). If we assume an average car weights 1500 kilograms (DOE Lightweighting Efforts for Sustainable Transportation”,2nd Lightweighting summit, March 5, 2015) and based on DOE estimates (“Materials Technologies: Goals, Strategies, and Top Accomplishments”, Department of Energy, Vehicle Technologies Program, August 2010), approximately 100 kilograms is comprised of polymers/composites (and certainly the potential for more in the future), and if graphene-related products were to be adopted at just 1 weight percent, then there is the potential demand of over 71,000 metric tons for graphene-related materials.
5
Commercialization Process
Because graphene is a new material, most of our customers are still developing applications that use our products. Commercialization is a process, the exact timing of which is often difficult to predict. It starts with our own internal R&D to validate performance for an identified market or customer-specific need. Our customers then validate the performance of our materials and determine whether our products can be incorporated into their manufacturing processes. This is initially done at pilot production scale levels. Our customers then introduce products incorporating our materials to their own customers to further validate performance. After their customers have validated performance, our customers will then move to commercial scale production. Every customer goes through the same process, but will do so at varying speeds, depending on the customer, the product application and the end-use market. Thus, we are not always able to predict when our customers will begin ordering commercial volumes of our materials or predict their expected volumes over time. However, as customers move through the process, we generally receive feedback and gain greater insights regarding their commercialization plans. According to our respective customers, the following are just some examples of where our products are providing value to our customers at levels that are either in commercial production or we believe will warrant their use on a commercial basis:
| ● | In 2018, Callaway Golf Company introduced new dual-core Chrome Soft and Chrome Soft X golf balls incorporating our xGnP®graphene nanoplatelets into the outer core, resulting in a new class of golf ball that enables higher driving speeds, greater distance and increased control, which is allowing Callaway to command a premium price for their golf balls in the marketplace and in 2019 expanded the technology to incorporate our xGnP® into the E·R·C Soft line of golf balls; and |
| ● | Ford Motor Company for polyurethane based foams for use in Ford and Lincoln light truck and passenger car, demonstrating a 17 percent enhancement in Noise, Vibration and Harshness (NVH) sound absorption, a 20 percent improvement in mechanical properties and a 30 percent improvement in heat endurance properties, compared with that of the foam used without graphene; and |
| ● | Light emitting diode module and product company demonstrated approximately 50% improvement in thermal management capability when compared to existing commercial thermal management products, translating into a 15% improvement in thermal management at the device level; and |
| ● | Lead acid battery manufacturers incorporating our materials in the commercial supply of batteries demonstrating improvements in measured cycle life, capacity and charge acceptance; and |
| ● | U.S. bottling company adopting commercial use of our graphene nanoplatelets in PET water bottles to improve modulus (10% with minimal affect to color and up to 200% with color change), shelf life and energy savings during processing; and |
| ● | Plastics composite part manufacturer demonstrating 7-30% improvement in strength and 40% improvement in modulus when used in sheet molding compound; and |
| ● | Plastic composite parts manufacturer demonstrating 25% increase in tensile strength and 15% improvement in flex modulus for a high-density polyethylene composite, and Plastics manufacturer demonstrating up to 25% increase in tensile modulus, 15% increase in tensile strength and 8x increase in puncture impact for nylon-based thermoplastics. |
The process of “designing-in” new materials is relatively complex and involves the use of relatively small amounts of the new material in laboratory and engineering development for an extended period of time. Following successful development, customers that incorporate our materials into their products will then order much larger quantities of material to support commercial production. Although, our customers are under no obligation to report to us on the usage of our materials, some have indicated that they have introduced or will soon introduce commercial products that use our materials. Thus, while many of our customers are currently purchasing our materials in kilogram (one or two pound) quantities, some have begun to purchase at multiple ton quantities and we believe many will require tens of tons or even hundreds of tons of material as they commercialize products that incorporate our materials. We also believe that those customers already in production will increase their order volume as demand increases and others will begin to move into commercial volume production as they gain more experience in working with our materials and engage new customers.
Commercialization Trends
We are tracking the commercial and development status of more than 75 different customer applications using our materials with some customers pursuing multiple applications. As of December 31, 2019, we had twenty-two (22) specific customer applications where our materials are incorporated into our customers’ products and such customers are actively selling these products to their own customers. In addition, we have another seventeen (17) customer applications where our customers have indicated that they expect to begin shipping product incorporating our materials in the next 3 – 6 months and have another twenty-six (26) customer applications where our customers have indicated an intent to commercialize in the next 6 – 9 months. We are also working with numerous additional customers that have not yet indicated an exact date for commercialization, but we believe have the potential to contribute to meaningful revenue in 2020 and 2021. The following graphic demonstrates the commercialization trends over the past 8 fiscal quarters as an increasing number of customers indicate their intent to commercialize applications and move into actively selling or promoting products for future sales.
6
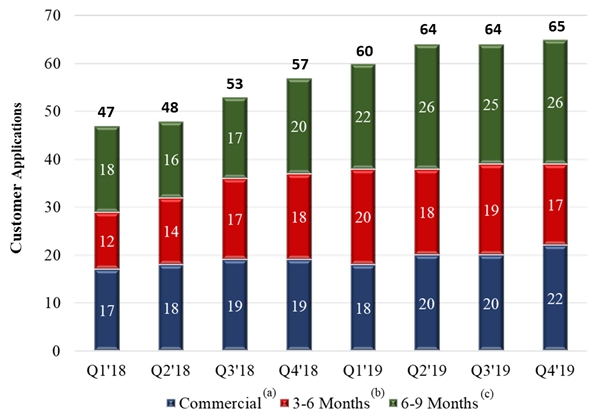
| (a) | Customer applications where our materials are used in customer products and they are actively selling them to their customers or actively promoting them for future sales. |
| (b) | Customer applications where our customers are indicating that they expect to begin shipping products incorporating our materials in the next 3-6 months. |
| (c) | Customer applications where our customers are indicating an intent to commercialize in the next 6-9 months. |
Our Intellectual Property
Some of our proprietary manufacturing processes were developed at Michigan State University (MSU) and licensed to us in 2006. We license three U.S. patents and patent applications from MSU. On August 8, 2016, we signed an agreement acquiring an exclusive license with Metna Co. (Metna) to Metna’s background IP for use of graphene nanoplatelets as additives to concrete mixtures. For purposes of the agreement, Metna’s background IP relates to the U.S. Patent 8,951,343. Also, on August 8, 2016, we entered into a second agreement for an exclusive license related to all Metna’s background technology and foreground technology, including any jointly owned foreground technology where the end use is known to be any graphite additive dispersed in concrete mixtures. On April 25, 2019 we signed an Intellectual Property License, Joint Development and Commercialization Agreement with Niagara Bottling acquiring exclusive license to certain of Niagara’s patents and applications relating to the use of graphene nanoplatelets in packaging applications. For purposes of the agreement, Niagara’s IP relates to five (5) U.S. patents and patent applications and two (2) international applications. Over time, our scientists and engineers have made many further discoveries and inventions that, as of December 31, 2019, are embodied in the form of : seventeen (17) additional U.S. patents, twenty-three (23) foreign patents, twelve (12) additional U.S. patent applications and numerous trade secrets. For many of the applications filed in the U.S., additional filings are made in other countries such as the European Union, Japan, South Korea, China, Taiwan or other applicable countries. As of December 31, 2019, we maintained twenty-three (23) international patent applications. These filings and analyses are made on a case-by-case basis. Typically, patents that are defensive in nature are not filed abroad, while those that are protective of active XGS products or applications are filed in relevant countries abroad. Our general IP strategy is to keep as trade secrets those manufacturing processes that are difficult to enforce should they be disclosed and to seek patent coverage for other manufacturing processes, materials derived from those processes, unique combinations of materials and end uses of materials containing graphene nanoplatelets. We believe that the combination of our rights under various licenses, our patents and patent applications, and our trade secrets create a robust intellectual property position.
7
Our Manufacturing Capacity
We completed the first phase of expansion in our 64,000 square-foot facility located in Mason, MI in the first half of 2018. The expansion has added 90 metric tons of graphene nanoplatelet production capacity, bringing the total capacity of the facility up to approximately 180 metric tons of dry powder. Phase two of the expansion was partially complete by year-end 2018 and resulted in up to ~270 metric tons of total graphene nanoplatelet output capacity at the facility. We completed the last portion of the Phase two expansion in the first half of 2019, resulting in up to ~400 metric tons of total graphene nanoplatelet output capacity. Manufacturing capacity is a function of product mix within a given facility. We make a range of products in our facilities that vary in their manufacturing time. It is therefore practical to consider capacity as an output range in order to reflect product mix. Our annual graphene nanoplatelet output capacity across both of our manufacturing facilities, as of December 31, 2019, ranged from approximately 225 metric tons up to approximately 450 metric tons. XG’s increasing graphene nanoplatelet capacity will support the growing demand for our products over the next several fiscal quarters and will serve as input materials to integrated products containing our graphene nanoplatelets. However, additional manufacturing capabilities for certain value-added products and certain bulk materials remain to be developed and may require the acquisition of additional facilities. In particular, the production processes for certain integrated products will require additional capital and may require additional facilities to meet expected future customer demand.
Some of the Company’s products are new products that have not yet been fully developed and for which manufacturing operations have not yet been fully scaled. Although we believe we will continue to scale our production capability and revenue in 2020, for some target products we have not yet demonstrated the capability to produce sufficient materials to generate the ongoing revenues necessary to sustain our operations in the long-term. For other target products, we have demonstrated sufficient scale to reach revenue and profits resulting in cash positive status, however, the impact of COVID-19 on our customer’s production plans and the ability for those customer with whom we are in development to introduce new products, may impact our ability to meet our commercial targets for the foreseeable future. For additional information please see “Risk Factors” herein.
Our Lead Investors
Since inception and through December 31, 2019, we have raised approximately $50 million of capital through the issuance of equity and equity-linked securities, $4 million through licensing fees and $10 million through the issuance of certain lease and senior debt obligations. Each of the entities below, after considerable due diligence, decided to take an equity position, license some of our technology or provided funding in the form of a secured loan. Notable investors and licensees in the Company are included in the funding history of the Company summarized below.
| Timing | Investor Class | Amount | ||
| 2006-2009 | Angel Groups, HNW Individuals | $2 million | ||
| 2010 | Hanwha Chemical | $3 million | ||
| 2010 | ASC-XGS, LLC | $1.6 million | ||
| 2011 | POSCO | $4 million | ||
| 2011 | Cabot Corp | $4 million license agreement | ||
| 2014 | Samsung Ventures | $3 million | ||
| 2014 | POSCO pre-emptive rights | $1.2 million | ||
| 2014/15 | Aspen/XGS II | $10 million + $1 million lease | ||
| 2015 | Series B | $4.3 million | ||
| 2016 | Dow Chemical | $10 million senior credit facility | ||
| 2016-2019 | Public Offering | $21 million (including $3 million from Soulbrain) |
8
Our Competitive Strengths
We believe that we are a leader in the emerging global market for graphene nanoplatelets. The following competitive strengths distinguish us in our industry:
Our know-how and ability to tailor our products for use in multiple applications. Many of our products and product-development activities target use of our xGnP® graphene nanoplatelets in various matrices to form composite products that are then used by our customers. We have extensive knowledge of how to tailor our products to deliver performance as composite products in various applications and we also have knowledge of how to tailor other components of a composite to adjust the performance of the composite for use in various applications. We also have knowledge of how to tailor our products for a range of other end-use markets and applications that may include: 3-D printing, anti-corrosive coatings, automotive coatings, thermally conductive adhesives, thermal interface materials/compounds, tires, thermal coatings and sporting goods equipment.
The strength of our intellectual property. Because of our focus on manufacturing process development, we believe we have one of the world’s strongest internal knowledge bases in graphene nanoplatelet manufacturing, with most of our proprietary knowledge maintained as trade secrets to avoid the disclosures required by patenting. The thirty-three (33) U.S. patents and patent applications that we are currently managing (including those under license from MSU and Metna) and thirty-seven (37) international patents and patent applications add value by protecting specific equipment, or high-value end-user product applications. The fact that two global companies have evaluated and licensed certain aspects of our production technology provides independent evidence of our technology’s effectiveness.
The breadth of our product offering. To our knowledge, we have the broadest product offering in our industry. In addition to offering four standard grades of bulk graphene nanoplatelet materials in a range of diameters and surface areas, we offer three standard ink formulations, resistive heating formulations and optional custom dispersions and formulations of our bulk materials. We also offer an XG TIM® thermal interface material and a GNP® Cement Additive product. We also offer polymer resins containing various forms of our xGnP®graphene nanoplatelets and in varying concentration and including polyurethane (XGPU), polypropylene (XGPP), polyethylene terephthalate (XGPET), vinyl ester (XGVE), polyetherimide (XGPEI) and high-density polyethylene (XGHDPE). Other polymers may be added over time depending on the end-market and customer needs.
The low-cost nature of our manufacturing processes. We believe our manufacturing processes are among the lowest-cost approaches to the manufacturing graphene nanoplatelets (subject to economies of scale) based on our internal modelling of competitive processes as well as our analysis of alternative technologies.
Our corporate partners. Four global corporations (Samsung, POSCO, Soulbrain and Hanwha Chemical) have invested over $14 million in XGS, giving us a significant global reach as well as the ability to leverage the assets of our partners. In addition, The Dow Chemical Company, or Dow, has extended $10 million in senior debt financing, of which we have drawn down $9 million as of December 31, 2019.
Our licensees should accelerate our entry into large markets. Cabot Corporation, the largest U.S.-based manufacturer of carbon particles, and POSCO, one of the world’s largest steel producers, have licensed parts of our production technology. We believe these licensees will help us distribute our products and value-added products made with our xGnP®nanoplatelets more rapidly than we could do on our own.
The number of development partners that are working with our materials. As of December 31, 2019, we had supplied materials to 303 universities or government laboratories in 41 different countries around the world. We also supply our products to a number of other organizations for the purposes of commercial design and manufacture. These other organizations include 3M, Amazon, BASF, Callaway Golf, Dow Chemical, Ford Motor Company, LG Chem, Michelin, Niagara Bottling, PPG, Raytheon, Samsung, Sherwin-Williams and The Boeing Company.
The number of commercial customers purchasing and working with our materials. As of December 31, 2019, we have supplied materials to 1,237 commercial companies around the world (in addition to universities and research laboratories) who are assessing their performance and potentially designing them into products. We have more than 75 active development relationships where we are working with end-use customers to design products for commercial use. We believe that these relationships will continue to expand.
As a result of these factors, we believe XG Sciences is a leader in the emerging global market for graphene nanoplatelets. Other independent observers have agreed with this assessment. For example, Lux Research, in a January 2019 summary stated “XG Sciences, while still in the red, now has a clear path to profitability; it is a leader in high quality graphene, and clients looking to develop novel graphene-containing products should consider it as a partner.”
Self-Underwritten Offering
We filed a Registration Statement on Form S-1 (File No. 333-209131) with the SEC on April 11, 2016 which was declared effective by the SEC on April 13, 2016 (as amended, the “Registration Statement”). The Registration Statement registered up to 3,000,000 shares of common stock at a fixed price of $8.00 per share to the general public in a self-underwritten offering (the “Offering” or our “IPO”). Post-Effective Amendment No. 1 to the Registration Statement was declared effective August 26, 2016, Post-Effective Amendment No. 2 was declared effective August 31, 2016, Post-Effective Amendment No. 3 was declared effective January 17, 2017, and Post-Effective Amendments No. 4 and No. 5 were dated April 12, 2017 and were declared effective April 14, 2017. Post-Effective Amendment No. 6 was declared effective June 26, 2018. The Offering terminated upon expiration of the Registration Statement on April 12, 2019. Although we have sold shares of our common stock in our IPO pursuant to an effective Registration Statement, we have not yet listed any of our capital stock for trading or quotation on any exchanges or the over-the-counter bulletin board.
9
As of December 31, 2019, the Company sold 2,615,425 shares under the Registration Statement at a price of $8.00 per share for proceeds of $20,923,400.
Pursuant to the Certificate of Designation for our Series A Preferred Stock, as amended (“Series A Stock”), and our Series B Preferred Stock, as amended (“Series B Stock”), all then-outstanding shares of Series A Stock and Series B Stock will automatically convert into shares of common stock upon (i) the listing of the Company’s common stock on a Qualified National Exchange (a securities exchange registered with the SEC under Section 6(a) of the Securities Exchange Act of 1934, as amended “Exchange Act”), such as the NASDAQ Capital Market or the New York Stock Exchange, or (ii) the quotation of our common stock on the OTCQB or OTCQX marketplaces operated by OTC Markets Group, Inc. (“OTC Markets”), and the act of achieving such listing or quotation is referred to hereafter as a “Public Listing” in this report. As a result, there will only be one class of equity securities outstanding — common stock — after we achieve a Public Listing, because there will be no other series of preferred stock issued and outstanding at such time. Prior to any such listing, the Series A Stock and Series B Stock may be voluntarily converted into shares of common stock at the then-current conversion rate for each series of Preferred Stock (current rate is 1.875 shares of common stock for 1 share of Series A Stock and 1 share of common stock for 1 share of Series B Stock). While there are no definitive plans to do a Public Listing as of the date of this report, the Company targets achieving a Public Listing in the next 24 months, market conditions permitting.
Public Listing
In order to achieve a Public Listing, we will have to meet certain initial listing qualifications of the Qualified National Exchange or the OTC Markets on which we are seeking the Public Listing. In addition, we will need to have market makers agree to make a market in our common stock and file a FINRA Form 15c211 with the SEC on our behalf before we can achieve a Public Listing, and we will also need to remain current in our quarterly and annual filings with the SEC. Although we intend to seek a Public Listing, we cannot make any assurances that our common stock will ever be quoted or traded on a Qualified National Exchange or the OTC Markets or that any market for our common stock will develop.
Employees
As of December 31, 2019, we had 39 full-time employees and 2 part-time employees. 3 of these employees were contract employees who may generally be hired as permanent employees after 3 – 6 months. Employees include the following four senior managers that report to the CEO: Chief Financial Officer, Chief Commercial Officer, Vice President of Operations and Chief Technologist. The Company employs a total of 7 full-time scientists and technicians in its R&D group, including the Chief Technologist.
Corporate Information
XG Sciences, Inc. was incorporated on May 23, 2006 in the State of Michigan and is organized as a “C” corporation under the applicable laws of the United States and State of Michigan. We do not currently have any affiliated companies or joint venture partners, and we have one wholly owned subsidiary called XG Sciences IP, LLC. This subsidiary was created in 2014 for the purpose of holding our intellectual property. Our headquarters and principal executive offices are located at 3101 Grand Oak Drive, Lansing, Michigan, 48911 and our telephone number is (517) 703-1110. Our Annual Report on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed or furnished pursuant to section 13(a) or 15(d) of the Exchange Act are available free of charge through our website as soon as reasonably practicable after we electronically file with or furnish them to the SEC, and are available in print to any stockholder who requests a copy.
Our website address ishttp://www.xgsciences.com, although the information contained in, or that can be accessed through, our website is not part of this filing. You may also contact Dr. Philip Rose, our Chief Executive Officer via email atp.rose@xgsciences.com. Additionally, the SEC maintains a website that contains reports, proxy statements, information statements and other information regarding issuers, including us, that file electronically with the SEC at www.sec.gov.
10
Risks Relating to Our Business and Industry
We will need to raise substantial additional capital in the future to fund our operations and we may be unable to raise such funds when needed and on acceptable terms, which could have a materially adverse effect on our business.
Developing, manufacturing and selling nanomaterials in commercially-viable quantities requires substantial funding. As of December 31, 2019, we had cash, cash equivalents and restricted cash on hand of $1,129,702. We believe our cash is sufficient to fund our operations for the next twelve months (through April 2021) when taking into account various sources of funding and cash received from continued commercial sales transactions. We intend that the primary means for raising funds will be through equity offerings that may include: private placement with one or more strategic investors, offering of shares of common stock to the public through a self-underwritten offering or through an underwritten offering with a bank in parallel with a listing on a Qualified National Exchange. There can be no assurance that we will be able to raise additional equity capital in subsequent equity offerings or that the terms and conditions of any future financings will be workable or acceptable to us and our stockholders. Our continuation as a going concern is dependent upon continued financial support from our shareholders, our ability to obtain necessary equity and/or debt financing to continue operations, and the attainment of profitable operations. In the event that we are not able to raise substantial additional funds in the future with terms that are acceptable, or adjust our business model accordingly, we may be forced to curtail or cease operations and investors could lose all or a significant part of their investment, or we could be prevented from making our periodic filings under the Exchange Act.
A pandemic, epidemic or outbreak of an infectious disease in the markets in which we operate or that otherwise impacts our facilities or suppliers could adversely impact our business.
We expect that a pandemic, epidemic, or outbreak of an infectious disease including the recent outbreak of respiratory illness caused by the novel COVID-19 virus first identified in Wuhan, Hubei Province, China (“COVID-19) will continue to affect our markets, facilities, customers and employees, and our business could continue to be adversely affected in the future. Consequences of the coronavirus outbreak are resulting in disruptions in or restrictions on our ability to travel and the ability of manufacturers, including many of our customers, to maintain normal operations. If such an infectious disease broke out at our facilities, or if, due to governmental or other restrictions, our facilities are unable to operate for an extended period of time, our operations may be affected significantly, our productivity may be affected, our ability to complete projects in accordance with our contractual obligations may be affected, and our ability to manufacture and sell our graphene nanoplatelets may be hindered. If the suppliers that we rely on for raw materials and other supplies to conduct operations are affected by COVID-19, we may not be able to fill orders that are received or our cost of sales may increase, and our financial position may suffer. If our customers are affected by COVID-19, orders for our products may be delayed or cancelled, and our financial position may suffer. Further, infectious outbreak may continue to cause disruption to the U.S. economy, or the economies of the markets in which we operate such as China and South Korea, and cause shortages of materials, labor and transportation which could affect our customers’ ability to produce finished products utilizing our graphene nanoplatelets, affect production capacity and business confidence, or cause economic changes that we cannot anticipate. Overall, the potential impact of a pandemic, epidemic or outbreak of an infectious disease with respect to our markets or our facilities is difficult to predict and could continue to adversely impact our business for an indefinite period of time. In response to the COVID-19 situation, federal, state and local governments (or other governments or bodies in markets in which we operate) have placed restrictions on travel and conducting or operating business activities. At this time, those restrictions are very fluid and evolving, and when such restrictions will be lifted is unknown. We have been and will continue to be impacted by those restrictions. Given that the type, degree and length of such restrictions are not known at this time, we cannot predict the overall impact of such restrictions on us, our customers, our employees, our supply and distribution chains, others that we work with or the overall economic environment. As such, the impact these restrictions may have on our financial position, operating results and liquidity cannot be reasonably estimated at this time, but we expect the impact to be material. In addition, due to the speed with which the COVID-19 situation is developing and evolving, there is uncertainty around its ultimate impact on public health, business operations and the overall economy; therefore, the negative impact on our financial position, operating results and liquidity cannot be reasonably estimated at this time, but we expect the impact to be material.
We could be adversely affected by our exposure to customer concentration risk.
We are subject to customer concentration risk as a result of our reliance on relatively few customers for a significant portion of our revenues. We had 1 customer whose purchases accounted for 41% and 72% of revenue in the twelve months ended December 31, 2019 and 2018, respectively. In 2019, we had 4 customers whose purchases accounted for 79% of revenue. Due to the nature of our business and the relatively large size of many of the applications our customers are developing, we anticipate that we will be dependent on a relatively small number of customers for the majority of our revenues for the next few years, and if one or more of these customers does not place orders, there would be a risk of significant loss of future revenues, which could in turn have a material adverse effect on our business and on your investment.
11
Downturns in general economic conditions could adversely affect our profitability.
Downturns in general economic conditions such as the economic conditions that have coincided with COVID 19 can cause fluctuations in demand for our products, product prices, volumes and gross margins. Future economic conditions may not be favorable to our industry. A decline in the demand for our products or a shift to lower-margin products due to deteriorating economic conditions could adversely affect sales of our products and our profitability and could also result in impairments of certain of our assets.
Furthermore, any uncertainty in economic conditions may result in a slowdown to the global economy that could affect our business by reducing the prices that our customers may be able or willing to pay for our products or by reducing the demand for our products. Due to our customer concentration risk, the loss of one or more of our large customers could significantly affect our business, operating results and financial condition.
If we are unable to continue as a going concern, our securities will have little or no value.
The report of our independent registered public accounting firm that accompanies our consolidated financial statements for the year ended December 31, 2019 contains a going concern qualification in which such firm expressed substantial doubt about our ability to continue as a going concern. We currently anticipate that our cash and cash equivalents will be sufficient to fund our operations through April 2021, without raising additional capital. We believe that we will need to raise approximately $500,000 to $1,000,000 of additional capital in order to continue our operations for the next twelve months in a minimal to no revenue growth environment. Our continuation as a going concern is dependent upon continued financial support from our shareholders, our ability to obtain necessary equity and/or debt financing to continue operations, and growth in revenue from operations. These factors raise substantial doubt regarding our ability to continue as a going concern. We cannot make any assurances that additional financings will be available to us and, if available, completed on a timely basis, on acceptable terms or at all. If we are unable to complete an equity or debt offering, or otherwise obtain sufficient financing when and if needed, it would negatively impact our business and operations. It could also lead to the reduction or suspension of our operations and ultimately force us to cease our operations.
We have a limited operating history, an accumulated deficit and a stockholders’ deficit, making it difficult for you to evaluate our business and your investment.
XG Sciences, Inc. was incorporated on May 23, 2006, and is an advanced materials company. We sell bulk nanomaterials or products made with these materials to other companies for incorporation into their products. To date, there has been limited incorporation of our materials or products into customer products that are released for commercial sale. Because there is a limited demonstrated history of commercial success for our products, it is difficult to evaluate whether our products will ultimately be successful in the market. It is possible that larger and or extended commercial success may never happen and that we will never achieve the level of revenues necessary to sustain our business or continue to attract additional financing.
Many of our products represent new products that have not yet been fully developed and for which manufacturing operations have not yet been fully scaled. This means that investors are subject to all of the risks incident to the creation and development of multiple new products and their associated manufacturing processes.
As of December 31, 2019, and December 31, 2018, we have an accumulated deficit from operations of $(65,581,128) and $(55,687,160), respectively. As of December 31, 2019, and December 31, 2018, our total stockholder’s equity was $(2,146,797) and $4,990,719, respectively. The deficit reflects net losses in each period since our inception incurred through development of nanomaterials without the presence of large-scale adoption to generate substantial revenues to cover development costs and generate a profit. We have never paid a dividend. Also, since inception, we have not generated sufficient revenues to cover our fixed expenses or sustain our business in any financial reporting period. Nor have we demonstrated the capability to produce sufficient materials to generate the ongoing revenues necessary to sustain our operations in the long-term. There can be no assurance that we will ever produce a profit.
Because we are subject to these uncertainties, there may be risks that management has failed to anticipate and you may have a difficult time evaluating our business and your investment in our Company. Our ability to become profitable depends primarily on our ability to successfully commercialize our products in the future. Even if we successfully develop and market our products, we may not generate sufficient or sustainable revenue to achieve or sustain profitability, which could cause us to cease or curtail operations. In such case, you would likely lose all or a significant part of your investments.
We have limited experience in the higher volume manufacturing that will be required to support profitable operations, and the risks associated with scaling to larger production quantities may be substantial.
We have limited experience manufacturing our products. We have established initial commercial or pilot-scale production facilities for our bulk powders and certain integrated products. In order to develop the capacity to produce much higher volumes, it will be necessary to produce multiples of existing processes or engineer new production processes in some cases.
We have completed the initial phases of building out our 64,000 square foot manufacturing facility in Mason, MI, but there can be no assurance that this new facility will continue to be expanded in the future on time or as planned. In addition, due to recent events surrounding our business and the COVID-19 pandemic, in March of 2020, we restructured our organization by reducing headcount by 45%, by furloughing substantially all manufacturing employees and by implementing temporary salary reductions ranging from 15-20% which has resulted in a 58% reduction in annual payroll and related costs. In April 2020, we furloughed additional employees in our R&D and Engineering departments. We expect that these actions could have a material impact on our ability to scale up our production processes, and there is no guarantee that we will be able to economically scale-up our production processes to the levels required. If we are unable to scale-up our production processes and facilities to support sustainable sales levels, the Company may be forced to curtail or cease operations and investors could lose all or a significant part of their investment.
12
Projection of fixed monthly expenses and operating losses for the near future means that investors may not earn a return on their investment or may lose their investment.
Because of the nature of our business, we project considerable fixed expenses that will lead to projected monthly deficits for the near future. Fixed manufacturing expenses to maintain production facilities, compensation expenses for scientists and other critical personnel, and ongoing rent and utilities amount to several hundred thousand dollars per month, and we believe that such expenses are required as a precursor to significant customer sales. However, there can be no assurance that monthly sales will ever reach a sufficient level to cover the cost of ongoing monthly expenses and if they do, are maintained for a sustainable period of time. If sufficient regular monthly sales are not generated to cover these fixed expenses, we will continue to experience monthly cash flow deficits which, if not eliminated, will require continuing new investment in the Company. If monthly cash flow deficits continue beyond levels that investors find tolerable, we may not be able to raise additional funds and may be forced to curtail or cease operations and investors could lose all or a significant part of their investment.
We have a long and complex sales cycle and have not demonstrated the ability to operate successfully in this environment.
It has been our experience since our inception that the average sales cycle for our products can range from one to seven years from the time a customer begins testing our products until the time that they could be successfully used in a commercial product. The product introduction timing will vary based on the target market, with automotive uses typically being toward the long end and consumer products toward the shorter end. We have a limited track record of success in completing customer development projects, which makes it difficult for investors to fully evaluate the likelihood of our future success. The sales and development cycle for our products is subject to customer budgetary constraints, internal acceptance procedures, competitive product assessments, scientific and development resource allocations, and other factors beyond our control. If we are not able to successfully accommodate these factors to enable customer development success, we will be unable to achieve sufficient sales to reach profitability. In this case, we may not be able to raise additional funds and may be forced to curtail or cease operations and investors could lose all or a significant part of their investment.
Our revenues often fluctuate significantly based on one-off orders from customers or from the recognition of non-product revenues which vary from period-to-period, which may materially impact our financial results from period to period.
Because of the potential for large revenue swings from one-time, large orders or grants (or other such non-product revenue), it may be difficult to accurately forecast the needs for inventory, working capital, and other financial resources from period-to-period. Such orders would require a significant short-term increase in our production capacity and would require the financial resources to add staff and support the associated working capital. If such large, one-time orders were not handled smoothly, customer confidence in us as a viable supplier could be reduced and we might not succeed in capturing the additional larger orders that may be reflected in our business plan.
We operate in an advanced technology arena where hypothesized properties and benefits of our products may not be achieved in practice, or in which technological change may alter the attractiveness of our products.
Because there is only limited sustained history of successful use of our products in commercial applications, there is no assurance that broad successful commercial applications may be broadly technically feasible. Many of the scientific and engineering data related to our products has been generated in our own laboratories or in laboratory environments at our customers or third-parties, like universities and national laboratories. It is well known that laboratory data is not always representative of commercial applications.
Likewise, we operate in a market that is subject to rapid technological change. Part of our business strategy is to monitor such change and take steps to remain technologically current, but there is no assurance that such strategy will be successful. If we are not able to adapt to new advances in materials sciences, or if unforeseen technologies or materials emerge that are not compatible with our products and services or that could replace our products and services, our revenues and business prospects would likely be adversely affected. Such an occurrence may have severe consequences, including the potential for our investors to lose all or a significant part of their investment.
13
Competitors that are larger and better funded may cause us to be unsuccessful in selling our products.
We operate in a market in which there are competitors. Global research is being conducted by substantially larger companies who have greater financial, personnel, technical, and marketing resources. There can be no assurance that our strategy of offering better materials based on our proprietary graphene nanoplatelets will be able to compete with other companies, many of whom will have significantly greater resources, on a continuing basis. In the event that we cannot compete successfully, we may be forced to cease or curtail operations and investors may lose all or a significant part of their investment.
We are dependent on key employees.
Our operations and product development are dependent upon the experience and knowledge of Philip Rose, our Chief Executive Officer. If he was to resign or be terminated, our business would be adversely affected in the short term, and his departure could disrupt the business enough to endanger your investment. We also depend on Jacqueline Lemke, Chief Financial Officer, Dr. Leroy Magwood, Chief Technologist, and Scott Murray, Vice President of Operations. If the services of any of these individuals should become unavailable, our business operations might be adversely affected. We hold “Key Person” insurance of $5 million on our CEO, Philip Rose, but do not hold any other “Key Person” insurance, and, if several of these individuals became unavailable at the same time, our ability to continue normal business operations might be adversely affected, to the extent that revenue or profits could be diminished and you could lose all or a significant part of your investment.
Our success depends in part on our ability to protect our intellectual property rights, and our inability to enforce these rights could have a material adverse effect on our competitive position.
We rely on the patent, trademark, copyright and trade-secret laws of the United States and the countries where we do business to protect our intellectual property rights. We may be unable to prevent third parties from using our intellectual property without our authorization. The unauthorized use of our intellectual property could reduce any competitive advantage we have developed, reduce our market share or otherwise harm our business. In the event of unauthorized use of our intellectual property, litigation to protect or enforce our rights could be costly, and we may not prevail.
Many of our technologies are not covered by any patent or patent application, and our issued and pending U.S. and non-U.S. patents may not provide us with any competitive advantage and could be challenged by third parties. Our inability to secure issuance of our pending patent applications may limit our ability to protect the intellectual property rights these pending patent applications were intended to cover. Our competitors may attempt to design around our patents to avoid liability for infringement and, if successful, our competitors could adversely affect our market share. Furthermore, the expiration of our patents may lead to increased competition.
Our pending trademark applications may not be approved by the responsible governmental authorities and, even if these trademark applications are granted, third parties may seek to oppose or otherwise challenge these trademark applications. A failure to obtain trademark registrations in the United States and in other countries could limit our ability to protect our products and their associated trademarks and impede our marketing efforts in those jurisdictions.
In addition, effective patent, trademark, copyright and trade secret protection may be unavailable or limited in some foreign countries. In some countries, we do not apply for patent, trademark or copyright protection. We also rely on unpatented proprietary manufacturing expertise, continuing technological innovation and other trade secrets to develop and maintain our competitive position. Although we enter into confidentiality agreements with our employees and generally with third parties to protect our intellectual property, these confidentiality agreements are limited in duration and could be breached and may not provide meaningful protection of our trade secrets or proprietary manufacturing expertise. Adequate remedies may not be available if there is an unauthorized use or disclosure of our trade secrets and manufacturing expertise. In addition, others may obtain knowledge about our trade secrets through independent development or by legal means. The failure to protect our processes, apparatuses, technology, trade secrets and proprietary manufacturing expertise, methods and compounds could have a material adverse effect on our business by jeopardizing critical intellectual property.
Where a product formulation or process is kept as a trade secret, third parties may independently develop or invent and patent products or processes identical to our trade-secret products or processes. This could have an adverse impact on our ability to make and sell products or use such processes and could potentially result in costly litigation in which we might not prevail.
We could face intellectual property infringement claims that could result in significant legal costs and damages and impede our ability to produce key products, which could have a material adverse effect on our business, financial condition and results of operations.
14
If we are unable to implement and maintain effective internal control over financial reporting, our stock could be less attractive to potential investors.
We are required to establish and maintain appropriate internal controls over financial reporting, subject to exemptions that we avail ourselves of under the JOBS Act discussed below. Failure to establish such controls, or any failure of such controls once established, could adversely impact our public disclosures regarding our business, financial condition or results of operations. Any failure of our controls could also prevent us from maintaining accurate accounting records and discovering accounting errors and financial frauds. Rules adopted by the SEC pursuant to Section 404 of the Sarbanes-Oxley Act of 2002 require annual assessment of our internal control over financial reporting, and the standards that must be met for management to assess the internal control over financial reporting as effective are new and complex, and require significant documentation, testing and possible remediation to meet the detailed standards. In November 2018, the Company hired a CFO to oversee the finance and reporting function, modify processes to mitigate segregation of duties issues and increase supervisory and review controls.
In addition, management’s assessment of internal controls over financial reporting may identify material weaknesses and conditions that need to be addressed in our internal controls over financial reporting or other matters that may raise concerns for investors in the future. Any actual or perceived weaknesses and conditions that need to be addressed in our internal control over financial reporting, disclosure of management’s assessment of our internal controls over financial reporting, or at such time as we are no longer subject to exemptions under the JOBS Act, disclosure of our independent registered public accounting firm’s report on management’s assessment of our internal controls over financial reporting, may have an adverse impact on our ability to sell our common stock.
Future adverse regulations could affect the viability of the business.
Our bulk products have been approved for sale in the United States by the U.S. Environmental Protection Agency after a detailed review of our products and production processes. In most cases, as far as we are aware, there are no current regulations elsewhere in the world that prevent or prohibit the sale of our products. Nevertheless, the sale of nano-materials is a subject of regulatory discussion and review in many countries around the world. In some cases, there is a discussion of potential testing requirements for toxicity or other health effects of nano-materials before they can be sold in certain jurisdictions. If such regulations are enacted in the future, our business could be adversely affected because of the requirement for expensive and time-consuming tests or other regulatory compliance. If our nano-materials are found to be toxic, such finding could have a material impact on our business and on the production and sale of our products. There can be no assurance that future regulations might not severely limit or even prevent the sale of our products in major markets, in which case our financial prospects might be severely limited, causing investors to lose all or a significant part of their investment.
Compliance with changing regulation of corporate governance and public disclosure will result in additional expenses and will divert time and attention away from revenue generating activities.
Changing laws, regulations and standards relating to corporate governance and public disclosure, including the Sarbanes-Oxley Act of 2002 and related SEC regulations, have significantly increased the costs and risks associated with accessing the public markets and public reporting. Our management team will need to invest significant management time and financial resources to comply with both existing and evolving standards for public companies, which will lead to increased general and administrative expenses and a diversion of management time and attention from revenue generating activities to compliance activities, which could have an adverse effect on our business.
Given our limited resources, we may not effectively manage our growth.
There is no guarantee that we have the resources, financial or operational, required to manage our growth. This is particularly true as we expand facilities and manufacture our products on a greater commercial scale. Furthermore, rapid growth in our operations may place a significant strain on our management, administrative, operational and financial infrastructure. The inability to adequately manage our growth could have a material and adverse effect on our business, financial condition or results of operations, thus resulting in a lower quoted price of our common stock.
15
An increase in the cost of raw materials or electricity might affect our profits.
Any increase in the prices of our raw materials or energy might affect the overall cost of our products. If we are not able to raise our prices to pass on increased costs to our customers, we would be unable to maintain our existing profit margins. Our major cost components include items such as graphite, acids and bases, and electricity, which items are normally readily available industrial commodities. During our history as a business, we have not seen any material impact (as defined by GAAP) on our cost structure from fluctuations in raw material or energy costs, but this could change in the future, especially given the impact of COVID-19 and its impact on the global economy.
Our results of operations could deteriorate if our manufacturing operations were substantially disrupted for an extended period.
In addition to the disruptions to our manufacturing operations caused by COVID-19, our manufacturing operations are subject to disruption due to extreme weather conditions, floods and similar events, major industrial accidents, strikes and lockouts, adoption of new laws or regulations, changes in interpretations of existing laws or regulations or changes in governmental enforcement policies, civil disruption, riots, terrorist attacks, war, and other events. We cannot assure you that no such events will occur. If such an event occurs, it could have a material adverse effect on us.
Some health effects of nanotechnology are unknown.
There is scientific debate on the health effects of nano-materials such as graphene nanoplatelets, but some scientists believe that certain nano-materials may be hazardous to human health, including the respiratory system if inhaled. Although there is no conclusive evidence of any danger associated with the handling of the Company’s products, there is a theoretical risk of danger to health if an individual is exposed to and/or inhales/ingests some of the Company’s products. The specific health effects of nanoplatelets are unknown and can depend on how they are incorporated and/or bonded to other materials. We carefully evaluate potential health effects of our products and the effects of handling materials on our employees and those who manufacture for us, but as any specific health risks are unknown, we cannot be certain our products are free of danger to our employees and customers. If graphene nanoplatelets are found to be hazardous to human health, this may adversely affect market acceptance of our products, subject us to additional regulation and have an adverse effect on our business.
Defects in our products or poor performance of our customers’ products could result in lost sales and subject us to substantial liability.
We have limited experience with large scale commercialization by our customers of products incorporating our nanoplatelets, and the chance of variability in the performance of our products as shipped may impact the performance of our customers’ products. If our customers’ products incorporating nanoplatelets perform poorly, whether due to design, engineering, production or other reasons, our customers may scale back or cancel orders. In certain cases, if our nanoplatelets are found to be the component that leads to failure or a failure to meet the performance specifications of a customer, we could be required to pay monetary damages. Real or perceived defects in our products could result in claims by our customers for losses they sustain. If our customers make such claims, we may be required, or may choose, for customer relations or other reasons, to expend additional resources to help correct any real or perceived defects. Liability provisions in our terms and conditions of sale may not be enforceable under some circumstances or may not fully or effectively protect us from claims and related liabilities and costs. In addition, regardless of the party at fault, errors of these kinds divert the attention of our engineering personnel from our product development efforts, damage our reputation and the reputation of our products, cause significant customer relations problems and can result in product liability claims. In addition, even claims that ultimately are unsuccessful could result in expenditures of funds in connection with litigation and divert management’s time and other resources. We also may incur costs and expenses relating to a recall of one or more of our products, and the occurrence of such claims could result in the delay or loss of market acceptance of our products and could adversely affect our business, operating results and financial condition.
16
If product liability lawsuits are brought against us, we may incur substantial liabilities.
We have limited experience with the large-scale manufacturing and distribution of our products. The commercialization of our products and the sale of our products in significant quantities involves exposure to product liability claims. We have limited product liability insurance. If existing coverage is insufficient to cover claims or we cannot obtain sufficient insurance coverage at an acceptable cost or otherwise protect against potential product liability claims, the commercialization of products that we develop may be prevented or inhibited. We cannot predict all of the adverse health events that our products or use of our products in our customer’s products may cause. As a result, our future coverages may not be adequate to protect us from all of the liabilities that we may incur. If losses from product liability claims exceed any insurance coverage, we may incur substantial liabilities that exceed our financial resources. In addition, we may not be able to maintain future product liability insurance at an acceptable cost, if at all, and this insurance may not provide adequate coverage against potential claims or losses. If we are required to pay a product liability claim, we may not have sufficient financial resources and our business and results of operations may be harmed. Whether or not we are ultimately successful in product liability litigation, such litigation could also consume substantial amounts of our financial and managerial resources, and might result in adverse publicity, all of which could have a material adverse effect on our business.
Cybersecurity incidents could disrupt business operations, result in the loss of critical and confidential information, and adversely impact our reputation and results of operations.
Global cybersecurity threats and incidents can range from uncoordinated individual attempts to gain unauthorized access to information technology systems to sophisticated and targeted measures known as advanced persistent threats, directed at the Company, its plants and operations, its products, its customers and/or its third-party service providers. We rely on third party service providers to protect information technology systems, a breach of which could expose our confidential intellectual property, including trade secrets. The failure to protect such intellectual property could create a diminution in the value of our investment in research, development and engineering, and increased cybersecurity protection and remediation costs, which in turn could adversely affect our competitiveness and results of operations. We cannot be sure that our information technology infrastructure is safe from cybersecurity threats or other forms of intellectual property appropriation. Cybersecurity incidents, depending on their nature and scope, could potentially result in the misappropriation, destruction, corruption or unavailability of critical data and confidential or proprietary information (our own or that of third parties) and the disruption of business operations. The potential consequences of a material cybersecurity incident or other such intellectual property appropriation include reputational damage, claims from and litigation with third parties, fines levied by governmental authorities, and competitive disadvantages in our business.
Risks Relating to Our Common Stock
There is a risk of dilution of your percentage ownership of common stock in the Company.
We have the right to raise additional capital or incur borrowings from third parties to finance the business. We may also implement public or private mergers, business combinations, business acquisitions and similar transactions pursuant to which we would issue substantial additional capital stock to outside parties, causing substantial dilution in the ownership of the Company by our existing stockholders. Subject to certain exceptions, our Board of Directors has the authority, without the consent of any of the stockholders, to cause the Company to issue more shares of common stock and/or preferred stock at such price and on such terms and conditions as are determined by the Board in its sole discretion.
The sale of the shares offered by us in our past Registration Statement, as well as the shares of common stock issuable upon the exercise of options, convertible notes and warrants, the shares issuable upon conversion of Series A Preferred Stock (including the shares of Series A Preferred Stock issuable upon the exercise of warrants), the shares issuable upon Conversion of any Series B Preferred Stock that we issue and the issuance of additional shares of capital stock by us will dilute an investor’s ownership percentage in the Company and could impair our ability to raise capital in the future through the sale of equity securities.
Certain stockholders who are also officers and directors of the Company may have significant control over our management, which may not be in your best interests.
As of December 31, 2019, the directors, or the entities they represent, and executive officers of the Company owned approximately 44.9% of the voting stock of the Company. Certain of these executives and directors, or the companies they represent, converted secured convertible notes into an additional 1,383,900 shares of Series A Preferred Stock on December 31, 2015 and have made commitments to purchase additional convertible notes in the near future.
17
Additionally, certain existing shareholders are party to a certain Shareholder Agreement, as amended on March 18, 2013 and amended on February 24, 2016, effective as of April 13, 2016, and amended on May 30, 2018 (the “Shareholder Agreement”). Although shareholders having purchased shares under our Registration Statement will not be subject to the Shareholder Agreement, certain provisions of such Shareholder Agreement may impact the governance of the Company. Pursuant to the Shareholder Agreement, (a) so long as Aspen Advance Opportunity Fund, LP, or “AAOF” or its affiliates own 10% of more of the aggregate outstanding Shareholder Stock (as defined in the Shareholder Agreement), (i) the size of the Board of Directors shall be set at seven individuals (provided, however, that the number of directors on the Board of Directors may be increased or decreased with the prior written consent of AAOF and shareholders (including AAOF) who in the aggregate then own Shareholder Stock representing a majority of the then issued and outstanding voting stock of the Company), (ii) one person nominated by AAOF shall be elected to the Board of Directors, (iii) two members of the Board of Directors, other than those nominated by AAOF, POSCO or Hanwha Chemical, shall qualify as independent Directors.
The Shareholder Agreement will continue in effect, unless the Shareholder Agreement is earlier terminated in accordance with its terms, until the date on which the Company’s common stock is listed on the NASDAQ Stock Market of the New York Stock Exchange. As a result, the Shareholder Agreement will continue to remain in effect and certain of our larger shareholders will be entitled to continue to exercise their rights under such Shareholder Agreement, but purchasers of shares of common stock under the Registration Statement filed in connection with our last Offering are not required to adopt the Shareholder Agreement.
As a result, such entities have a significant influence on the affairs and management of the Company, as well as on all matters requiring stockholder approval, including electing and removing members of our Board of Directors, causing us to engage in transactions with affiliated entities, causing or restricting the sale or merger of the Company, and certain other matters. Such concentration of ownership and control could have the effect of delaying, deferring or preventing a change in control of the Company even when such a change of control would be in the best interests of our shareholders.
We may, in the future, issue additional shares of common stock, which would reduce investors’ percent of ownership and may dilute our share value.
Our Articles of Incorporation, as amended, authorize the issuance of up to 25,000,000 shares of common stock and up to 8,000,000 shares of preferred stock. As of December 31, 2019, the Company had 4,024,443 shares of common stock and 1,890,354 shares of Series A Preferred Stock issued and outstanding.
Upon a Public Listing on a Qualified National Exchange, all Series A Preferred Stock then currently outstanding will automatically convert into shares of common stock at the then-current Series A Conversion Rate (current ratio is 1.875 shares of common for 1 share of Series A), which would result in the issuance of 3,544,408 shares of common stock assuming the conversion of all 1,890,354 shares of Series A Preferred Stock. Series A Preferred Stockholders may also voluntarily convert at the then-current rate at any time prior to any such Public Listing on a Qualified National Exchange.
As of December 31, 2019, the Company had also granted options to purchase up to 839,625 shares of common stock and had issued warrants to purchase up to (i) 487,017 shares of common stock, (including the warrants for 225,000 shares issued under the Dow Facility) and (ii) 972,720 shares of Series A Preferred Stock which, if exercised, would be convertible into 1,823,845 shares of common stock at the then-current Series A Conversion Rate (currently 1.875 shares of common for 1 share of Series A). Therefore, we have committed to issue up to an additional 6,694,895 shares of common stock, which includes the issuance of (a) 3,544,408 shares upon conversion of all 1,890,354 shares Series A Preferred Stock currently outstanding at the Series A Conversion Rate, (b) 1,823,845 shares upon the conversion of 972,720 shares of Series A Preferred Stock (at the current Series A Conversion Rate) which are issuable upon exercise of 972,720 Series A warrants, (c) 839,625 shares upon the exercise of options and (d) the issuance of 487,017 shares upon the exercise of warrants. If we issued all 6,694,895 shares, we would have, including the 4,024,443 shares currently outstanding, 10,719,338 shares issued and outstanding, with 14,280,662 authorized shares available for future issuance. The future issuance of common stock or other securities convertible into our common stock may result in substantial dilution in the percentage of our common stock held by our existing shareholders. Other than in our past IPO, we may value any common stock issued in the future on an arbitrary basis. The issuance of common stock for future services or acquisitions or other corporate actions may have the effect of diluting the value of the shares held by our investors, might have an adverse effect on any trading market for our common stock and could impair our ability to raise capital in the future through the sale of equity securities.
18
The Dow Facility obligates us to issue a warrant to purchase a share of our common stock for each $40 in debt drawn. Subsequent to the 2019 year-end, on February 12, 2020, we drew the remaining $1 million from the Dow Facility and issued warrants to purchase up to 25,000 more of our shares. With the issue of these warrants, further dilution has occurred.
We are considered a smaller reporting company and are exempt from certain disclosure requirements, which could make our stock less attractive to potential investors.
Rule 12b-2 of the Exchange Act, as amended and effective September 10, 2018, defines a “smaller reporting company” as an issuer that is not an investment company, an asset-backed issuer, or a majority-owned subsidiary of a parent that is not a smaller reporting company and that:
| ● | Had a public float of less than $250 million as of the last business day of its most recently completed second fiscal quarter, computed by multiplying the aggregate worldwide number of shares of its voting and non-voting common equity held by non-affiliates by the price at which the common equity was last sold, or the average of the bid and asked prices of common equity, in the principal market for the common equity; or |
| ● | In the case of an initial registration statement under the Securities Act or Exchange Act for shares of its common equity, had a public float of less than $250 million as of a date within 30 days of the date of the filing of the registration statement, computed by multiplying the aggregate worldwide number of such shares held by non-affiliates before the registration plus, in the case of a Securities Act registration statement, the number of such shares included in the registration statement by the estimated public offering price of the shares; or |
| ● | In the case of an issuer who had a public float as calculated under paragraph (1) or (2) of this definition of less than $700 million or had no float and had annual revenues of less than $100 million during the most recently completed fiscal year for which audited financial statements are available. |
As a “smaller reporting company” (in addition to and without regard to our status as an “emerging growth company”) (i) we are not required and may not include a “Discussion and Analysis” section in our proxy statements; (ii) we provide only 3 years of business development information; (iii) we provide fewer years of selected financial data in certain tables; and (iv) we have other “scaled” disclosure requirements that are less comprehensive than issuers that are not “smaller reporting companies” which may make our stock less attractive to potential investors, which could make it even more difficult for you to sell your shares.
We are subject to the periodic reporting requirements of the Exchange Act, which will require us to incur audit fees, legal fees and valuation fees in connection with the preparation of such reports. These additional costs will negatively affect our ability to earn a profit.
We are required to file periodic reports with the SEC pursuant to the Exchange Act and the rules and regulations thereunder by virtue of our effective Registration Statement and our status as a smaller reporting company. In order to comply with such requirements, our independent registered auditors have to review our financial statements on a quarterly basis and audit our financial statements on an annual basis. Moreover, our legal counsel will have to review and assist in the preparation of such reports. Factors such as the number and type of transactions that we engage in and the complexity of our reports cannot accurately be determined at this time and may have a major negative effect on the cost and amount of time to be spent by our auditors and attorneys. However, the incurrence of such costs will be an expense to our operations and thus have a negative effect on our ability to meet our overhead requirements and earn a profit, in the event that we are unable to raise additional capital and do not have sufficient funds to pay these costs.
However, for as long as we remain an “emerging growth company” we intend to take advantage of certain exemptions from various reporting requirements until we are no longer an “emerging growth company.”
For so long as we remain a smaller reporting company, we benefit from many of the same exemptions and exclusions as an emerging growth company. In the event that we cease to be an emerging growth company as a result of a lapse of the five-year period, but continue to be a smaller reporting company, we would continue to be subject to the exemptions available to emerging growth companies until such time as we were no longer a smaller reporting company.
After, and if ever, we are no longer an “emerging growth company,” we expect to incur significant additional expenses and devote substantial management effort toward ensuring compliance with those requirements applicable to companies that are not “emerging growth companies,” including Section 404 of the Sarbanes-Oxley Act.
19
For so long as we are an emerging growth company, we may rely on certain exemptions provided in the JOBS Act, which could make our common stock less attractive to investors due to the nature of the reduced disclosure.
We are an “emerging growth company,” as defined in the JOBS Act, and may remain an emerging growth company up to the last day of the fiscal year following the fifth anniversary of the date of the completion of our Offering. For so long as we remain an emerging growth company, we are permitted and plan to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002, or SOX Section 404, not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, reduced disclosure obligations regarding executive compensation and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. In this Annual Report on Form 10-K, we have provided only two years of audited financial statements and have not included all of the executive compensation related information that would be required if we were not an emerging growth company. We cannot predict whether investors will find our common stock less attractive if we rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile. In addition, the JOBS Act provides that an emerging growth company can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an emerging growth company to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, we will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
If our common stock becomes a “penny stock,” you may have greater difficulty selling your shares.
Penny stocks are generally equity securities with a price of less than $5.00, other than securities registered on certain national securities exchanges or quoted on the NASDAQ system, provided that current price and volume information with respect to transactions in such securities is provided by the exchange or quotation system. After a Public Listing, our common stock may become a “penny stock” within the meaning of the rules, the rules apply to us and to our securities if we are not listed on a national securities exchange. These rules may further affect the ability of owners of shares to sell our securities in any market that might develop for them. As long as the trading price of our common stock is less than $5.00 per share, even if our common stock is quoted on either the OTCQX or OTCQB market place operated by the OTC Markets, our common stock will be subject to Rule 15g-9 under the Exchange Act (the “Penny Stock Rules”). The Penny Stock Rules require a broker-dealer, prior to a transaction in a penny stock, to deliver a standardized risk disclosure document prepared by the SEC, that:
| ● | contains a description of the nature and level of risk in the market for penny stocks in both public offerings and secondary trading; |
| ● | contains a description of the broker’s or dealer’s duties to the customer and of the rights and remedies available to the customer with respect to a violation to such duties or other requirements of securities laws; |
| ● | contains a brief, clear, narrative description of a dealer market, including bid and ask prices for penny stocks and the significance of the spread between the bid and ask price; |
| ● | contains a toll-free telephone number for inquiries on disciplinary actions; |
| ● | defines significant terms in the disclosure document or in the conduct of trading in penny stocks; and |
| ● | contains such other information and is in such form, including language, type, size and format, as the SEC shall require by rule or regulation. |
20
The broker-dealer also must provide, prior to effecting any transaction in a penny stock, the customer with: (a) bid and offer quotations for the penny stock; (b) the compensation of the broker-dealer and its salesperson in the transaction; (c) the number of shares to which such bid and ask prices apply, or other comparable information relating to the depth and liquidity of the market for such stock; and (d) a monthly account statements showing the market value of each penny stock held in the customer’s account. In addition, the penny stock rules require that, prior to a transaction in a penny stock not otherwise exempt from those rules, the broker-dealer must make a special written determination that the penny stock is a suitable investment for the purchaser and receive the purchaser’s written acknowledgment of the receipt of a risk disclosure statement, a written agreement to transactions involving penny stocks, and a signed and dated copy of a written suitability statement. These disclosure requirements may have the effect of reducing the trading activity in the secondary market for our stock if it becomes designated as a Penny Stock.
Because we do not intend to pay any cash dividends on our common stock, our stockholders will not be able to receive a return on their shares unless they sell them.
We intend to retain any future earnings to finance the development and expansion of our business. We do not anticipate paying any cash dividends on our common stock in the foreseeable future. Unless we pay dividends, our stockholders will not be able to receive a return on their shares unless they sell them. There is no assurance that stockholders will be able to sell shares when desired.
Risks Related to Equity Offerings
Because the offering price for the primary shares has been arbitrarily set by us, you may not realize a return on your investment upon the resale of your shares.
The offering price of each share and other terms and conditions relative to the shares being sold in our past IPO and potential future offerings not a part of an underwritten offering have been arbitrarily determined by us and do not bear any relationship to assets, earnings, book value or any other objective financial criteria. Additionally, as the Company was formed on May 23, 2006, and has only a limited operating history with no earnings, the price of the offered shares in future offerings is not based on its past earnings, and no investment banker, appraiser, or other independent third party, has been consulted concerning the offering price for the shares or the fairness of the offering price used for the shares, as such our stockholders may not be able to receive a return on their investment when they sell shares of common stock purchased historically.Additionally, our last offering terminated on April 12, 2019, and we no longer have the ability to utilize this method for the sale of our equity to investors, unless we were to seek registration under a new S-1 filing, which could hamper our overall ability to raise capital which could have a material adverse effect on our operations.
Our officers and directors have limited prior experience offering and selling securities to the public, and as a result, we may not be able to raise sufficient funds to sustain our business.
Certain of our officers and directors participated in selling shares in our IPO, where allowed to do so under applicable state blue sky laws, on our behalf. We conducted a “best efforts” offering under our Registration Statement which does not require a minimum amount to be raised, and there is no guarantee sufficient funds will be raised to sustain our business. Also, the Offering terminated on April 12, 2019.
Our officers and directors have limited experience conducting a publicly registered securities Offerings. If we are not able to raise sufficient funds in the future, we may not be able to fund our operations as planned, and we may be forced to curtail or cease operations and investors could lose all or a significant part of their investment. Our inability to successfully conduct an offering could be the basis for investors losing all or a significant part of their investment in us.
21
Due to the lack of any quotation or trading market for our securities, you may have difficulty selling any shares you purchase in our IPO.
We are not registered on any market or public stock exchange. There is presently no demand for our common stock and no public market exists for our shares. In the future, we intend to seek a listing or a quotation on a Qualified National Exchange or the OTC Markets. However, we cannot make any assurance that our common stock will ever be quoted or traded on a Qualified National Exchange or the OTC Markets or that any public market for our stock will develop. In order to achieve a Public Listing, we will have to meet certain initial listing qualifications of the Qualified National Exchange or the OTC Markets on which we are seeking the Public Listing. In addition, we will need to have market makers agree to make a market in our common stock and file a FINRA Form 15c211 with the SEC on our behalf before we can achieve a Public Listing. As of the date of this Annual Report on Form 10-K, there are no assurances or understandings between the Company and anyone acting on our behalf, with any market maker regarding participation in a future trading market for our securities. If we are successful in achieving a Public Listing for our common stock, we will also need to remain current in our quarterly and annual filings with the SEC to achieve and maintain such Public Listing. Market makers are not permitted to begin quotation of a security whose issuer does not meet these filing requirements. Furthermore, if we are not able to pay the expenses associated with our ongoing reporting obligations, we will not be able to achieve or maintain a Public Listing. If no public market is ever developed for our common stock, it will be difficult for investors to sell any of our shares. In such a case, investors may find that they are unable to achieve any benefit from their investment or liquidate their shares without considerable delay, if at all. In addition, if we fail to have our common stock quoted on a public trading market, any common stock purchased by investors will not have a quantifiable value and it may be difficult, if not impossible, to ever resell their shares, resulting in an inability to realize any value from their investment.
ITEM 1B. UNRESOLVED STAFF COMMENTS
None.
We lease three separate facilities. Administrative offices and a manufacturing operation are in a 25,000 square foot building with an operating lease that expires in December 2022. Research and development laboratories are in a 14,300 square foot space with an operating lease that extends to December 2022. In October 2017, we signed a lease for a 64,000 square foot manufacturing facility, the site on which we expanded our mechanical exfoliation capacity. This facility replaced the 6,600 square foot facility in the early part of 2018. The term of the operating lease for the new facility is 5 years expiring December 2022. All of the physical assets of the Company are contained within these facilities. XG Sciences has achieved and maintains an ISO9001:2015 quality certification for all of its locations.
None.
ITEM 4. MINE SAFETY DISCLOSURES
Not applicable.
22
| ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Market for Common Equity
No public market currently exists for our shares and a public market may never develop, or, if any market does develop, it may not be sustained.
Following our Offering which terminated on April 12, 2019, we now intend to seek either (i) a listing of our common stock on a securities exchange registered with the SEC under Section 6(a) of the Exchange Act, such as the NASDAQ Capital Market or the NYSE, or (ii) the quotation of our common stock on the OTCQB or OTCQX marketplaces operated by OTC Markets Group, Inc., market conditions permitting. In order to achieve such a Public Listing, we will have to meet certain initial listing qualifications of the Qualified National Exchange or the OTC Markets on which we are seeking the Public Listing. In addition, we will need to have market makers agree to make a market in our common stock and file a FINRA Form 15c211 with the SEC on our behalf before we can achieve a Public Listing, and we will also need to remain current in our quarterly and annual filings with the SEC. As of the date of this report, there are no assurances or understandings between the Company and anyone acting on our behalf, with any market maker regarding participation in a future trading market for our securities. There can be no assurance that our common stock will ever be quoted or traded on a Qualified National Exchange or the OTC Markets or that any market for our common stock will develop.
Holders of Equity
As of April 29, 2020, we had 385 record holders of our common stock, and a total of 4,024,443 shares of common stock issued and outstanding. We had 13 record holders of Series A Preferred Stock and a total of 1,890,354 shares of Series A Preferred Stock issued and outstanding. In total, we had 390 shareholders of record of all classes of our capital stock as of April 29, 2020.
Each share of Series A Preferred Stock is voluntarily convertible, at the option of the holder thereof, at any time after the date of issuance and prior to any Public Listing of common stock on a Qualified National Exchange, into that number of fully paid, non-assessable shares of common stock determined by dividing the Original Issue Price by the Conversion Price then in effect, as such terms are defined in the Series A Designations. The current Series A Conversion Rate is 1.875 shares of common for 1 share of Series A. The Conversion Price of the Series A Preferred Stock is subject to adjustments pursuant to the occurrence of stock splits and certain other specified events, and therefore the respective conversion rates are subject to change.
Furthermore, under the terms of the Series A Designations (as amended), all outstanding shares of Series A Preferred Stock will automatically convert into shares of common stock upon a Public Listing on a Qualified National Exchange at the then-current Series A Conversion Rate. At the current Series A Conversion Rate, if all outstanding shares of Series A Preferred Stock were voluntarily converted or automatically converted, we would issue 3,544,408 shares of common stock to satisfy such conversions.
As of December 31, 2019, the Company had granted options to purchase up to 839,625 shares of common stock and had issued warrants to purchase up to (i) 487,017 shares of common stock (including the warrants for 225,000 shares issued under the Dow Facility) and (ii) 972,720 shares of Series A Preferred Stock which, if exercised, would be convertible into 1,823,845 shares of common stock at the then-current Series A Conversion Rate (currently of 1.875 shares of common for 1 share of Series A). Therefore, we have committed to issue up to an additional 6,694,895 shares of common stock, which includes the issuance of (a) 3,544,408 shares upon conversion of all 1,890,354 shares Series A Preferred Stock currently outstanding at the Series A Conversion Rate, (b) 1,823,845 shares upon the conversion of 972,720 shares of Series A Preferred Stock (at the current Series A Conversion Rate) which are issuable upon exercise of 972,720 Series A warrants, (c) 839,625 shares upon the exercise of options and (d) the issuance of 487,017 shares upon the exercise of warrants. If we issued all 6,694,895 shares, we would have, including the 4,024,443 shares currently outstanding, 10,719,338 shares issued and outstanding, with 14,280,662 authorized shares available for future issuance. The future issuance of common stock may result in substantial dilution in the percentage of our common stock held by our then existing stockholders. Except with respect to the IPO (which was priced at $8.00 per share), we may value any common stock issued in the future on an arbitrary basis. The issuance of common stock for future services or acquisitions or other corporate actions may have the effect of diluting the value of the shares held by our investors, might have an adverse effect on any future trading market for our common stock and could impair our ability to raise capital in the future through the sale of equity securities.
23
Securities Authorized for Issuance under Equity Compensation Plans
As of December 31, 2019, and 2018, the Company had outstanding stock options to purchase up to 839,625 and 797,875 shares of common stock, respectively.
On September 30, 2019 and September 30, 2018, the Company granted each Board member 2,500 stock options and 2,500 shares of restricted stock for their Board services. The options were granted at a price of $8.00 per share and vest ratably over a four-year period beginning on the one-year anniversary. The options had an aggregate grant date fair value of $38,295 and $29,580 on September 30, 2019 and, September 30, 2018, respectively. The restricted stock issued to the Board members in 2019 and 2018 has an aggregate fair value of $180,000 and vests ratably in arrears over four quarters on the last day of each fiscal quarter following the grant date. As of December 31, 2019 and December 31, 2018, 26,250 and 15,000 of the shares of restricted stock issued had vested, resulting in compensation expense of $90,000 and $80,000 for the years ended December 31, 2019 and December 31, 2018, respectively.
During the year ended December 31, 2019 and December 31, 2018, we granted 70,000 and 138,250 stock options, respectively, to employees with an exercise price of $8.00 and a seven-year term. The aggregate fair value of the awards on the date of grant was $177,111 and $408,237, respectively, as of December 31, 2019 and December 31, 2018. The stock options vest equally over four years beginning on the first anniversary of the date of grant.
As of December 31, 2019, stock options to purchase a total of 839,625 shares at a price of $8.00 per share had been granted to Company employees and Directors, with expiration dates ranging from July 2024 to December 2026.
24
Equity Compensation Plan as of December 31, 2019
| Equity Compensation Plan Information | ||||||||||||
| Plan category | Number of securities to be issued upon exercise of outstanding options, warrants and rights (a) | Weighted- average exercise price of outstanding options, warrants and rights (b) | Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in column (a) (c) | |||||||||
| Equity compensation plans approved by security holders(1) | 839,625 | $ | 8.00 | 360,375 | ||||||||
| Equity compensation plans not approved by security holders | — | — | — | |||||||||
| Total | 839,625 | $ | 8.00 | 360,375 | ||||||||
| (1) | Reflects our 2017 Plan, for the benefit of our directors, officers, employees and consultants as of December 31, 2019. As of December 31, 2019, we had reserved 1,200,000 shares of common stock for such persons pursuant to that plan and had granted stock options to purchase 839,625 shares at a price of $8.00 with 360,375 shares remaining available for future issuance. |
DIVIDEND POLICY
Since inception, we have not paid any dividends on our common stock. We currently do not anticipate paying any cash dividends in the foreseeable future on our common stock, because we intend to retain our earnings, if any, to finance the growth of our business. Our Board of Directors will have the discretion to declare and pay dividends in the future. Payment of dividends in the future will depend upon our earnings, capital requirements, and other factors, which our Board of Directors may deem relevant.
UNREGISTERED SALES OF EQUITY SECURITIES
During the period from May 17, 2016 through December 31, 2018 we issued shares of Series A Preferred Stock to AAOF as payment for lease financing obligations under the terms of a Master Leasing Agreement as detailed below.
Shares of Series A Preferred Stock Issued to AAOF as Lease Financing Payments
| ||||||||||
| Date | Shares | Date | Shares | Date | Shares | |||||
| 5/17/2016 | 7,140 | 1/18/2017 | 7,140 | 3/26/2018 | 7,140 | |||||
| 5/17/2016 | 7,140 | 4/3/2017 | 7,140 | 4/2/2018 | 7,140 | |||||
| 8/17/2016 | 4,760 | 7/3/2017 | 7,140 | 7/2/2018 | 2,716 | |||||
| 9/30/2016 | 2,380 | 10/3/2017 | 7,140 | 7/16/2018 | 7,947 | |||||
| 10/1/2016 | 7,140 | 10/4/2018 | 2,716 | |||||||
| 12/31/2018 | 4,879 | |||||||||
| Total | 28,560 | 28,560 | 32,538 | |||||||
With the final issuance of Series A Preferred Stock to AAOF on December 31, 2018, all obligations under the terms of the Master Lease have been fulfilled and the Master Lease has expired.
On December 14, 2016, we issued a promissory note in the amount of $2,000,000 and warrants to purchase 50,000 shares of common stock of the Company to Dow pursuant to the Dow Facility. The notes are non-convertible and mature on December 1, 2021. The warrants have a seven-year maturity date with a strike price of $8.00 per share.
On July 18, 2017, September 22, 2017, and December 4, 2017 we issued promissory notes in the amount of $1,000,000 and warrants to purchase 25,000 shares of common stock, respectively for each date ($3,000,000 in total for the notes and 75,000 for the warrants) to Dow pursuant to the Dow Facility. The notes are non-convertible and mature on December 1, 2021. The warrants have a seven-year maturity date with a strike price of $8.00 per share.
On July 8, 2019 and November 11, 2019, we issued promissory notes in the amounts of $2,000,000 each and warrants to purchase 50,000 shares of common stock, respectively for each date ($4,000,000 in total for the notes and 100,000 for the warrants) to Dow pursuant to the Dow Facility. The notes are non-convertible and mature on December 1, 2021. The warrants have a seven-year maturity date with a strike price of $8.00 per share.
25
ITEM 6. SELECTED FINANCIAL DATA
Not applicable.
| ITEM 7. | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
This section includes a number of forward-looking statements that reflect our current views with respect to future events and financial performance. Forward-looking statements are often identified by words like “believe”, “expect”, “estimate”, “anticipate”, “intend”, “project” and similar expressions, or words which, by their nature, refer to future events. You should not place undue certainty on these forward-looking statements. These forward-looking statements are subject to certain risks and uncertainties that could cause actual results to differ materially from our predictions.
Overview of our Business
XG Sciences was formed in May 2006 for the purpose of commercializing certain technology to produce graphene nanoplatelets and products containing graphene nanoplatelets. First isolated and characterized in 2004, graphene is a single layer of carbon atoms configured in an atomic-scale honeycomb lattice. Among many noted properties, monolayer graphene is harder than diamonds, lighter than steel but significantly stronger, and conducts electricity better than copper. Graphene nanoplatelets are particles consisting of multiple layers of graphene. Graphene nanoplatelets have unique capabilities for energy storage, thermal conductivity, electrical conductivity, barrier properties, lubricity and the ability to impart strength when incorporated into plastics or other matrices.
We believe the unique properties of graphene and graphene nanoplatelets will enable numerous new product applications and the market for such products will grow to be a significant market opportunity. Our business model is to design, manufacture and sell advanced materials we call xGnP®graphene nanoplatelets and value-added products based on these nanoplatelets. We currently have hundreds of customers trialing our products for numerous applications, including, but not limited to lithium ion batteries, lead acid batteries, thermally conductive adhesives, composites, thermal management and heat transfer, inks and coatings, printed electronics, construction materials, cement, and in a range of other industrial uses. We believe our proprietary processes have enabled us to be among the low-cost producers of high quality, graphene nanoplatelets and value-added products containing graphene nanoplatelets and that we are well positioned to address a wide range of end-use applications.
We sell products to customers around the world and have sold materials to over 1,300 customers in 47 countries since 2008. Some of these customers are research organizations and some are commercial organizations. Our customers have included well-known automotive and OEM suppliers around the world (Ford, Johnson Controls, Magna, Honda Engineering), world-scale lithium ion battery manufacturers in the US, South Korea and China (Samsung SDI, LG Chemical, Lishen, A123), diverse specialty material companies (3M, BASF, Henkel, Dow, DuPont, Callaway), and leading research centers such as Lawrence Livermore National Laboratory and Oakridge National Laboratory. We have also licensed some aspects of our base manufacturing technology to other companies. Our licensees include POSCO, the fifth largest steel manufacturer in the world by tonnage of output (as reported by Worldsteel Associates in “World Steel in Figures 2018”), and Cabot Corporation (“Cabot”), a leading global specialty chemicals and performance materials company. These licensees further extend our technology through their customer networks.
We target our xGnP® nanoplatelets for use in a range of large and growing end-use markets. Our proprietary manufacturing processes allow us to produce nanoplatelets with varying performance characteristics that can be tuned to specific end-use applications based on customer requirements. We manage our business with five major product lines at this time:
| 1. | Bulk Materials. We sell bulk materials under the trademarked brand name of xGnP®graphene nanoplatelets. These materials are produced in various grades, which are analogous to average platelet thickness, and average platelet diameters. There are three commercial grades (Grades H, M & R), each of which is offered in three standard platelet diameters and a fourth, C Grade, which is offered in three standard surface areas. We also have access to other development grades (Grade T, for example), but which are not yet made available commercially and have been used internally for those products containing graphene nanoplatelets. These bulk materials, which normally ship in the form of a dry powder, are especially applicable for use as additives in polymeric or metallic composites, or in coatings or other formulations where particular electrical, thermal or barrier performance are desired by our customers. We also offer our material in the form of dispersions of nanoplatelets in liquids such as water, alcohol, or organic solvents, or mixed into resins or polymers such as thermoplastics or thermosets. |
26
| 2. | Composites. Our composites, integrated-products portfolio includes resins derived from a range of thermoplastics and thermosets into which our xGnP® graphene nanoplatelets are compounded. These products are supplied as master batches of resins with nanoplatelets pre-dispersed such that their performance has been optimized for use in various end-use applications that may include industrial, automotive and sporting goods and which have demonstrated efficacy in standard injection molding, compression molding, blow molding and 3-D processes, to name but a few. Our current product portfolio of polymer resins containing various forms of our xGnP®graphene nanoplatelets and in varying concentration includes polyurethane (XGPU), polypropylene (XGPP), polyethylene terephthalate (XGPET), vinyl ester (XGVE), polyetherimide (XGPEI) and high-density polyethylene (XGHDPE). Other polymers may be added over time depending on the end-market and customer needs. In addition, we offer various bulk materials with demonstrated efficacy in plastic composites to impart improved physical performance to such matrices, which may be supplied as dry powders or as aqueous-based or solvent-based dispersions or cakes as described previously. We have also targeted use of our graphene nanoplatelets as an additive in cement mixtures, which we believe results in improved barrier resistance, durability, toughness and corrosion protection. Our GNP Concrete Additive product promotes the formation of more uniform and smaller-grain cement structure, improving both flexural and compressive strength. In addition, the embedded graphene nanoplatelets will stop cracks from forming and retard crack propagation, should any cracks form – the combination of which will further improve cement lifetime and durability. |
| 3. | Inks and Coatings. These consist of specially formulated dispersions of xGnP®together with solvents, binders, and other additives to make electrically or thermally conductive products designed for printing or coating and which are showing promise in diverse customer applications such as advanced packaging, electrostatic dissipation and thermal management. We also offer a set of standardized ink formulations suitable for printing. These inks offer the capability to print electrical circuits or antennas and may be suitable for other electrical or thermal applications and can be further customized for specific customer requirements. |
| 4. | Energy Storage Materials. These consist of specialty advanced materials that have been formulated for specific applications in the energy storage segment. We offer various bulk materials for use as conductive additives for cathodes and anodes in lithium-ion batteries, as additives to anode slurries for lead-carbon batteries, as a component in coatings for current collectors used in lithium-ion batteries and we are investigating the use of our materials as part of other energy-storage components. |
| 5. | Thermal Management Materials. These consist mainly of various thermal interface materials (“TIM’s”) in the form of custom greases or pastes. Our custom thermal interface materials are also designed to be used in various high temperature environments. Additionally, we offer various bulk materials for use as active components in adhesives, liquids, coatings and plastic composites to impart improved thermal management performance to such matrices. |
Because graphene is a new material, many of our customers are still developing applications that use our products, and thus historically those customers have purchased products in quantities consistent with development purposes, although some are now purchasing in quantities consistent with larger volume commercial production. The process of “designing-in” new materials is a relatively complex one and involves the use of relatively small amounts of the new material in laboratory and engineering development for an extended period. Following successful development, we believe customers that incorporate our materials into their products will then order much larger quantities of material to support commercial production. Thus, while many of our customers are currently purchasing our materials in kilogram (one or two pound) quantities, and some have begun to purchase in metric tons, we believe many will require tens of tons or even hundreds of tons of material when they commercialize products that incorporate our materials. Although, our customers are under no obligation to report to us on the usage of our materials, some have indicated that they have introduced or will soon introduce commercial products that use our materials.
27
We are tracking the commercial and development status of more than 75 different customer applications using our materials with some customers pursuing multiple applications. As of December 31, 2019, we had twenty-two (22) specific customer applications where our materials are incorporated into our customers’ products and such customers are actively selling these products to their own customers. In addition, we have another seventeen (17) customer applications where our customers have indicated that they expect to begin shipping product incorporating our materials in the next 3 – 6 months and have another twenty-six (26) customer applications where our customers have indicated an intent to commercialize in the next 6 – 9 months. We are also working with numerous additional customers that have not yet indicated an exact date for commercialization, but we believe have the potential to contribute to meaningful revenue in 2020 and 2021. The following graphic demonstrates the commercialization trends over the past 8 fiscal quarters as an increasing number of customers indicate their intent to commercialize applications and move into actively selling or promoting products for future sales.
Operating Segment
We have one operating segment that manufactures xGnP® graphene nanoplatelets and value-added products produced therefrom, we conduct research on graphene nanoplatelets and related products, and we license our technology, as appropriate. As of December 31, 2019, we shipped products on a worldwide basis, but all of our assets were located within the United States.
Our Critical Accounting Policies
US generally accepted accounting principles (“GAAP”) requires management to make estimates, judgments and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses, together with amounts disclosed in the related notes to the consolidated financial statements. Actual results and outcomes may differ from management’s estimates, judgments and assumptions. Significant estimates, judgments and assumptions used in our consolidated financial statements include, but are not limited to, those related to revenues, the fair values of stock-based compensation, derivative financial instrument liabilities, and liquidity. These estimates, judgments, and assumptions are reviewed periodically and the effects of material revisions in estimates are reflected in the consolidated financial statements prospectively from the date of the change in estimate.
Revenue Recognition
On January 1, 2018, the Company adopted ASU No. 2014-09,Revenue from Contracts with Customers (Topic 606), which supersedes nearly all previous revenue recognition guidance.
Revenues are recognized at a point in time, typically when control of the promised goods is transferred to customers, in an amount that reflects the consideration the Company expects to be entitled to in exchange for those goods. The Company does not recognize revenue in cases where collectability is not probable, and defers the recognition until collection is probable or payment is received.
Customer deposits, deferred revenue and other receipts are deferred and recognized when the revenue is realized and earned.
Revenue related to licensing agreements is recorded upon substantial performance of the terms of the licensing contract. In the case of licensing arrangements that involve up-front payments, revenue is recorded when management determines that the appropriate terms of the contract have been fulfilled. For example, this may occur when technology has been transferred via written documents or, if training is involved, whenever all contracted training has occurred. In the case of licenses where product delivery is also embedded in the deliverable, a portion of revenue would be recognized when products are delivered.
In the case of licensing arrangements that involve ongoing royalties based on sales of products produced with our technology, royalty income is recorded when received or, in the case of minimum royalties due, in the period when due.
Grant contract revenue is recognized over the life of the contracts as the services are performed.
Amounts received in excess of revenues earned are recorded as deferred revenue.
Stock-Based Compensation
We recognize compensation expense in our statement of operations for all share-based option and stock awards, based on estimated grant-date fair values.
28
We estimate the grant-date fair value of stock-based compensation awards using the Black-Scholes option valuation model. This model is affected by the estimated value of our common stock on the date of the grant as well as assumptions regarding a number of highly complex and subjective variables. These variables include the expected term of the option, the exercise price, expected risk-free rates of return, the expected volatility of our common stock, and expected dividend yield, each of which is more fully described below. The assumptions for the estimated value of our common stock, expected term and expected volatility are the assumptions that most significantly affect the grant date fair value.
Expected Term: Because we have limited experience related to the exercise of employee stock options, we use the simplified method permitted by SEC Staff Accounting Bulletin Topic 14 to estimate the expected term of the options. The expected term of an option is estimated to be equal to the mid-point between the vesting and expiration dates of the option.
Risk-free Interest Rate: We base the risk-free interest rate used on the implied yield at the grant date of U.S. Treasury zero-coupon issues with a term approximately equal to the expected term of the stock-based award being valued.
Expected Stock Price Volatility: Because we are a company with very limited stock sales history (we had an Offering whereby we are selling stock directly to investors without the assistance of an investment bank nor are we registered on any market or public exchange), we use a blended average weekly volatility of certain publicly traded peer companies. We believe that the use of this blended average peer volatility is reflective of market conditions and a reasonable indicator of our expected future volatility.
Dividend Yield: Because we have never paid a dividend and do not expect to begin doing so in the foreseeable future, we have assumed a 0% dividend yield in valuing our stock-based awards.
The grant-date fair value of the award is recognized as expense over the requisite service period using the straight-line method.
Fair Value Measurements
FASB ASC 820: “Fair Value Measurements and Disclosures” defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. ASC 820 also establishes a fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure fair value:
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Inputs other than quoted prices included within Level 1 which are either directly or indirectly observable.
Level 3: Inputs to the valuation methodology are unobservable and significant to the fair value measurement.
A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
Derivative Financial Instruments
We do not use derivative instruments to hedge exposures to cash flow, market or foreign currency risk. The terms of convertible preferred stock and convertible notes that we have issued in the past were reviewed to determine whether or not they contain embedded derivative instruments that are required by ASC 815: “Derivatives and Hedging” to be accounted for separately from the host contract and recorded at fair value. In addition, freestanding warrants are also reviewed to determine if they achieve equity classification.
Recent Accounting Pronouncements
In December of 2019, the FASB issued Accounting Standards Update (ASU) 2019-12, Accounting for Income Taxes. The new standard simplifies the accounting for income taxes and is effective for fiscal years beginning after December 15, 2020..
29
Adoption of Lease Accounting Policy
We applied ASU 2016-02 and all related amendments (“ASC 842”) using the modified retrospective method by recognizing the cumulative effect of adoption as an adjustment to the opening balance of retained earnings at January 1, 2019. Therefore, the comparative information has not been adjusted and continues to be reported under prior leasing guidance. As a result, in the first quarter of 2019 we recorded ROU assets of $1,871,366. We also recorded lease liabilities of $1,981,795. The decrease to retained earnings was $116,319, reflecting the cumulative impact of the accounting change. The standard did not have a material effect on consolidated net income or cash flows.
Right-of-use assets represent our right to use an underlying asset for the lease term and lease liabilities represent our obligation to make lease payments arising from the lease. As our leases do not provide an implicit rate, we used our incremental borrowing rate based on the information available at the commencement date in determining the present value of lease payments. Lease expense for lease payments is recognized on a straight-line basis over the lease term.
We do not record a ROU asset or lease liability for leases with an expected term of 12 months or less. The comparative information has not been adjusted and continues to be reported under prior leasing guidance.
With the exception of the standards discussed above, we believe there have been no new accounting pronouncements effective or not yet effective which have significance, or potential significance, to our Consolidated Financial Statements.
JOBS Act
In April 2012, the Jumpstart Our Business Startups Act of 2012, or the JOBS Act, was enacted. Section 107 of the JOBS Act provides that an “emerging growth company,” or EGC, can take advantage of the extended transition period for complying with new or revised accounting standards. Thus, an EGC can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have irrevocably elected not to avail ourselves of this extended transition period and, as a result, we will adopt new or revised accounting standards on the relevant dates on which adoption of such standards is required for other public companies.
We are in the process of evaluating the benefits of relying on other exemptions and reduced reporting requirements under the JOBS Act. Subject to certain conditions, as an EGC, we intend to rely on certain exemptions, including exemptions from the requirement to provide an auditor’s attestation report on our system of internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and from any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion and analysis. We will remain an EGC until the earlier of: the last day of the fiscal year in which we have total annual gross revenues of $1.07 billion or more; the last day of the fiscal year following the fifth anniversary of the date of the completion of our Offering; the date on which we have issued more than $1.0 billion in nonconvertible debt during the previous three years; or the date on which we are deemed to be a large accelerated filer under the rules of the SEC.
30
Results of Operations for the Year Ended December 31, 2019 Compared with the Year Ended December 31, 2018
The following table summarizes the results of our operations for the years ended December 31, 2019 and 2018.
| Year Ended December 31, | Change 2019 to 2018 | |||||||||||||||
| 2019 | 2018 | $ | % | |||||||||||||
| Total Revenues | 1,369,556 | 4,301,820 | (2,932,264 | ) | -68.2 | % | ||||||||||
| Cost of Goods Sold | 3,763,473 | 5,506,321 | 1,742,848 | 31.7 | % | |||||||||||
| Gross Loss | (2,393,917 | ) | (1,204,501 | ) | (1,189,416 | ) | -98.7 | % | ||||||||
| Research & Development Expense | 1,506,716 | 1,414,259 | (92,457 | ) | -6.5 | % | ||||||||||
| Sales, General & Administrative Expense | 5,467,698 | 4,972,595 | (495,103 | ) | -10.0 | % | ||||||||||
| Total Operating Expenses | 6,974,414 | 6,386,854 | (587,560 | ) | -9.2 | % | ||||||||||
| Operating Loss | (9,368,331 | ) | (7,591,355 | ) | (1,776,976 | ) | -23.4 | % | ||||||||
| Other Expense | 409,318 | 328,261 | (81,057 | ) | -24.7 | % | ||||||||||
| Net Loss | (9,777,649 | ) | (7,919,616 | ) | (1,858,033 | ) | -23.5 | % | ||||||||
Revenues
Revenues for the years ended December 31, 2019 and 2018, by category, are shown below.
| Year Ended December 31, | Change 2019 to 2018 | |||||||||||||||
| 2019 | 2018 | % | ||||||||||||||
| Product Sales | $ | 1,369,556 | $ | 4,125,567 | $ | (2,756,011 | ) | (66.8 | ) | |||||||
| Grants | - | 176,253 | (176,253 | ) | (100.0 | ) | ||||||||||
| Total Revenues | $ | 1,369,556 | $ | 4,301,820 | $ | (2,932,264 | ) | (68.2 | ) | |||||||
Product sales consist of two broad categories: (1) material sold to customers for research or development purposes; and (2) production orders for customers. Typically, the order sizes for the first category are relatively small, however we expect orders in the second category to be much larger in the future. For the year ended December 31, 2019, product sales decreased by $2,756,011 or 67% from the prior year. The decrease in revenue for the year ended December 31, 2019 is due to our largest customer suspending orders during the period. The customer has not stopped using our products, but rather, is working down material ordered and delivered in the second half of 2018 and currently held in inventory and being used to produce their products. This customer continues to ship their products incorporating our material and we expect that will continue working down existing inventory and will resume purchasing our products at historical levels in the coming year. In addition, other customers have been moving through development programs towards commercialization, requiring larger quantities of our materials for advanced testing, pilot production and commercial-scale production activities. We believe that those customers already in production will increase their order volume as demand increases and others will begin to move into commercial volume production as they gain more experience in working with our materials and engage their own customers.
We ship our products from our Lansing, MI manufacturing facilities to customers around the world. During the year ended December 31, 2019, we shipped materials to customers in 29 countries, as compared to 23 countries during the same period in 2018. For the year ended December 31, 2019, sales to two foreign countries, South Korea and Switzerland, accounted for more than 10% of revenue. For the year ended December 31, 2018, no product sales to any one country accounted for more than 10% of product sales.
Order Summary
The table below shows a comparison of domestic and international orders fulfilled (note that this does not include orders for free samples). The table also includes the average order size for product sales. These numbers indicate that our customer base remains active with research and development projects that use our materials. The average order size for the product revenue during the year ended December 31, 2019 decreased by 56% as compared to the same period in 2018 primarily due to a 2019 decrease in orders from one customer while working down their inventory.
| Year Ended December 31 | Change 2019 to 2018 | |||||||||||||||
| 2019 | 2018 | # of Orders | % | |||||||||||||
| Number of orders - domestic | 119 | 122 | (3 | ) | -2 | % | ||||||||||
| Number of orders - international | 99 | 168 | (69 | ) | -41 | % | ||||||||||
| Number of orders - Total | 218 | 290 | (72 | ) | -25 | % | ||||||||||
| Average order size - $s | $ | 6,282 | $ | 14,303 | $ | (8,021 | ) | -56 | % | |||||||
31
Grant Revenue
Grant revenues of $176,253 in 2018 consisted of proceeds from sources as shown in the table below. The largest of these sources in 2018 came from the State of Michigan. In 2018, an agreement was initiated with the Michigan Strategic Fund relating to our new manufacturing facility in Mason, Michigan. Grant contract revenue was recognized at a point in time when the first increment of qualified new jobs over the base employment level as defined by the grant was met in 2018 and recorded as grant revenue in the amount of $173,000.
| Year Ended December 31 | ||||||||
| 2019 | 2018 | |||||||
| State of Michigan | $ | — | $ | 173,000 | ||||
| US Department of Energy | — | — | ||||||
| Daimler / University of Michigan | — | — | ||||||
| Michigan Economic Development Corporation | — | 3,253 | ||||||
| Total | $ | — | $ | 176,253 | ||||
Licensing Revenue
There were no licensing agreements in place nor amounts due and recorded on our balance sheet for Licensing Revenue at December 31, 2019 or December 31,2018.
Cost of Goods Sold
We use a standard cost system to estimate the direct costs of products sold. Variances between the standard costs and the actual costs are then properly capitalized to the inventories and expensed to cost of goods sold. Direct costs include estimates of raw material costs, packaging, freight charges net of those billed to customers, and an allocation for direct labor and manufacturing overhead. Because of the nature of our production processes, there is a substantial fixed manufacturing expense requirement that represents the ongoing cost of maintaining production facilities that are not directly related to products sold, so we use a “full capacity” allocation of overhead based on an estimate of what product costs would be if the manufacturing facilities were operating on a full-time basis and producing products at the designed capacity.
Gross Profit Summary
The following table shows the relationship of direct costs to product sales for the years ended December 31, 2019 and 2018:
| Year Ended December 31, | Change 2019 to 2018 | |||||||||||||||
| 2019 | 2018 | $ | % | |||||||||||||
| Product Sales | $ | 1,369,556 | $ | 4,125,567 | $ | (2,756,011 | ) | (66.8 | ) | |||||||
| Direct Costs | 1,006,594 | 2,344,937 | (1,338,343 | ) | (57.1 | ) | ||||||||||
| Direct Cost Margin | $ | 362,962 | $ | 1,780,630 | $ | (1,417,668 | ) | (79.6 | ) | |||||||
| % of Sales | 26.5 | % | 43.2 | % | ||||||||||||
| Unallocated Manufacturing Expense | 2,756,879 | 3,161,384 | (404,505 | ) | (12.8 | ) | ||||||||||
| Gross Loss on Product Sales | $ | (2,393,917 | ) | $ | (1,380,754 | ) | $ | (1,013,163 | ) | 73.4 | ||||||
32
We believe that the fluctuations in gross loss on product sales and direct cost from period to period are not indicative of future margins because of the relatively small size of our sales in comparison to our future expectations. Direct costs vary depending on the size of an order, the specific products being ordered, and other factors like shipping destination (which on small orders can represent a significant percentage of the cost).
The remaining “non-direct” costs of operating our manufacturing facilities are recorded as unallocated manufacturing expenses. These expenses include personnel costs, rent, utilities, indirect supplies, depreciation, and related indirect expenses. Unallocated manufacturing expenses are expensed as incurred. We allocate these costs to direct product costs based on the proportion of these expenses that would be representative direct product costs if we were operating our factory at full capacity.
For the year ended December 31, 2019, unallocated manufacturing expenses decreased by 13% to $2,756,879 as compared to $3,161,384 in 2018. The decrease of $404,505 is largely due to lower levels of manufacturing expenses as we adjusted to a lower volume of commercial orders versus the prior year.
Research and Development Expenses
Research and development expenses for the year ended December 31, 2019 increased by 7% to $1,506,717 as compared to $1,414,259 for the year ended December 31, 2018. The increase of $92,458 is largely due to the hiring of additional personnel to increase our core ability to deliver improved applications to customers.
Sales, General and Administrative Expenses
During 2019, we incurred selling, general and administrative expenses (SG&A) of $5,467,698. This is an increase of $703,903 or 14% from 2018, which represents an increase in sales personnel to support increased customer programs as well as the addition of our Chief Technologist.
Other Income (Expense)
Interest expense, net of interest income in the year ended December 31, 2019, increased by $81,057 compared to 2018. The net increase in expense reflects interest due on the increased Dow Facility balance which was partially offset by principal repayments and the elimination of interest expense on the capital lease which ended December 31, 2018.
33
Cash Flow Summary
The following condensed cash flow statement compares cash flow from operating, investing, and financing activities for the year ended 2019 and 2018.
| Year Ended December 31, | Change 2019 to 2018 | |||||||||||||||
| 2019 | 2018 | $ | % | |||||||||||||
| Cash, cash equivalents and restricted cash at beginning of period | 4,893,974 | 3,041,590 | 1,852,384 | 60.9 | % | |||||||||||
| Net Cash provided (used by): | ||||||||||||||||
| Operating activities | (8,589,598 | ) | (5,643,238 | ) | (2,946,360 | ) | -52.2 | % | ||||||||
| Investment activities | (514,327 | ) | (3,385,114 | ) | 2,870,787 | -84.8 | % | |||||||||
| Finance activities | 5,339,653 | 10,880,736 | (5,541,083 | ) | -50.9 | % | ||||||||||
| Net increase (decrease) in cash | (3,764,272 | ) | 1,852,384 | (5,616,656 | ) | -303.2 | % | |||||||||
| Cash, end of period | 1,129,702 | 4,893,974 | (3,764,272 | ) | 76.9 | % | ||||||||||
| (1) | Restricted cash, which is included in cash at the end of the year for a letter of credit, were $0 and $190,140 for the twelve months ended December 31, 2019 and 2018, respectively. |
Net cash used in operating activities for the year ended December 31, 2019 and 2018 was $8,589,598 and $5,643,238, respectively. Net cash used by operating activities decreased 52% during 2019 as compared to 2018 primarily due to a lower volume of commercial orders in 2019 than 2018.
Investing activities for the year ended December 31, 2019 included net capital expenditures for the purchase of property and equipment of $383,070 and $131,257 for intellectual property as compared with $3,209,632 for property and equipment and $175,482 for intellectual property during the same period in 2018. The 2019 levels of capital expenditures are lower than 2018 since we had completed our expanded production capacity in 2018 to meet anticipated customer orders for those customers who are moving into commercialization of products containing our materials.
Financing activities provided a net increase in cash of $5,339,653 and $10,880,736 for the year ended December 31, 2019 and 2018, respectively. For the year ended December 31, 2019 gross proceeds from the issuance of common stock was $2,013,400 and stock issuance expenses were $20,000 as compared to proceeds from the issuance of common stock for the year ended December 31, 2018 of $11,175,344 and stock issuance expenses of $102,880. Financing activities for the year ended December 31, 2019 included $4,000,000 of loan proceeds from the Dow Facility. No new loan proceeds were received from the Dow Facility in 2018 (see Note 7 of our financial statements in this report).
Liquidity and Capital Expenditures
We have historically incurred losses from operations and we may continue to generate negative cash flows as we implement our business plan. Our consolidated financial statements are prepared using US GAAP as applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business.
In December 2016, we entered into the Dow Facility to provide up to $10 million of secured debt financing at an interest rate of 5% per year, drawable at our request under certain conditions. We drew $2 million at closing, $1 million on each of July 18, 2017, September 22, 2017 and December 4, 2017. After December 1, 2017, an additional $5 million became available under the Dow Facility as a result of the Company having raised $10 million of equity capital after October 31, 2016. We drew $2 million on July 8, 2019, an additional $2 million on November 19, 2019 and, on February 12,2020, we drew the remaining $1 million.
As of April 24, 2020, we had cash on hand of $1,589,468.74 Due to lower than expected revenue in 2019 and the current COVID-19 global pandemic impacting both our operations and that of our customers, Management has taken several steps to ensure we are able to fund our operations from existing cash on hand, operating cash flows, additional borrowings and restructured debt obligations.
In March of 2020, we restructured our organization by reducing headcount by 45%, by furloughing substantially all manufacturing employees, and by implementing temporary salary reductions ranging from 15-20% which has resulted in a 58% reduction in annual payroll and related costs. In April 2020, we furloughed additional employees in our R&D and Engineering departments. We also reduced our annual cash run rate for all other expenses by 17%. Salary reductions will be in place until the Company has recorded quarterly revenue of at least $1 million for two consecutive quarters, at which time salaries will return to their previous levels.
34
In late March / early April of 2020, we applied for relief under the Coronavirus Aid, Relief and Economic Security Act (CARES) by submitting an application with the Small Business Administration (SBA) for an Economic Injury Disaster Loan (“EIDL”) and by submitting an application to an SBA lender bank, PNC, for a Paycheck Protection Plan (“PPP”) loan. The potential loan amounts under these programs are up to $2,000,000 and $825,200, respectively. An EIDL, if the Company qualifies, is for up to a 30-year term at a 3.75% annual interest rate. On April 18, 2020, we received an approved and fully executed PPP Term Note for $825,200 with a term of two years, a six-month repayment deferral period, and an annual rate of interest of 1%, with a potential for some or all of the loan to be forgiven, dependent upon use of the loan proceeds. On April 20, 2020, we received the $825,200 under the PPP loan.
On April 23, 2020, we entered into an amended and restated Draw Loan Note and Agreement and related transaction documents (collectively, the “Amended Dow Facility”) with the Dow Chemical Company to amend the terms of our current loan facility to allow us to structure an offering of subordinated, secured convertible notes (“Convertible Notes” and such offering, the “Unit Offering”), to support ongoing cash needs. In the Amended Dow Facility, the Company and Dow agreed to 1) extend the term of such loan facility by two years to December 1, 2023, 2) significantly reduce any required prepayment to Dow from the proceeds of new equity or equity-linked financings from the current 30-50% prepayment requirement on the pre-existing Dow Facility to a 10% prepayment requirement in the Amended Dow Facility, which does not begin until after we have raised an additional $7 million in equity or equity-linked capital from the date of the amendment, 3) capitalize all interest payable until such time as we have recorded GAAP revenue of at least $2 million for two consecutive calendar quarters 4) increase the rate of interest to 6.5% per annum and 5) allow for a subordinated security interest to be granted to new investors in the Unit Offering.
Immediately after the execution of the transaction documents related to the Amended Dow Facility, we began the Unit Offering in a private placement to accredited investors. The Unit Offering is comprised of the Convertible Notes and a fully assignable or transferrable right to exchange two shares of previously issued Common Stock of the Company for two shares of Series B Preferred Stock of the Company for every $8.00 invested in the Unit Offering (the “Exchange Rights”). The Convertible Notes are secured by a junior security interest in all the assets of the Company, bear an interest rate of 7.5% per annum and mature on December 31, 2024. Each investor’s Exchange rights are exercisable for a period of thirty (30) days after the Company has received a completed subscription agreement.
At the option of each holder, the Convertible Notes are convertible into either i) Series B Convertible Preferred Stock (“Series B Preferred Stock”) at a note conversion price of $8.00/share; or ii) any other form of preferred or common stock (“Subsequent Stock”) issued by the Company at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable).
If and when we raise at least $15 million of equity capital (excluding capital raised in this Unit Offering), the Convertible Notes will be automatically converted into whichever of the following equity securities would result in the greatest number of shares of Common Stock being issued to the holders on an “as-if-converted” basis at such time: (i) Series B Preferred Stock at a note conversion price of $8.00/share; or (ii) Subsequent Stock at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable); provided, however, in the event the Company raises at least $15 million of equity capital within one hundred and twenty (120) days after the first issue date of Convertible Notes, such percentages will be changed to 90% of the purchase price per share at which such Subsequent Stock is sold (or, if the value per share is fixed, 110% of the number of shares that might otherwise be issuable).
Each share of Series B Preferred Stock will have an original issue price of $8.00 per share (the “Series B Original Issue Price”) and a liquidation preference of $8.00 per share, with both the Series B Original Issue Price and the liquidation preference per share subject to adjustment for stock splits, recapitalizations, and the like. The Series B Preferred Stock will be senior to the Company’s Common Stock and pari-passu with the Series A Preferred Stock in terms of right of repayment in a liquidation.
The Series B Preferred Stock will have full ratchet antidilution protection that provides that each share of Series B Preferred Stock outstanding may be converted by an Investor at any time into that number of shares of Common Stock determined by dividing the then current Series B Original Issue Price by the applicable Conversion Price (as defined below) with the resulting fraction equal to the “Series B Conversion Rate”. The total number of shares of Common Stock issuable will be equal to the number of shares of Series B Preferred Stock being converted multiplied by the Series B Conversion Rate. The “Conversion Price” is, at any time, the price per share equal to the lesser of a) the Series B Original Issue Price per share and b) the lowest price per share at which the Company has sold equity or equity-linked securities (other than customary exclusions) at any future date while any shares of the Series B Preferred Stock remain outstanding. The Series B Original Issue Price and Conversion Price in effect at any time are also subject to proportional adjustment for share splits, share dividends, recapitalizations and the like.
On April 23, 2020, certain members of our Board of Directors and their affiliates purchased $550,000 of Units. In additions, these Board members and their affiliates have made additional commitments to the Company to match any purchases of Units by disinterested third parties on a dollar for dollar basis up to an additional $1.5 million of Units purchased by such disinterested parties.
Taking into consideration our current cash on hand, we estimate that we will need to raise approximately $500,000- $1,000,000 of additional capital in order to continue our operations for the next twelve months in a minimal to no revenue growth environment. If we are able to raise $1.5 million of capital from disinterested third parties, the commitments from our Board members to match this amount would result in $3.0 million of total capital to the Company, which we estimate would allow the Company to continue operating for 24 months in a minimal to no revenue growth environment.
| ITEM 7A. | QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISKS |
Not applicable.
35
| ITEM 8. | FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA |
XG SCIENCES, INC.
Consolidated Financial Statements
INDEX TO FINANCIAL STATEMENTS
TABLE OF CONTENTS
| Consolidated Financial Statements as of and for the years ended December 31, 2019 and 2018 | |
| Report of Independent Registered Public Accounting Firm | 37 |
| Consolidated Balance Sheets | 38 |
| Consolidated Statements of Operations | 39 |
| Consolidated Statement of Changes in Stockholders’ Equity (Deficit) | 40 |
| Consolidated Statements of Cash Flows | 41 |
| Notes to Consolidated Financial Statements | 42 |
36
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors of
XG Sciences, Inc.
Opinion on the Consolidated Financial Statements
We have audited the accompanying balance sheet of XG Sciences, Inc. (the Company) as of December 31, 2019 and 2018, the related statements of operations, stockholders’ equity (deficit) and cash flows, for the years then ended, and the related notes (collectively, the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2019 and 2018, and the results of its operations and its cash flows for the years then ended in conformity with accounting principles generally accepted in the United States of America.
Emphasis of Matter – Going Concern
The accompanying financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the financial statements, the Company has suffered recurring losses from operations and negative cash flows. The Company’s existing cash on hand, operating cash flows, additional borrowings and restructured debt obligations may not support the operations of the business over the next twelve months. This raises substantial doubt about the Company's ability to continue as a going concern. Management's plans in regard to these matters also are described in Note 1. The financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audit we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
/s/ RSM US LLP
We have served as the Company’s auditor since 2019.
Detroit, Michigan
April 29, 2020
37
XG SCIENCES, INC.
CONDENSED CONSOLIDATED BALANCE SHEETS
| December 31, 2019 | December 31, 2018 | |||||||
| ASSETS | ||||||||
| CURRENT ASSETS | ||||||||
| Cash | $ | 1,129,702 | $ | 4,703,834 | ||||
| Accounts receivable, less allowance for doubtful accounts of $179,600 at December 31, 2019 and $85,000 at December 31, 2018 | 72,227 | 859,054 | ||||||
| Inventories | 891,587 | 660,217 | ||||||
| Other current assets | 334,493 | 114,453 | ||||||
| Total current assets | 2,428,009 | 6,337,558 | ||||||
| LEASEHOLD IMPROVEMENTS AND EQUIPMENT, NET | 3,676,142 | 4,223,650 | ||||||
| RESTRICTED CASH FOR LETTER OF CREDIT | - | 190,140 | ||||||
| LEASE DEPOSIT | 77,544 | 20,156 | ||||||
| INTANGIBLE ASSETS, NET | 753,862 | 690,646 | ||||||
| RIGHT OF USE ASSET | 1,606,443 | - | ||||||
| TOTAL ASSETS | 8,542,000 | 11,462,150 | ||||||
| LIABILITIES AND STOCKHOLDERS’ EQUITY | ||||||||
| CURRENT LIABILITIES | ||||||||
| Accounts payable | 634,564 | 1,102,910 | ||||||
| Other current liabilities | 238,554 | 429,573 | ||||||
| Deferred revenue | - | 832 | ||||||
| Current portion of long-term debt | - | 196,723 | ||||||
| Current portion of lease liabilities | 520,197 | 3,613 | ||||||
| Total current liabilities | 1,393,315 | 1,733,651 | ||||||
| LONG-TERM LIABILITIES | ||||||||
| Long-term portion of lease liabilities | 1,183,872 | 11,914 | ||||||
| Long term debt | 8,111,610 | 4,725,866 | ||||||
| Total long-term liabilities | 9,295,482 | 4,737,780 | ||||||
| TOTAL LIABILITIES | 10,688,797 | 6,471,431 | ||||||
| STOCKHOLDERS’ EQUITY | ||||||||
| Series A convertible preferred stock, 3,000,000 shares authorized, 1,890,354 shares issued and outstanding, liquidation value of $22,684,248 at December 31, 2019 and December 31, 2018 | 22,307,480 | 22,307,480 | ||||||
| Common stock, no par value, 25,000,000 shares authorized, 4,024,443 and 3,760,268 shares issued and outstanding at December 31, 2019 and December 31, 2018, respectively | 32,351,876 | 30,268,476 | ||||||
| Additional paid-in capital | 8,774,975 | 8,101,923 | ||||||
| Accumulated deficit | (65,581,128 | ) | (55,687,160 | ) | ||||
| Total stockholders’ equity | (2,146,797 | ) | 4,990,719 | |||||
| TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY | 8,542,000 | 11,462,150 | ||||||
See notes to consolidated financial statements
38
XG SCIENCES, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
FOR THE YEARS ENDED DECEMBER 31, 2019 AND 2018
| 2019 | 2018 | |||||||
| REVENUE | ||||||||
| Product Sales | $ | 1,369,556 | $ | 4,125,567 | ||||
| Grants | - | 176,253 | ||||||
| Total Revenues | 1,369,556 | 4,301,820 | ||||||
| COST OF GOODS SOLD | ||||||||
| Direct costs | 1,006,594 | 2,344,937 | ||||||
| Unallocated manufacturing expenses | 2,756,879 | 3,161,384 | ||||||
| Cost of Goods Sold | 3,763,473 | 5,506,321 | ||||||
| GROSS LOSS | (2,393,917 | ) | (1,204,501 | ) | ||||
| OPERATING EXPENSES | ||||||||
| Research & Development Expense | 1,506,716 | 1,414,259 | ||||||
| Sales, General & Administrative Expense | 5,467,698 | 4,972,595 | ||||||
| Total Operating Expenses | 6,974,414 | 6,386,854 | ||||||
| OPERATING LOSS | (9,368,331 | ) | (7,591,355 | ) | ||||
| OTHER INCOME (EXPENSE) | ||||||||
| Interest expense, net | 409,318 | 328,261 | ||||||
| Total Other Expense | 409,318 | 328,261 | ||||||
| Net Loss | $ | (9,777,649 | ) | $ | (7,919,616 | ) | ||
| WEIGHTED AVERAGE NUMBER OF SHARES OUTSTANDING - Basic and diluted (in shares) | 3,951,178 | 2,964,805 | ||||||
| NET LOSS PER SHARE - Basic and diluted (in dollars per share) | $ | (2.47 | ) | $ | (2.67 | ) | ||
See notes to consolidated financial statements
39
CONSOLIDATED STATEMENT OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT)
FOR THE YEARS ENDED DECEMBER 31, 2019 AND 2018
| Preferred stock A | Common stock | Additional paid-in capital | Accumulated deficit | |||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | Amount | Amount | Total | ||||||||||||||||||||||
| Balances, December 31, 2017 | 1,857,816 | $ | 21,917,046 | 2,353,350 | $ | 19,116,012 | $ | 7,831,958 | $ | (47,767,544 | ) | $ | 1,097,472 | |||||||||||||||
| Stock issued for cash | 1,396,918 | 11,175,344 | $ | 11,175,344 | ||||||||||||||||||||||||
| Stock issuance fees and expenses | (102,880 | ) | $ | (102,880 | ) | |||||||||||||||||||||||
| Preferred stock issued to pay capital lease obligations | 32,548 | 390,434 | $ | 390,434 | ||||||||||||||||||||||||
| Stock-based compensation expense | 10,000 | 80,000 | 269,965 | $ | 349,965 | |||||||||||||||||||||||
| Net loss | $ | (7,919,616 | ) | $ | (7,919,616 | ) | ||||||||||||||||||||||
| Balances, December 31, 2018 | 1,890,354 | $ | 22,307,480 | 3,760,268 | $ | 30,268,476 | $ | 8,101,923 | $ | (55,687,160 | ) | $ | 4,990,719 | |||||||||||||||
| Stock issued for cash | 251,675 | 2,013,400 | $ | 2,013,400 | ||||||||||||||||||||||||
| Stock issuance fees and expenses | (20,000 | ) | $ | (20,000 | ) | |||||||||||||||||||||||
| Transition adjustment for adoption of ASC 842 | (116,319 | ) | $ | (116,319 | ) | |||||||||||||||||||||||
| Stock-based compensation expense | 12,500 | 90,000 | 369,885 | $ | 459,885 | |||||||||||||||||||||||
| Warrants issued with Dow financing | 303,168 | $ | 303,168 | |||||||||||||||||||||||||
| Net loss | $ | (9,777,649 | ) | $ | (9,777,649 | ) | ||||||||||||||||||||||
| Balances, December 31, 2019 | 1,890,354 | $ | 22,307,480 | 4,024,443 | $ | 32,351,876 | $ | 8,774,976 | $ | (65,581,128 | ) | $ | (2,146,796 | ) | ||||||||||||||
See notes to consolidated financial statements
40
CONSOLIDATED STATEMENTS OF CASH FLOWS
FOR THE YEARS ENDED DECEMBER 31, 2019 AND 2018
| For the Twelve Months Ended | ||||||||
| December 31, | ||||||||
| 2019 | 2018 | |||||||
| CASH FLOWS FROM OPERATING ACTIVITIES | ||||||||
| Net loss | $ | (9,777,649 | ) | $ | (7,919,616 | ) | ||
| Depreciation | 927,974 | 1,696,334 | ||||||
| Amortization of intangible assets | 68,040 | 56,776 | ||||||
| Loss on disposal of equipment and intangibles | 2,605 | 45,130 | ||||||
| Provision for bad debts | 181,679 | 45,000 | ||||||
| Stock-based compensation expense | 459,885 | 349,965 | ||||||
| Non-cash interest expense | 130,410 | 331,526 | ||||||
| Non-cash equipment rent expense | 0 | 106,163 | ||||||
| Changes in current assets and liabilities: | ||||||||
| Accounts receivable | 605,148 | (435,431 | ) | |||||
| Inventory | (231,370 | ) | (488,353 | ) | ||||
| Other current and non-current assets | (296,123 | ) | (98,672 | ) | ||||
| Accounts payable and other liabilities | (660,197 | ) | 667,940 | |||||
| NET CASH USED IN OPERATING ACTIVITIES | (8,589,598 | ) | (5,643,238 | ) | ||||
| CASH FLOWS FROM INVESTING ACTIVITIES | ||||||||
| Purchases of property and equipment | (383,070 | ) | (3,209,632 | ) | ||||
| Purchases of intangible assets | (131,257 | ) | (175,482 | ) | ||||
| NET CASH USED IN INVESTING ACTIVITIES | (514,327 | ) | (3,385,114 | ) | ||||
| CASH FLOWS FROM FINANCING ACTIVITIES | ||||||||
| Repayments of capital lease obligations | (15,527 | ) | (13,238 | ) | ||||
| Repayments of long-term loan debt | (638,220 | ) | (178,490 | ) | ||||
| Proceeds from issuance of common stock | 2,013,400 | 11,175,344 | ||||||
| Common stock issuance fees and expenses | (20,000 | ) | (102,880 | ) | ||||
| Proceeds from long-term loan | 4,000,000 | |||||||
| NET CASH PROVIDED BY FINANCING ACTIVITIES | 5,339,653 | 10,880,736 | ||||||
| NET CHANGE IN CASH, CASH EQUIVALENTS AND RESTRICTED CASH | (3,764,272 | ) | 1,852,384 | |||||
| CASH, CASH EQUIVALENTS AND RESTRICTED CASH, BEGINNING OF PERIOD | 4,893,974 | 3,041,590 | ||||||
| CASH, CASH EQUIVALENTS AND RESTRICTED CASH, END OF PERIOD | 1,129,702 | 4,893,974 | ||||||
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: | ||||||||
| Cash paid for interest | 208,286 | 1,241 | ||||||
| Value of preferred stock issued for AAOF capital lease obligations | 0 | 390,434 | ||||||
| Warrants issued with Dow financing | 303,168 | 0 | ||||||
See notes to consolidated financial statements
41
NOTE 1 — NATURE OF BUSINESS AND BASIS OF PRESENTATION
XG Sciences, Inc., a Michigan company located in Lansing, Michigan and its subsidiary, XG Sciences IP, LLC (collectively referred to as “we”, “us”, “our”, or the “Company”) manufactures graphene nanoplatelets, using two proprietary manufacturing processes to split natural flakes of crystalline graphite into very small and thin particles, which we sell as xGnP® graphene nanoplatelets. We sell our nanoparticles in the form of bulk powders or dispersions to other companies for use as additives to make composite and other materials with specially engineered characteristics. We also manufacture and sell integrated, value-added products containing these graphene nanoplatelets such as greases, composites, thin sheets, inks and coating formulations that we sell to other companies. Additionally, we license our technology to other companies in exchange for royalties and other fees.
Basis of Presentation
The accompanying consolidated financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (“US GAAP”).
The consolidated financial statements include the accounts of XG Sciences, Inc. and our wholly owned subsidiary, XGS IP, LLC. We operate as one operating segment. All intercompany accounts, transactions and profits have been eliminated in consolidation.
Going Concern and Liquidity
We have historically incurred losses from operations and we may continue to generate negative cash flows as we implement our business plan. Our consolidated financial statements are prepared using US GAAP as applicable to a going concern, which contemplates the realization of assets and liquidation of liabilities in the normal course of business.
In December 2016, we entered into the Dow Facility to provide up to $10 million of secured debt financing at an interest rate of 5% per year, drawable at our request under certain conditions pursuant to a draw loan note and agreement (the “Dow Facility”). We drew $2 million at closing, $1 million on each of July 18, 2017, September 22, 2017, and December 4, 2017. After December 1, 2017, an additional $5 million became available under the Dow Facility as a result of the Company having raised $10 million of equity capital after October 31, 2016. With access to these additional funds, we drew $2 million on July 8, 2019, an additional $2 million on November 19, 2019 and, on February 12, 2020, we drew the remaining $1,000,000.
As of April 24, 2020, we had cash on hand of $1,589,468.74. Due to lower than expected revenue in 2019 and the current COVID-19 global pandemic impacting both our operations and that of our customers, Management has taken several steps to ensure we are able to fund our operations from existing cash on hand, operating cash flows, additional borrowings and restructured debt obligations.
In March of 2020, we restructured our organization by reducing headcount by 45%, by furloughing substantially all manufacturing employees, and by implementing temporary salary reductions ranging from 15-20% which has resulted in a 58% reduction in annual payroll and related costs. We also reduced our annual cash run rate for all other expenses by 17%. Salary reductions will be in place until the Company has recorded quarterly revenue of at least $1 million for two consecutive quarters, at which time salaries will return to their previous levels.
In late March / early April of 2020, we applied for relief under the Coronavirus Aid, Relief and Economic Security Act (CARES) by submitting an application with the Small Business Administration (SBA) for an Economic Injury Disaster Loan (“EIDL”) and by submitting an application to an SBA lender bank, PNC, for a Paycheck Protection Plan (“PPP”) loan. The potential loan amounts under these programs are up to $2,000,000 and $825,200, respectively. An EIDL, if the Company qualifies, is for up to a 30-year term at a 3.75% annual interest rate. On April 18, 2020, we received an approved and fully executed PPP Term Note for $825,200 with a term of two years, a six-month repayment deferral period, and an annual rate of interest of 1%, with a potential for some or all of the loan to be forgiven, dependent upon use of the loan proceeds. On April 20, 2020, we received the $825,200 under the PPP loan.
On April 23, 2020, we entered into an amended and restated Draw Loan Note and Agreement and related transaction documents (collectively, the “Amended Dow Facility”) with the Dow Chemical Company to amend the terms of our current loan facility to allow us to structure an offering of subordinated, secured convertible notes (“Convertible Notes”) and such offering, the “Unit Offering”, to support ongoing cash needs. In the Amended Dow Facility, the Company and Dow has agreed to 1) extend the term of such loan facility by two years to December 1, 2023, 2) significantly reduce any required prepayments to Dow from the proceeds of new equity or equity-linked financings from the current 30-50% prepayment requirement on the pre-existing Dow Facility to a 10% prepayment requirement in the Amended Dow Facility, which does not begin until after we have raised an additional $7 million in equity or equity-linked capital from the date of the amendment, 3) capitalize all interest payable until such time as we have recorded GAAP revenue of at least $2 million for two consecutive calendar quarters 4) increase the rate of interest to 6.5% per annum and 5) allow for a subordinated security interest to be granted to new investors in the Unit Offering.
Immediately after the execution of the transaction documents related to the Amended Dow Facility, we began the Unit Offering in a private placement to accredited investors. The Unit Offering is comprised of the Convertible Notes and a fully assignable or transferrable right to exchange two shares of previously issued Common Stock of the Company for two shares of Series B Preferred Stock of the Company for every $8.00 invested in the Unit Offering (the “Exchange Rights”). The Convertible Notes are secured by a junior security interest in all the assets of the Company, bear an interest rate of 7.5% per annum and mature on December 31, 2024. Each investor’s Exchange rights are exercisable for a period of thirty (30) days after the Company has received a completed subscription agreement.
42
At the option of each holder, the Convertible Notes are convertible into either i) Series B Convertible Preferred Stock (“Series B Preferred Stock”) at a note conversion price of $8.00/share; or ii) any other form of preferred or common stock (“Subsequent Stock”) issued by the Company at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable).
If and when we raise at least $15 million of equity capital (excluding capital raised in this Unit Offering), the Convertible Notes will be automatically converted into whichever of the following equity securities would result in the greatest number of shares of Common Stock being issued to the holders on an “as-if-converted” basis at such time: (i) Series B Preferred Stock at a note conversion price of $8.00/share; or (ii) Subsequent Stock at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable); provided, however, in the event the Company raises at least $15 million of equity capital within one hundred and twenty (120) days after the first issue date of Convertible Notes, such percentages will be changed to 90% of the purchase price per share at which such Subsequent Stock is sold (or, if the value per share is fixed, 110% of the number of shares that might otherwise be issuable).
Each share of Series B Preferred Stock will have an original issue price of $8.00 per share (the “Series B Original Issue Price”) and a liquidation preference of $8.00 per share, with both the Series B Original Issue Price and the liquidation preference per share subject to adjustment for stock splits, recapitalizations, and the like. The Series B Preferred Stock will be senior to the Company’s Common Stock and pari-passu with the Series A Preferred Stock in terms of right of repayment in a liquidation.
The Series B Preferred Stock will have full ratchet antidilution protection that provides that each share of Series B Preferred Stock outstanding may be converted by an Investor at any time into that number of shares of Common Stock determined by dividing the then current Series B Original Issue Price by the applicable Conversion Price (as defined below) with the resulting fraction equal to the “Series B Conversion Rate”. The total number of shares of Common Stock issuable will be equal to the number of shares of Series B Preferred Stock being converted multiplied by the Series B Conversion Rate. The “Conversion Price” is, at any time, the price per share equal to the lesser of a) the Series B Original Issue Price per share and b) the lowest price per share at which the Company has sold equity or equity-linked securities (other than customary exclusions) at any future date while any shares of the Series B Preferred Stock remain outstanding. The Series B Original Issue Price and Conversion Price in effect at any time are also subject to proportional adjustment for share splits, share dividends, recapitalizations and the like.
On April 23, 2020, certain members of our Board of Directors and their affiliates purchased $550,000 of Units. In additions, these Board members and their affiliates have made additional commitments to the Company to match any purchases of Units by disinterested third parties on a dollar for dollar basis up to an additional $1.5 million of Units purchased by such disinterested parties.
Taking into consideration our current cash on hand, we estimate that we will need to raise approximately $500,000- $1,000,000 of additional capital in order to continue our operations for the next twelve months in a minimal to no revenue growth environment. If we are able to raise $1.5 million of capital from disinterested third parties, the commitments from our Board members to match this amount would result in $3.0 million of total capital to the Company, which we estimate would allow the Company to continue operating for 24 months in a minimal to no revenue growth environment.
We believe our cash is sufficient to fund our operations through April 2021 when taking into account various sources of funding and cash received from continued commercial sales transactions. However, the ultimate outcome is uncertain, therefore, there is substantial doubt with regard to continuing as a going concern one year beyond the issuance date.
In the event we are unable to fund our operations from existing cash on hand, operating cash flows, or additional borrowings, we may be forced to reduce our expenses, slow down our growth rate, or discontinue operations. Our consolidated financial statements do not include any adjustments relating to the recoverability and classification of recorded asset amounts or the amounts and classification of liabilities that might be necessary should we be unable to continue as a going concern.
43
NOTE 2 — SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Use of Estimates
US GAAP requires management to make estimates, judgments and assumptions that affect the reported amounts of assets, liabilities, revenues and expenses, together with amounts disclosed in the related notes to the consolidated financial statements. Actual results and outcomes may differ from management’s estimates, judgments and assumptions. Significant estimates, judgments and assumptions used in these consolidated financial statements include, but are not limited to, those related to revenues, accounts receivable and related allowances, contingencies, useful lives and recovery of long-term assets, income taxes, and the fair values of stock-based compensation, warrants, and derivative financial instrument liabilities. These estimates, judgments, and assumptions are reviewed periodically and the effects of material revisions in estimates are reflected in the financial statements prospectively from the date of the change in estimate.
Revenue Recognition
On January 1, 2018, the Company adopted ASU No. 2014-09,Revenue from Contracts with Customers (Topic 606), which supersedes nearly all previous revenue recognition guidance.
Revenues are recognized at a point in time, typically when control of the promised goods is transferred to customers, in an amount that reflects the consideration the Company expects to be entitled to in exchange for those goods. The Company does not recognize revenue in cases where collectability is not probable, and defers the recognition until collection is probable or payment is received.
Customer deposits, deferred revenue and other receipts are deferred and recognized when the revenue is realized and earned.
Revenue related to licensing agreements is recorded upon substantial performance of the terms of the licensing contract. In the case of licensing arrangements that involve up-front payments, revenue is recorded when management determines that the appropriate terms of the contract have been fulfilled. For example, this may occur when technology has been transferred via written documents or, if training is involved, whenever all contracted training has occurred. In the case of licenses where product delivery is also embedded in the deliverable, a portion of revenue would be recognized when products are delivered.
We have also licensed certain of our intellectual property to licensees under terms and conditions of license agreements that specify the intellectual property licensed, the territory, and the type of license. In exchange for these licenses, we have recorded revenues associated with the initial granting of the license and expect to receive royalties based on sales of products produced under these licenses. License revenues are recorded to reflect our performance of requirements under these license agreements. In addition, we record royalty revenues from licensees at the time they are earned.
In the case of licensing arrangements that involve ongoing royalties based on sales of products produced with our technology, royalty income is recorded when received or, in the case of minimum royalties due, in the period when due.
Grant contract revenue is recognized over the life of the contracts as the services are performed.
Amounts received in excess of revenues earned are recorded as deferred revenue.
Cost of Products Sold
We use a standard cost system to estimate the direct costs of products sold. Variances between the standard costs and the actual costs are then capitalized to the inventories and expensed to the cost of goods sold. Direct costs include estimates of raw material costs, packaging, freight charges net of those billed to customers, and an allocation for direct labor and manufacturing overhead. Because of the nature of our production processes, there is a substantial fixed manufacturing expense requirement that represents the ongoing cost of maintaining production facilities that are not directly related to products sold, so we use a “full capacity” allocation of overhead based on an estimate of what product costs would be if the manufacturing facilities were operating on a full-time basis and producing products at the designed capacity.
The remaining costs of operating the Company’s manufacturing facilities are recorded as Unallocated Manufacturing Expenses. Manufacturing expense includes the costs of operating our manufacturing facilities including personnel costs, rent, utilities, indirect supplies, depreciation, and related indirect expenses. Manufacturing expenses are expensed as incurred.
44
Research and Development
Research and development expenses include the compensation costs of research personnel, laboratory rent and utilities, depreciation of laboratory equipment, travel and laboratory supplies and are expensed as incurred.
Sales, General and Administrative Expense
Sales, General and Administrative expenses include the compensation costs of personnel, rent, utilities, supplies, travel, depreciation of office equipment, and related expenses not included in other expense categories. Sales and marketing costs include compensation, travel, and business development expenses including free samples provided to customers. These costs are expensed as incurred. Product marketing allowances are recorded at the estimated out of pocket cost necessary to produce the product in the period the allowance is granted. Advertising costs are expensed at the time they are incurred and were not material for the years ended December 31, 2019 and 2018. General and administrative expenses also include non-cash compensation expenses related to the Company’s deferred compensation, management incentive bonus, and employee stock option programs.
Income Taxes
It is our policy to provide for uncertain tax positions and the related interest and penalties based upon management’s assessment of whether a tax benefit is more likely than not to be sustained upon examination by tax authorities. To the extent that the probable tax outcome of these uncertain tax position changes, such changes in estimate will impact the income tax provision in the period in which such determination is made. At December 31, 2019, we believe we have appropriately accounted for any unrecognized tax benefits. We are not aware of any uncertain tax positions. To the extent we prevail in matters for which a liability for an unrecognized tax benefit is established or we are required to pay amounts in excess of the liability, our effective tax rate in a given financial statement period may be affected.
We account for income taxes using an asset and liability approach. The difference between the financial statement and tax basis of assets and liabilities is determined annually. Deferred income tax assets and liabilities are computed for those differences that have future tax consequences using the currently enacted tax laws and rates that apply to the periods in which they are expected to affect taxable income. Income tax expense is the current tax payable or refundable for the period plus or minus the net change in the deferred tax assets and liabilities. The deferred tax effects of state and local income taxes are considered immaterial and have not been recorded. Valuation allowances are established, if necessary, to reduce deferred tax assets to the amount that is estimated to be realized. Because of the uncertainty related to future realization of deferred tax assets (see Note 13), we have established a valuation allowance equal to one hundred percent of the deferred tax assets.
The Tax Cuts and Jobs Act (the Tax Act) was signed into law on December 22, 2017. The law includes significant changes to the U.S. corporate income tax system, including a federal corporate rate reduction from 35% to 21%, limitations on the deductibility of interest expense and executive compensation, and the transition of U.S. international taxation from a worldwide tax system to a territorial tax system. The rate reduction resulted in a $5,635,000 decrease in both the deferred tax asset and valuation allowance with no net impact on the 2018 or 2019 financial statements.
The Company will continue to assess its provision for income taxes as future guidance is issued but does not currently anticipate significant revisions will be necessary. Any such revisions will be treated in accordance with the measurement period guidance outlined in Staff Accounting Bulletin No. 118.
Net Income (Loss) Per Common Share
We compute net income or (loss) per share in accordance with Financial Accounting Standards Board (“FASB”) Accountings Standards Codification (“ASC”) Topic 260: Earnings Per Share. Under the provisions of ASC 260, basic net income (loss) per share is computed by dividing the net income (loss) available to common stockholders by the weighted average number of common shares outstanding during the period. Diluted net loss per share is computed using the weighted average number of shares outstanding during the applicable period, plus the effect of potentially dilutive securities. Potentially dilutive securities consist of shares potentially issuable pursuant to stock options and warrants as well as shares that would result from full conversion of all outstanding convertible securities. These potentially dilutive securities were 3,150,487 and 3,006,487 as of December 31, 2019 and 2018 and are excluded from diluted net loss per share calculations because they are anti-dilutive. As a result, for the years ended December 31, 2019 and 2018, basic and diluted net loss per share was the same.
45
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable are stated at the amount management expects to collect from outstanding balances. Management provides for probable uncollectible amounts through a provision for bad debt expense and an adjustment to a valuation allowance based on their assessment of the current status of individual accounts. Balances that are still outstanding after management has used reasonable collection efforts are written off through a charge to the allowance account and a credit to accounts receivable.
Statements of Cash Flows
For the purposes of the statements of cash flows, we consider all highly liquid investments purchased with an original maturity of three months or less to be cash equivalents.
Fair Value of Financial Instruments and Concentrations of Credit Risk
Financial instruments, which potentially subject the Company to concentrations of credit risk, consist principally of cash and accounts receivable. The Company places its cash with FDIC insured financial institutions. Although such balances may exceed the federally insured limits at certain times, in the opinion of management they are subject to minimal risk.
The Company has established policies for extending credit to customers based upon factors including the customers’ credit worthiness, historical trends and other information. Nonetheless the collectability of accounts receivable is affected by regional economic conditions and other factors.
Inventory
Inventory consists of raw materials, consumables inventory and finished goods, all of which are stated at the lower of cost or net realizable value. Cost is determined on a first in, first out basis. We inventory media used in the production process of our facilities as a Consumables inventory.
Property and Equipment
Property and equipment are recorded at cost, net of accumulated depreciation and amortization. Property and equipment generally include purchases of items with a cost greater than $3,000 and a useful life greater than one year. Depreciation and amortization are computed on the straight-line basis over the estimated useful lives of the assets. Leasehold improvements are amortized over the shorter of the related lease terms or their estimated useful lives.
We periodically review the estimated useful lives of property and equipment. Changes to the estimated useful lives are recorded prospectively from the date of the change. Upon retirement or sale, the cost of the assets disposed of and the related accumulated depreciation are removed from the accounts and any resulting gain or loss is included in the statement of operations. Repairs and maintenance costs are expensed as incurred.
Intangible Assets
We have entered into a license agreement with Michigan State University under which we have licensed certain intellectual property in the form of patents and patent applications and invention disclosures. We are responsible for managing the patent process and ongoing filings for this licensed intellectual property and for bearing the cost thereof. We capitalize all costs related to the acquisition and ongoing administration of this license agreement and we amortize these costs over 15 years or the remaining life of the license agreement, whichever is shorter.
In addition to the costs of managing in-licensed intellectual property, we also file for patent protection for inventions and other intellectual property generated by our employees. All patents are evaluated for filing in international markets on a case-by-case basis and are filed in the United States and in selected international markets as considered appropriate. All external legal and filing costs related to patent applications, patent filings, ongoing registrations, overseas filings, and legal opinions related thereto are capitalized as intangible assets at cost and amortized over a period of 15 years from the date incurred, or the remaining useful life of the associated patent, whichever is shorter.
The cost of royalties or minimum payments specified under the license agreement for in-licensed technology is expensed as incurred.
Recoverability and Impairment of Long-Lived Assets
Land, buildings and equipment and certain other assets, including definite-lived intangible assets, are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. The judgments we make related to the expected useful lives of long-lived assets, definitions of lease terms and our ability to realize undiscounted cash flows in excess of the carrying amounts of these assets are affected by factors such as the ongoing maintenance and improvements of the assets, changes in economic conditions, changes in usage or operating performance, and other factors, such as our ability to sell our assets held for sale. As we assess the ongoing expected cash flows and carrying amounts of our long-lived assets, significant adverse changes in these factors could cause us to realize an impairment loss. Based on a review of operating results, we believe the carrying values of our long-lived assets are recoverable at December 31, 2019 and 2018.
46
Per ASC 360-10-35-47, a long-lived asset to be abandoned is disposed of when it ceases to be used. In the year ended December 31, 2018, management decided not to pursue the xGnP Leaf line business. The asset line had not produced a saleable product for over a year. The Leaf inventory value was written off at the end of 2017. Any sales recorded of Leaf products has been nominal and is from the existing inventory with no plans to restart production. It was determined that the seven (7) year estimated useful life determined in late 2013 should be revised to reflect the use of the asset over a shortened useful life ending December 31, 2018. Depreciation estimates were revised in 2018 in accordance with the change in accounting estimate standards at ASC 250-10-45-17 through 45-20. The amount of acceleration of depreciation reported in operations in 2018 was $681,975. No prior period adjustments or restatements were required.
Stock-Based Compensation
We recognize compensation expense in our statement of operations for all share-based option and stock awards, based on estimated grant-date fair values.
We estimate the grant-date fair value of stock-based compensation awards using the Black-Scholes option valuation model. This model is affected by the estimated value of our common stock on the date of the grant as well as assumptions regarding a number of highly complex and subjective variables. These variables include the expected term of the option, the exercise price, expected risk-free rates of return, the expected volatility of our common stock, and expected dividend yield, each of which is more fully described below. The assumptions for the estimated value of our common stock, expected term and expected volatility are the assumptions that most significantly affect the grant date fair value.
Expected Term: Because we have limited experience related to the exercise of employee stock options, we use the simplified method permitted by SEC Staff Accounting Bulletin Topic 14 to estimate the expected term of the options. The expected term of an option is estimated to be equal to the mid-point between the vesting and expiration dates of the option.
Risk-free Interest Rate: We base the risk-free interest rate used on the implied yield at the grant date of U.S. Treasury zero-coupon issues with a term approximately equal to the expected term of the stock-based award being valued.
Expected Stock Price Volatility: Because we are a company with very limited stock sales history (we have an Offering whereby we are selling stock directly to investors without the assistance of an investment bank nor are we registered on any market or public exchange), we use a blended average weekly volatility of certain publicly traded peer companies. We believe that the use of this blended average peer volatility is reflective of market conditions and a reasonable indicator of our expected future volatility.
Dividend Yield: Because we have never paid a dividend and do not expect to begin doing so in the foreseeable future, we have assumed a 0% dividend yield in valuing our stock-based awards.
The grant-date fair value of the award is recognized as expense over the requisite service period using the straight-line method.
47
Fair Value Measurements
FASB ASC 820: “Fair Value Measurements and Disclosures” defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. ASC 820 also establishes a fair value hierarchy which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The standard describes three levels of inputs that may be used to measure fair value:
Level 1: Quoted prices in active markets for identical assets or liabilities.
Level 2: Inputs other than quoted prices included within Level 1 which are either directly or indirectly observable.
Level 3: Inputs to the valuation methodology are unobservable and significant to the fair value measurement.
A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
Derivative Financial Instruments
We do not use derivative instruments to hedge exposures to cash flow, market or foreign currency risk. The terms of convertible preferred stock and convertible notes that we issue are reviewed to determine whether or not they contain embedded derivative instruments that are required by ASC 815: “Derivatives and Hedging” to be accounted for separately from the host contract and recorded at fair value. The Company’s freestanding warrants are classified as equity.
48
Recent Accounting Pronouncements
In December of 2019, the FASB issued Accounting Standards Update (ASU) 2019-12, Accounting for Income Taxes. The new standard simplifies the accounting for income taxes and is effective for fiscal years beginning after December 15, 2020.
Adoption of Lease Accounting Policy
We applied ASU 2016-02 and all related amendments (“ASC 842”) using the modified retrospective method by recognizing the cumulative effect of adoption as an adjustment to the opening balance of retained earnings at January 1, 2019. Therefore, the comparative information has not been adjusted and continues to be reported under prior leasing guidance. As a result, in the first quarter of 2019 we recorded ROU assets of $1,871,366. We also recorded lease liabilities of $1,981,795. The decrease to retained earnings was $116,319, reflecting the cumulative impact of the accounting change. The standard did not have a material effect on consolidated net income or cash flows.
Right-of-use assets represent our right to use an underlying asset for the lease term and lease liabilities represent our obligation to make lease payments arising from the lease. As our leases do not provide an implicit rate, we used our incremental borrowing rate based on the information available at the commencement date in determining the present value of lease payments. Lease expense for lease payments is recognized on a straight-line basis over the lease term.
We do not record a ROU asset or lease liability for leases with an expected term of 12 months or less. The comparative information has not been adjusted and continues to be reported under prior leasing guidance.
With the exception of the standards discussed above, we believe there have been no new accounting pronouncements effective or not yet effective that have significance, or potential significance, to our Consolidated Financial Statements.
NOTE 3 — INVENTORY
Inventory is carried at the lower of cost or net realizable value. Cost is determined using the first-in, first-out method for raw material and finished goods inventory. Net realizable value is based on current replacement cost for raw materials and net realizable value for finished goods. Raw materials include all components used in the production of our finished goods, which are our four major product lines, bulk materials, energy storage materials, thermal management materials and inks and coatings.
The following amounts were included in inventory at the end of the period:
| Year Ended December 31, | ||||||||
| 2019 | 2018 | |||||||
| Raw Materials | $ | 68,784 | $ | 48,371 | ||||
| Consumables | 70,103 | 188,764 | ||||||
| Finished Goods | 752,700 | 423,082 | ||||||
| Total | $ | 891,587 | $ | 660,217 | ||||
49
NOTE 4 — LEASEHOLD IMPROVEMENTS AND EQUIPMENT
Leasehold improvements and equipment as of December 31 consist of the following:
| Depreciable | |||||||||||
| Life (Years) | 2019 | 2018 | |||||||||
| Equipment not yet placed in service | - | 1,194,417 | |||||||||
Leasehold improvements and equipment | 5-10 | 804,460 | 702,362 | ||||||||
| Lab equipment | 3-7 | 1,134,129 | 1,107,603 | ||||||||
| Production and other equipment | 3-7 | 8,857,169 | 7,487,707 | ||||||||
| Software | 3 | 14,177 | 14,177 | ||||||||
| Total Cost | 10,809,935 | 10,506,266 | |||||||||
| Less accumulated depreciation and amortization | (7,133,793 | ) | (6,282,616 | ) | |||||||
| Net leasehold improvements and equipment | 3,676,142 | 4,223,650 | |||||||||
Depreciation expense on property and equipment, including leased assets, for the years ended December 31, 2019 and 2018, was $927,974 and $1,696,334, respectively. These amounts are included as part of our statement of operations in Cost of Goods Sold, Research and Development, and Sales, General and Administrative Expenses. For the year ended December 31, 2019, $805,114 was recorded in Cost of Goods Sold, $96,065 in Research and Development, and $26,795 in Sales, General and Administrative Expenses. For the year ended December 31, 2018, $1,607,313 was recorded in Cost of Goods Sold, $85,289 in Research and Development, and $3,732 in Sales, General and Administrative Expenses.
NOTE 5 — INTANGIBLE ASSETS
Intangible assets and related accumulated amortization as of December 31, 2019 and 2018 are as follows:
| Year Ended December 31 | ||||||||
| 2019 | 2018 | |||||||
| Licenses | $ | 159,868 | $ | 159,868 | ||||
| Patents | 888,066 | 775,838 | ||||||
| Trademarks, other intangibles | 24,728 | 5,698 | ||||||
| Total Carrying Amount | 1,072,662 | 941,404 | ||||||
| Less: Accumulated Amortization – Licenses | (110,312 | ) | (99,206 | ) | ||||
| Less: Accumulated Amortization – Patents | (202,328 | ) | (148,500 | ) | ||||
| Less: Accumulated Amortization – Trademarks, other intangibles | (6,159 | ) | (3,053 | ) | ||||
| Total Accumulated Amortization | (318,799 | ) | (250,759 | ) | ||||
| Net Carrying Amount – Licenses | 49,556 | 60,662 | ||||||
| Net Carrying Amount – Patents | 685,738 | 627,338 | ||||||
| Net Carrying Amount – Trademarks, other intangibles | 18,569 | 2,645 | ||||||
| Total Net Carrying Amount | $ | 753,863 | $ | 690,645 | ||||
Amortization expense of $68,040 and $56,776 was recorded for the years ended December 31, 2019 and 2018, respectively. Amortization expense for the next five years is estimated to be approximately $68,000 annually.
50
NOTE 6 — OTHER CURRENT LIABILITIES
As of December 31, 2019, and 2018, our other current liabilities on our balance sheet consisted of the following:
| 2019 | 2018 | $ | % | |||||||||||||
| Accrued Compensation | 47,453 | 331,589 | (284,136 | ) | -85.7 | % | ||||||||||
| Accrued Expenses | 188,360 | 59,818 | 128,542 | 214.9 | % | |||||||||||
| 401 (k) Employer Contribution Expense | 2,741 | 38,166 | (35,425 | ) | 92.8 | % | ||||||||||
| Total Other Current Liabilities | 238,554 | 429,573 | (191,019 | ) | 44.5 | % | ||||||||||
NOTE 7 —WARRANTS AND FINANCING AGREEMENTS
Dow Facility
In December 2016, we entered into the Dow Facility which provides us with up to $10 million of secured debt financing at an interest rate of 5% per year, drawable at our request under certain conditions. We received $2 million at closing and an additional $1 million on July 18, 2017, September 22, 2017 and December 4, 2017, respectively. After December 1, 2017, when we had raised $10 million of equity capital post October 31, 2016, an additional $5 million of the $10 million of secured debt financing became available. In 2019, we drew $2 million on July 8, 2019 and an additional $2 million on November 19, 2019. Subsequent to the 2019 year-end, on February 12, 2020, we requested and subsequently received the remaining $1 million draw from the Dow Facility and have issued warrants to purchase up to 25,000 more of our shares. See Note 1.
The Dow Facility is senior to most of our other debt and is secured by all of our assets (Dow was subordinate only to the capital leases with AAOF which was paid off and closed December 31, 2018, see Note 12). The loan matures on December 1, 2021 (subject to certain mandatory prepayments based on our equity financing activities). When we raise a cumulative amount of equity capital exceeding $15 million, we are required to prepay an amount equal to 30% of the amount raised over $15 million, but less than $25 million. We began these prepayments on equity raised as of September 10, 2018. Interest is payable beginning January 1, 2017 although we elected, per the loan documents, to capitalize the interest as part of the outstanding debt through January 1, 2019. Beginning April 1, 2019, current interest was payable in cash on the first day of each quarter.
Dow received warrant coverage of one share of common stock for each $40 in loans received by us, equating to 20% warrant coverage, with an exercise price of $8.00 per share for the warrants issued at closing of the initial $2 million draw. After the initial closing, the strike price of future warrants issued is subject to adjustment if we sell shares of common stock at a lower price. As of December 31, 2019, we had issued 225,000 warrants to Dow, which are exercisable on or before the expiration date of December 1, 2023.
The aforementioned warrants meet the criteria for classification within stockholders’ equity. Proceeds were allocated between the debt and the warrants at their relative fair value. The total debt discount on the Dow Facility was approximately $676,000. The debt discount is being amortized to interest expense using the effective interest method over the term of the loans using an average effective interest rate of 8.2%. During the year ended December 31, 2019, we recognized $432,769 of amortization expense consisting of $302,360 of interest expense paid and $130,410 of amortization from debt discount accretion related to the Dow Facility Loan. We repaid $638,220 of outstanding principal on the debt, and borrowed an additional $4,000,000, resulting in a carrying value of $8,111,611 for the Dow Facility as of December 31, 2019.
The Dow Facility entitles Dow to appoint an observer to our Board. Dow will maintain this observation right until the amount of principal and interest outstanding under the Dow Facility is less than $5 million.
2014 Samsung Financing and Warrants
On January 15, 2014, we sold $3,000,000 of Secured Convertible Notes to SVIC No. 15 New Technology Business Investment L.L.P, a subsidiary of the Samsung Group (“Samsung”). The Notes were converted into Series A Convertible Preferred Stock on December 31, 2015. In connection with the sale of the Notes, the Company issued to Samsung a total of 100,000 warrants with a term of 4 years that were exercisable into shares of Series A Convertible Preferred Stock at a price of $12.00. These warrants vested in four annual installments according to the amount of future cash payments made by Samsung for licensing, royalties and product purchases. As of December 31, 2018, no payments had been made by Samsung and none of the warrants associated with the Samsung note had vested. The warrants expired on January 15, 2018.
NOTE 8 — PRIVATE PLACEMENT
In connection with the private placement of previously issued Series B Preferred Stock warrants to purchase 222,262 shares of common stock and preemptive rights warrants to purchase 2,635 shares of common stock remain outstanding at December 31, 2019. Such warrants have an exercise price of $16.00 per share and expire between April 21 and June 30, 2022.
51
NOTE 9 – STOCK WARRANTS ACCOUNTED FOR AS EQUITY INSTRUMENTS
The following table summarizes the warrants (including the warrants previously accounted for as derivatives) outstanding at December 31, 2019, which are accounted for as equity instruments, all of which are exercisable:
| Exercise | Number of | |||||||||||
| Date Issued | Expiration Date | Indexed Stock | Price | Warrants | ||||||||
| 10/8/2012 | 10/8/2027 | Common | $ | 12.00 | 5,000 | |||||||
| 1/15/2014 - 12/31/2014 | 1/15/2024 | Series A Convertible Preferred | $ | 6.40 | 972,720 | |||||||
| 4/30/2015 - 5/26/2015 | 4/30/2022 | Common | $ | 16.00 | 218,334 | |||||||
| 6/30/2015 | 6/30/2022 | Common | $ | 16.00 | 6,563 | |||||||
| 12/31/2015 | 12/31/2020 | Common | $ | 8.00 | 20,625 | |||||||
| 3/31/2016 | 3/31/2021 | Common | $ | 10.00 | 10,600 | |||||||
| 4/30/2016 | 4/30/2021 | Common | $ | 10.00 | 895 | |||||||
| 12/14/2016 | 12/1/2023 | Common | $ | 8.00 | 50,000 | |||||||
| 7/18/2017 | 12/1/2023 | Common | $ | 8.00 | 25,000 | |||||||
| 9/22/2017 | 12/1/2023 | Common | $ | 8.00 | 25,000 | |||||||
| 12/14/2017 | 12/1/2023 | Common | $ | 8.00 | 25,000 | |||||||
| 7/8/2019 | 12/1/2023 | Common | $ | 8.00 | 50,000 | |||||||
| 11/11/2019 | 12/1/2023 | Common | $ | 8.00 | 50,000 | |||||||
| Total | 1,459,737 | |||||||||||
The warrants indexed to Series A Convertible Preferred Stock are currently exercisable and are exchangeable into 1.875 shares of common stock for each share of Series A Preferred.
NOTE 10 — STOCKHOLDERS’ EQUITY (DEFICIT)
Series A Convertible Preferred Stock
As of December 31, 2019, the Company is authorized to issue up to 3,000,000 shares of Series A Convertible Preferred Stock, or Series A Preferred. Each share of the Series A Preferred, which has a liquidation preference of $12.00 per share, is convertible at any time, at the option of the holder, into one share of Common Stock at the lower of: (a) $12.00 per share, or (b) 80% of the price at which the Company sells any equity or equity-linked securities in the future. The Series A Preferred also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. The Series A Preferred is subject to mandatory conversion into Common Stock upon the listing of the Company’s Common Stock on a Qualified National Exchange. However, the Series A Preferred is not subject to the mandatory conversion until all outstanding Convertible Securities are also converted into common stock. The Series A Preferred ranks senior to all other equity or equity equivalent securities of the Company other than those securities which are explicitly senior or pari passu in rights and liquidation preference to the Series A Preferred.
52
The Company issued 1,456,126 shares of Series A Preferred in connection with the conversion of certain convertible notes on December 31, 2015. There were 1,890,354 shares of Series A Convertible Preferred Stock outstanding as of both December 31, 2018 and 2019.
Series B Convertible Preferred Stock
As of December 31, 2019, and 2018, 1,500,000 shares have been designated as Series B Convertible Preferred Stock, or Series B Preferred, of which none were issued and outstanding as of December 31, 2019 and 2018. Each share of the Series B Preferred, which has a liquidation preference of $16.00 per share, is convertible at any time, at the option of the holder, into one share of common stock at $16.00 per share. The Series B Preferred also contains typical anti-dilution provisions that provide for adjustment of the conversion price to reflect stock splits, stock dividends, or similar events. Each share of Series B Preferred is subject to mandatory conversion into common stock at the then-effective Series B conversion rate upon the public listing by the Company of its common stock on a Qualified National Exchange. However, the Series B Preferred is not subject to the mandatory conversion until all outstanding Convertible Securities are also converted into common stock. The Series B Preferred ranks senior to all other equity or equity equivalent securities of the Company other than those securities which are explicitly senior or pari passu in rights and liquidation preference to the Series B Preferred and pari passu with the Company’s Series A Preferred.
The Series A Preferred and Series B Preferred are not redeemable for cash and the Company concluded that they are more akin to equity-type instruments than debt-type instruments. Accordingly, the embedded conversion option in each agreement is clearly and closely related to an equity-type host and the conversion option does not require classification and measurement as a derivative financial instrument. Therefore, the securities meet the conditions for stockholders’ equity classification. See Note 18.
Common Stock
The Company is authorized to issue 25,000,000 shares of common stock, no par value per share. During the years ended December 31, 2019 and 2018, the Company issued 264,175 and 1,406,918 shares of common stock, respectively. There were 4,024,443 and 3,760,268 shares of common stock issued and outstanding at December 31, 2019 and 2018, respectively.
Each outstanding share of common stock is entitled to one vote on each matter submitted to a vote, unless provided in our Articles of Incorporation, as amended. Each common stockholder is entitled to receive dividends, if declared. Holders of the common stock have no other preemptive or preferential rights to purchase additional shares of any class of the Company’s capital stock in subsequent stock offerings.
NOTE 11 — EQUITY INCENTIVE PLAN
We previously established the 2007 Stock Option Plan (the “2007 Plan”), which was scheduled to expire on October 30, 2017 and under which we granted key employees and directors options to purchase shares of our common stock at not less than fair market value as of the grant date. On May 4, 2017, the Board approved the 2017 Equity Incentive Plan (the “2017 Plan”) to replace the 2007 Plan, which became effective upon the approval of the stockholders holding a majority of the voting power in the Company on July 18, 2017. The 2017 Plan replaced the 2007 Plan and authorizes us to issue awards (stock options and restricted stock) with respect of a maximum of 1,200,000 shares of our common stock, which equals the number of shares authorized under the 2007 Plan.
53
On September 30, 2019 and September 30, 2018, the Company granted each Board member 2,500 stock options and 2,500 shares of restricted stock for their Board services. The options were granted at a price of $8.00 per share and vest ratably over a four-year period beginning on the one-year anniversary. The options had an aggregate grant date fair value of $38,295 and $29,580 on September 30, 2019 and September 30, 2018, respectively. The restricted stock issued to the Board members has an aggregate fair value of $260,000 and vests ratably in arrears over four quarters on the last day of each fiscal quarter following the grant date. As of December 31, 2019, and 2018, 26,250 and 15,000 of the 32,500 shares of restricted stock issued had vested, resulting in compensation expense of $90,000 and $80,000 for the years ended December 31, 2019 and 2018, respectively.
The Company granted 32,500 employee stock options on November 1, 2017. The options were granted at a price of $8.00 per share and had an aggregate grant date fair value of $88,946. The options vest ratably over a four-year period beginning on the one-year anniversary.
The following table shows the stock option activity during the years ended December 31, 2019 and 2018:
| 2019 | 2018 | |||||||||||||||
| Number Of Options | Weighted Average Exercise Price | Number Of Options | Weighted Average Exercise Price | |||||||||||||
| Options outstanding at beginning of year | 797,875 | $ | 8.00 | 677,125 | $ | 8.00 | ||||||||||
| Changes during the year: | ||||||||||||||||
| Cancelled | (28,250 | ) | 8.00 | (27,500 | ) | 8.00 | ||||||||||
| New options granted- at market price | 70,000 | 8.00 | 148,250 | 8.00 | ||||||||||||
| Expired | — | — | ||||||||||||||
| Options outstanding at end of year | 839,625 | 8.00 | 797,875 | 8.00 | ||||||||||||
| Options exercisable at end of year | 498,393 | 8.00 | 396,525 | 8.00 | ||||||||||||
| Weighted average fair value exercise price of options granted during the year | $ | 8.00 | $ | 8.00 | ||||||||||||
| Weighted average remaining contractual term (in months) | 57.8 | 40.2 | ||||||||||||||
Costs incurred in respect of stock-based compensation for employees and directors, for the years ended December 31, 2019 and 2018 were $459,885 and $349,965, respectively. Unrecognized compensation cost as of December 31, 2019 and 2018 was $854,405 and $362,462, respectively. Unrecognized compensation expense includes options which have not yet vested and is expected to be recognized over the next four years.
As of December 31, 2019, none of the currently exercisable stock options had intrinsic value. The intrinsic value of each option share is the difference between the fair value of our common stock and the exercise price of such option share to the extent it is “in-the-money”. Aggregate intrinsic value represents the value that would have been received by the holders of in-the-money options had they exercised their options on the last trading day of the year and sold the underlying shares at the closing stock price on such day. The intrinsic value calculation is based on the assumed market value of our common stock on December 31, 2019 of $8.00 per share, which is the price per share at which we have been selling shares of common stock to third parties in our Offering. There were no in-the-money options outstanding and exercisable as of December 31, 2019, since the exercise prices of the stock options outstanding and expected to vest were all equal to the fair value of our common stock.
54
The following table presents changes in the number of non-exercisable options during 2019:
| 2019 | ||||
| Total- non-exercisable options outstanding- December 31, 2018 | 401,350 | |||
| Options granted | 70,000 | |||
| Options vested | (101,868 | ) | ||
| Options cancelled/forfeited | (28,250 | ) | ||
| Outstanding non-exercisable options outstanding as of December 31, 2019 | 341,232 | |||
| Weighted average grant date fair value | $ | 8.00 | ||
| Weighted average remaining vested period (in months) | 16 | |||
The fair value of options granted during the year ended December 31, 2019 totaled $215,406. The total fair value of options granted during the year ended December 31, 2018 was $437,817.
The fair value of the options granted during the years ended December 31, 2019 and 2018 was estimated on the grant date using the Black-Scholes option-pricing model with the following assumptions:
| 2019 | 2018 | |||
| Fair value of underlying stock | $8.00 | $8.00 | ||
| Expected option life | 4.75 years | 4.75 years | ||
| Expected stock price volatility | 41.07% – 42.95% | 37.34% – 39.09% | ||
| Risk free interest rate | 1.51%- 2.23% | 2.51%- 2.94% | ||
| Expected dividend yield | 0.00% | 0.00% |
Information with respect to restricted stock awards outstanding was as follows:
| 2019 | 2018 | |||||||
| Outstanding non-vested restricted stock at beginning of year: | 5,000 | — | ||||||
| Granted | 12,500 | 10,000 | ||||||
| Vested | 11,250 | 10,000 | ||||||
| Cancelled/forfeited | — | — | ||||||
| Outstanding non-vested restricted stock | 6,250 | 5,000 | ||||||
| Weighted average grant date fair value | $ | 8.00 | $ | 8.00 | ||||
| Weighted average remaining vested period (in months) | 4.5 | 4.5 | ||||||
55
NOTE 12 – LEASES
Right of Use Asset and Leased Liability:
Estimated Lease Life – Lease term through December 2022
| Right-of-use lease assets- operating as of January 1, 2019 | $ | 1,982,739 | ||
| Less: Accumulated amortization | (376,295 | ) | ||
| Right-of-use lease assets- operating as of December 31, 2019 | $ | 1,606,443 | ||
| Lease liability-operating as of January 1, 2019 | $ | 2,094,958 | ||
| Less: Accumulated Amortization | (390,889 | ) | ||
| Lease liability operating-as of December 31, 2019 | $ | 1,704,069 | ||
| Operating lease expense for the twelve months ended December 31, 2019 | $ | 617,896 | ||
| Actual remaining lease payments, December 31, 2019 | $ | 1,964,373 | ||
| Present value of remaining payments, December 31, 2019 | $ | 1,704,069 |
| Supplemental cash flow information related to leases: | Operating Leases Twelve months ended December 31, 2019 | |||
| Cash paid for amounts included in the measurement of lease liabilities: | ||||
| Operating cash flows from operating leases | $ | 632,490 | ||
| Weighted average remaining lease term- operating leases ( in months) | 32.04 | |||
| Weighted average discount rate- operating leases (annual) | 9.98 | % | ||
| Maturities of leases liabilities were as follows: | ||||
| Year ending December 31, 2020 | 660,387 | |||
| Year ending December 31, 2021 | 660,438 | |||
| Year ending December 31, 2022 | 643,548 | |||
| Total Lease payments | $ | 1,964,373 | ||
| Less imputed interest | (260,304 | ) | ||
| Total | $ | 1,704,069 | ||
56
NOTE 13 — INCOME TAXES
| Net operating loss carry forwards | $ | 13,315,000 | $ | 11,330,000 | ||||
| Miscellaneous temporary differences | 50,000 | 55,000 | ||||||
| Property and equipment | 50,000 | |||||||
| Research and development credits | 760,000 | 635,000 | ||||||
| Deferred tax liabilities: | ||||||||
| Property and equipment | (20,000 | ) | ||||||
| Miscellaneous temporary differences | (5,000 | ) | ||||||
| Net deferred tax asset | $ | 14,105,000 | $ | 12,065,000 | ||||
| Valuation allowance | $ | (14,105,000 | ) | $ | (12,065,000 | ) | ||
| Net deferred tax asset | $ | — | $ | — |
Federal net operating loss carry forwards of $60,400,000 and $50,700,000 exist at December 31, 2019 and 2018, respectively. State net operating loss carry forwards of $9,000,000 and $6,500,000 exist at December 31, 2019 and 2018, respectively.
The primary difference between the net operating loss carry forwards and the accumulated deficit arises from certain stock option, warrants and other debt and equity transactions that are considered permanent differences. These losses were incurred in the years 2006 through 2019 and will expire between 2026 and 2039 and their utilization may be limited if we experience significant ownership changes. The Company has not conducted a full IRC Section 382 analysis to determine if a reduction in net operating loss carry forwards is required due to ownership changes. The analysis has not been undertaken due to the financial burden it would cause and changes, if any, resulting in a reduction to the deferred tax asset and related valuation would have no impact on the netdeferred tax asset or expense recognized. The research and development credits will expire between 2028 and 2038. A rate of 28% has been used to calculate the deferred tax assets and liabilities based on the expected effective tax rates, net of applicable credits, upon reversal of the differences above. A valuation allowance has been established against the entire deferred tax asset at December 31, 2019 and 2018.
NOTE 14 —Customer, Supplier, country and Product Concentrations
Grants and Licensing Revenue Concentration
We received no grants or licensing revenue in 2019. One grantor accounted for 98% of total grant revenue in 2018.
Product Concentration
For 2019, we had a concentration of product revenue from one product that was greater than 10% of total product revenues. Revenue from one of the Company’s graphene nanoplatelets materials, Grade C 500 m²/g, was 62%. For 2018, we had a concentration of product revenue from one product that was greater than 10% of total product revenues. Revenue from one of the Company’s graphene nanoplatelets materials, Grade C 500 m²/g, was 74%. We attempt to minimize the risk associated with product concentrations by continuing to develop new markets and products to add to our portfolio.
Customer Concentration
We had one customer whose purchases accounted for 41% and 72% of total product revenues in the twelve months ended December 31, 2019 and December 31, 2018, respectively. At December 31, 2019, there were two customers who had an accounts receivable balance greater than 10% of our outstanding receivable balance. For 2018 we had one customer whose purchases accounted for 72% of total product revenues. At December 31, 2018, there were three customers who had an accounts receivable balance greater than 10% of our outstanding receivable balance.
57
Country Concentration
We sell our products on a worldwide basis. International revenues in 2019 and 2018, as a percentage of total product revenue, were 49% and 12%, respectively. All of these sales are denominated in U.S. dollars.
For 2019, revenue from three countries, Korea, Switzerland and the UK, other than in the United States accounted for more than 5% of total product revenue. For 2018, no one country other than the United States accounted for more than 5% of total product revenue.
Suppliers
We buy raw materials used in manufacturing from several sources. These materials are available from a large number of sources. A change in suppliers has no material effect on the Company’s operations. We did not have any purchases from one supplier that were more than 10% of total purchases in either 2019 or 2018.
NOTE 15 — RELATED PARTY TRANSACTIONS AND COMMITMENTS
We have a licensing agreement for exclusive use of patents and pending patents with Michigan State University (MSU), a stockholder via the MSU Foundation. During 2019 and 2018, we incurred $50,000 each year for royalties for these licenses. We have also entered into product licensing agreements with certain other stockholders. No royalty expenses have been recognized related to these agreements during 2019 and 2018.
During 2019, affiliates of Mr. Pendall, our director, purchased 3,125 shares of common stock with an investment of $25,000. During 2019, Ms. Jacqueline Lemke, the Chief Financial Officer, purchased 5,000 shares of common stock with an investment value of $40,000.
During the year ended December 31, 2018, we issued 32,538 shares, of Series A Preferred to AAOF as payment under the terms of a Master Leasing Agreement. No shares were issued during the year ended December 31, 2019.
During 2018, our directors Mr. Allemang, Jones and Pendell and certain of their affiliates invested the following amounts into our Offering and purchased the number of shares indicated below.
| Purchase Dates (2018) | Amount Invested | Shares Purchased | ||||||||
| Arnold Allemang and affiliates | April 19 and June 27, 2018 | $ | 1,500,000 | 187,500 | ||||||
| Steven C. Jones and affiliates | June 27 – August 29, 2018 | $ | 1,065,944 | 133,243 | ||||||
| David G. Pendell and affiliates | September 30, 2018 | $ | 20,000 | 2,500 | ||||||
| Total | $ | 2,585,944 | 323,243 | |||||||
In conjunction with a financing with AAOF, we and our stockholders listed therein entered into a Shareholder Agreement on March 18, 2013 that contains a number of specific provisions pertaining to the Board of Directors as well as individual Directors. On February 26, 2016, we amended the Shareholder Agreement, which was effective as of April 14, 2016. We further amended the Shareholder Agreement on May 31, 2018.
58
Among other things, the Shareholder Agreement provides for certain voting and nomination rights to be calculated on the basis of “Full Conversion” stock ownership (under which calculation, all convertible notes, preferred shares, or other convertible equity securities are deemed converted into common stock) as follows:
| ● | So long as AAOF or its affiliates own 10% of more of the aggregate outstanding Shareholder Stock (as defined in the Shareholder Agreement): |
| - | the size of the Board of Directors shall be set at seven individuals. |
| - | one person nominated by AAOF shall be elected to the Board of Directors. |
| - | two members of the Board of Directors, other than those nominated by AAOF, POSCO or Hanwha Chemical, shall qualify as independent Directors. |
| ● | So long as POSCO owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by POSCO shall be elected to the Board of Directors. POSCO does not currently own at least 10% of the aggregate outstanding Shareholder Stock and therefore, there is no POSCO representative on the Board of Directors. |
| ● | So long as Hanwha Chemical owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by Hanwha Chemical shall be elected to the Board of Directors. Hanwha does not currently own at least 10% of the aggregate outstanding Shareholder Stock and therefore, there is no Hanwha representative on the Board of Directors. |
As of December 31, 2016, the ownership percentage of AAOF, as calculated for purposes of director voting, required the shareholders bound by the Shareholder Agreement to vote for a director nominated by AAOF. Mr. Jones is the AAOF representative to the Board pursuant to the terms of the Shareholder Agreement.
The Shareholder Agreement grants preemptive rights to shareholders and holders of convertible notes who are parties to the Shareholder Agreement. Pursuant to the terms therein, such shareholders and noteholders have the right to purchase their pro rata share of all shareholder stock that the Company may, from time to time, propose to sell, issue, or exchange after the date of the Shareholder Agreement, other than certain excluded stock which includes stock granted to employees or as merger consideration. Each shareholder’s pro rata shares shall be equal to the ratio of (i) the aggregate number of shares of the Company’s common stock on a fully diluted basis, owned by the such shareholder at the time of the delivery of a preemptive rights notice to (ii) the aggregate number of shares of Company’s common stock on a fully diluted basis owned by all of the Company’s shareholders at the time of the delivery of a preemptive rights notice.
The Shareholder Agreement may be amended or terminated by agreement (either generally or in a particular instance and either retroactively or prospectively), only with the written consent of (i) a majority of the Board, and (ii) persons holding, in the aggregate, shares of Shareholder Stock representing at least sixty percent (60%) of the voting power of all shares of Shareholder Stock then held by Shareholders and their permitted assignees.
The February 26, 2016 amendment provides that holders of Excluded Stock are not subject to the terms of the Shareholders Agreement. Excluded Stock means shares of common stock that are subject to a registration statement that has been filed with the SEC and has been declared effective, and, for the avoidance of any doubt, includes the 3,000,000 shares being offered under the Registration Statement. This amendment took effect upon the effectiveness of our Registration Statement.
The Amendment to the Shareholder Agreement further clarifies that preemptive rights shall not apply to Excluded Stock (including, without limitation, the 3,000,000 shares being offered under the Registration Statement) and amends the termination date of the Shareholders Agreement. The Shareholder Agreement will continue in effect, unless the Shareholder Agreement is earlier terminated in accordance with its terms until the date on which the Company’s common stock is listed on the NASDAQ Stock Market of the New York Stock Exchange. As a result, the Shareholder Agreement will continue to remain in effect and certain of our larger shareholders will be entitled to continue to exercise their rights under such Shareholder Agreement, but purchasers of shares of common stock under the registration statement filed in connection with our current offering are not required to adopt the Shareholder Agreement.
On April 23, 2020, the Dow Chemical Company amended and restated certain terms of our current loan facility to allow the Company to structure an offering of subordinated, secured convertible notes (“Convertible Notes”) and such offering, the “Unit Offering”, instrument to support ongoing cash needs. In the Amended Dow Facility, Dow has agreed to 1) extend the term of our current loan for two years December 1, 2023, 2) significantly reduce any pre-payment requirements from the proceeds of new equity or equity-linked investments from the current 30-50% repayment requirement on the pre-existing Dow Facility to a 10% repayment requirement in the Amended Dow after we have raised an additional $7 million in capital from the date of the amendment, 3) capitalize all interest payable until such time as we have recorded GAAP revenue of at least $2 million for two consecutive calendar quarters 4) increase the rate of interest to 6.5% per year and 5) allow for a subordinated security interest for a Convertible Note offering.
Immediately after the execution of the transaction documents related to the Amended Dow Facility, we began the Unit Offering in a private placement to accredited investors. The Unit Offering is comprised of the Convertible Notes and a fully assignable or transferrable right to exchange two shares of previously issued Common Stock of the Company for two shares of Series B Preferred Stock of the Company for every $8.00 invested in the Unit Offering (the “Exchange Rights”). The Convertible Notes are secured by a junior security interest in all the assets of the Company, bear an interest rate of 7.5% per annum and mature on December 31, 2024. Each investor’s Exchange rights are exercisable for a period of thirty (30) days after the Company has received a completed subscription agreement.
59
At the option of each holder, the Convertible Notes are convertible into either i) Series B Convertible Preferred Stock (“Series B Preferred Stock”) at a note conversion price of $8.00/share; or ii) any other form of preferred or common stock (“Subsequent Stock”) issued by the Company at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable).
If and when we raise at least $15 million of equity capital (excluding capital raised in this Unit Offering), the Convertible Notes will be mandatorily converted into whichever of the following equity securities would result in the greatest number of shares of Common Stock being issued to the holders on an “as-if-converted” basis at such time: (i) Series B Preferred Stock at a note conversion price of $8.00/share; or (ii) Subsequent Stock at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable); provided, however, in the event the Company raises at least $15 million of equity capital within one hundred and twenty (120) days after the first issue date of Convertible Notes, such percentages will be changed to 90% of the purchase price per share at which such Subsequent Stock is sold (or, if the value per share is fixed, 110% of the number of shares that might otherwise be issuable).
Each share of Series B Preferred Stock will have an original issue price of $8.00 per share (the “Series B Original Issue Price”) and a liquidation preference of $8.00 per share, with both the Series B Original Issue Price and the liquidation preference per share subject to adjustment for stock splits, recapitalizations, and the like. The Series B Preferred Stock will be senior to the Company’s Common Stock and pari-passu with the Series A Preferred Stock in terms of right of repayment in a liquidation.
The Series B Preferred Stock will have full ratchet antidilution protection that provides that each share of Series B Preferred Stock outstanding may be converted by an Investor at any time into that number of shares of Common Stock determined by dividing the then current Series B Original Issue Price by the applicable Conversion Price (as defined below) with the resulting fraction equal to the “Series B Conversion Rate”. The total number of shares of Common Stock issuable will be equal to the number of shares of Series B Preferred Stock being converted multiplied by the Series B Conversion Rate. The “Conversion Price” will be the price per share equal to the lesser of a) the Series B Original Issue Price per share and b) the lowest price per share at which the Company has sold equity or equity-linked securities (other than customary exclusions) at any future date while any shares of the Series B Preferred Stock remain outstanding. The Series B Original Issue Price and Conversion Price in effect at any time are also subject to proportional adjustment for share splits, share dividends, recapitalizations and the like.
On April 23, 2020, certain members of our Board of Directors and their affiliates purchased $550,000 of Units. In additions, these Board members and their affiliates have made additional commitments to the Company to match any purchases of Units by disinterested third parties on a dollar for dollar basis up to an additional $1.5 million of Units purchased by such disinterested parties.
NOTE 16 — RETIREMENT PLAN
We maintain a defined-contribution 401(k) retirement plan covering substantially all employees (as defined by our plan document). Employees may make voluntary contributions to the plan, subject to limitations based on IRS regulations and compensation. The plan allows for an employer match contribution. The employer match expense was $110,743 and $76,009 for the years ended December 31, 2019 and 2018, respectively.
NOTE 17 — LETTER OF CREDIT
In April of 2019, we were relieved of the requirement by one of our lease agreements to maintain a letter of credit of approximately $190,000 in exchange for placing one month rent on deposit with the lessee. Through April of 2019, to support this letter of credit, we were required to maintain an equivalent cash deposit. As of December 31, 2019, there was no outstanding letter of credit nor supporting cash deposit. The cash deposit had been restricted and classified as a non-current asset. As of December 31, 2019, and 2018, the cash deposit for the letter of credit was $0 and $190,140, respectively.
NOTE 18 — SUBSEQUENT EVENTS
On February 12, 2020, we requested and subsequently received a draw from our Dow line of $1,000,000 for which we issued 25,000 warrants for the purchase of common stock.
In March of 2020, we restructured our organization by reducing headcount by 45%, by furloughing substantially all manufacturing employees, and by implementing temporary salary reductions ranging from 15-20% which has resulted in a 58% reduction in annual payroll and related costs. We also reduced our annual cash run rate for all other expenses by 17%. Salary reductions will be in place until the Company has recorded quarterly revenue of at least $1 million for two consecutive quarters, at which time salaries will return to their previous levels.
In late March / early April of 2020, we applied for relief under the Coronavirus Aid, Relief and Economic Security Act (CARES) by submitting an application with the Small Business Administration (SBA) for an Economic Injury Disaster Loan (“EIDL”) and by submitting an application to an SBA lender bank, PNC, for a Paycheck Protection Plan (“PPP”) loan. The potential loan amounts under these programs are up to $2,000,000 and $825,200, respectively. An EIDL, if the Company qualifies, is for up to a 30-year term at a 3.75% annual interest rate. On April 18, 2020, we received an approved and fully executed PPP Term Note for $825,200 with a term of two years, a six-month repayment deferral period, and an annual rate of interest of 1%, with a potential for some or all of the loan to be forgiven, dependent upon use of the loan proceeds. On April 20, 2020, we received the $825,200 under the PPP loan.
60
On April 23, 2020, we entered into an amended and restated Draw Loan Note and Agreement and related transaction documents (collectively, the “Amended Dow Facility”) with the Dow Chemical Company to amend the terms of our current loan facility to allow us to structure an offering of subordinated, secured convertible notes (“Convertible Notes” and such offering, the “Unit Offering”), to support ongoing cash needs. In the Amended Dow Facility, the Company and Dow agreed to 1) extend the term of such loan facility by two years to December 1, 2023, 2) significantly reduce any required prepayment to Dow from the proceeds of new equity or equity-linked financings from the current 30-50% prepayment requirement on the pre-existing Dow Facility to a 10% prepayment requirement in the Amended Dow Facility, which does not begin until after we have raised an additional $7 million in equity or equity-linked capital from the date of the amendment, 3) capitalize all interest payable until such time as we have recorded GAAP revenue of at least $2 million for two consecutive calendar quarters 4) increase the rate of interest to 6.5% per annum and 5) allow for a subordinated security interest to be granted to new investors in the Unit Offering.
Immediately after the execution of the transaction documents related to the Amended Dow Facility, we began the Unit Offering in a private placement to accredited investors. The Unit Offering is comprised of the Convertible Notes and a fully assignable or transferrable right to exchange two shares of previously issued Common Stock of the Company for two shares of Series B Preferred Stock of the Company for every $8.00 invested in the Unit Offering (the “Exchange Rights”). The Convertible Notes are secured by a junior security interest in all the assets of the Company, bear an interest rate of 7.5% per annum and mature on December 31, 2024. Each investor’s Exchange rights are exercisable for a period of thirty (30) days after the Company has received a completed subscription agreement.
At the option of each holder, the Convertible Notes are convertible into either i) Series B Convertible Preferred Stock (“Series B Preferred Stock”) at a note conversion price of $8.00/share; or ii) any other form of preferred or common stock (“Subsequent Stock”) issued by the Company at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable).
If and when we raise at least $15 million of equity capital (excluding capital raised in this Unit Offering), the Convertible Notes will be automatically converted into whichever of the following equity securities would result in the greatest number of shares of Common Stock being issued to the holders on an “as-if-converted” basis at such time: (i) Series B Preferred Stock at a note conversion price of $8.00/share; or (ii) Subsequent Stock at a note conversion price per share equal to 80% of the purchase price per share at which such Subsequent Stock is sold (or if the value per share is fixed, 120% of the number of shares that might otherwise be issuable); provided, however, in the event the Company raises at least $15 million of equity capital within one hundred and twenty (120) days after the first issue date of Convertible Notes, such percentages will be changed to 90% of the purchase price per share at which such Subsequent Stock is sold (or, if the value per share is fixed, 110% of the number of shares that might otherwise be issuable).
Each share of Series B Preferred Stock will have an original issue price of $8.00 per share (the “Series B Original Issue Price”) and a liquidation preference of $8.00 per share, with both the Series B Original Issue Price and the liquidation preference per share subject to adjustment for stock splits, recapitalizations, and the like. The Series B Preferred Stock will be senior to the Company’s Common Stock and pari-passu with the Series A Preferred Stock in terms of right of repayment in a liquidation.
The Series B Preferred Stock will have full ratchet antidilution protection that provides that each share of Series B Preferred Stock outstanding may be converted by an Investor at any time into that number of shares of Common Stock determined by dividing the then current Series B Original Issue Price by the applicable Conversion Price (as defined below) with the resulting fraction equal to the “Series B Conversion Rate”. The total number of shares of Common Stock issuable will be equal to the number of shares of Series B Preferred Stock being converted multiplied by the Series B Conversion Rate. The “Conversion Price” is, at any time, the price per share equal to the lesser of a) the Series B Original Issue Price per share and b) the lowest price per share at which the Company has sold equity or equity-linked securities (other than customary exclusions) at any future date while any shares of the Series B Preferred Stock remain outstanding. The Series B Original Issue Price and Conversion Price in effect at any time are also subject to proportional adjustment for share splits, share dividends, recapitalizations and the like.
On April 23, 2020, certain members of our Board of Directors and their affiliates purchased $550,000 of Units. In additions, these Board members and their affiliates have made additional commitments to the Company to match any purchases of Units by disinterested third parties on a dollar for dollar basis up to an additional $1.5 million of Units purchased by such disinterested parties.
Taking into consideration our current cash on hand, we estimate that we will need to raise approximately $500,000- $1,000,000 of additional capital in order to continue our operations for the next twelve months in a minimal to no revenue growth environment. If we are able to raise $1.5 million of capital from disinterested third parties, the commitments from our Board members to match this amount would result in $3.0 million of total capital to the Company, which we estimate would allow the Company to continue operating for 24 months in a minimal to no revenue growth environment.
61
| ITEM 9. | CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURES |
None.
| ITEM 9A. | CONTROLS AND PROCEDURES |
Evaluation of Disclosure Controls and Procedures
At the conclusion of the period ended December 31, 2019, we carried out an evaluation, under the supervision and with the participation of our management, including our Chief Executive Officer and Chief Financial Officer, of the effectiveness of the design and operation of our disclosure controls and procedures (as such term is defined in Rules 13a-15(e) and 15d-15(e) under the Securities Exchange Act of 1934 (the “Exchange Act”)). Based upon that evaluation, because of the addition of the new CFO in November 2018 and because the increase in the number of accounting team personnel did not occur until early 2019, our Chief Executive Officer and Chief Financial Officer concluded that as of December 31, 2018 , our disclosure controls and procedures were not effective and adequately designed to ensure that the information required to be disclosed by us in the reports we submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the applicable rules and forms and that such information was accumulated and communicated to our Principal Executive Officer and Principal Financial Officer, in a manner that allowed for timely decisions regarding required disclosure.
We do not expect that our disclosure controls and procedures will prevent all errors and all instances of fraud. Disclosure controls and procedures, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the disclosure controls and procedures are met. Further, the design of disclosure controls and procedures must reflect the fact that there are resource constraints, and the benefits must be considered relative to their costs. Because of the inherent limitations in all disclosure controls and procedures, no evaluation of disclosure controls and procedures can provide absolute assurance that we have detected all our control deficiencies and instances of fraud, if any. The design of disclosure controls and procedures also is based partly on certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions.
Management’s Annual Report on Internal Control over Financial Reporting
The Company’s management is responsible for establishing and maintaining adequate internal control over financial reporting as defined in Rule 13a-15(f) of the Securities Exchange Act of 1934. The Company’s internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with United States generally accepted accounting principles. Internal control over financial reporting includes policies and procedures that (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the assets of the Company; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with United States generally accepted accounting principles, and that receipts and expenditures of the Company are being made only in accordance with authorizations of management and directors of the Company; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisitions, use or disposition of the Company’s assets that could have a material effect on the financial statements.
Management assessed the effectiveness of the Company’s internal control over financial reporting as of December 31, 2019. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (“COSO”) in Internal Control-Integrated Framework published in 2013. Based on this assessment, and on those criteria, management concluded that the Company has a material weakness in its internal control over financial reporting. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the Company’s annual or interim financial statements will not be prevented or detected on a timely basis. We consider the lack of proper segregation of duties and reviews to be a material weakness: The Company operates with a small finance and accounting team where there is a lack of segregation of duties and limited review controls in place. In December 2019,s the Company’s Controller departed XG Science, leaving the Chief Financial Officer (CFO) of the Company to perform the responsibilities previously performed by the Controller as well as the duties of the CFO. As a result, there is inherent risk in the CFO both preparing and reviewing documentation on behalf of the Company.
This report does not include an attestation report of the Company’s registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by the Company’s registered public accounting firm pursuant to rules of the SEC.
Changes in Internal Controls over Financial Reporting
62
| ITEM 9B. | OTHER INFORMATION |
None
| ITEM 10. | DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE |
Directors and Executive Officers of the Company
The following table sets forth the name and age of the Company’s executive officers, Directors and significant employees as of the date of this filing. There are no family relationships among any of our executive officers or directors.
| Name | Age | Position(s) | ||
| Philip L. Rose | 57 | Chief Executive Officer, President, Secretary, Treasurer & Director | ||
| Arnold A. Allemang | 77 | Chairman of the Board, Chairman of the Executive Committee, and member of the Audit and Compensation Committees | ||
| Steven C. Jones | 56 | Director, AAOF representative to the Board, Chairman of the Audit, Nominating, and Corporate Governance Committees, member of the Executive and Compensation Committees | ||
| Molly P. Zhang | 58 | Director, member of the Audit Committee, Nominating, and Corporate Governance Committees | ||
| Dave Pendell | 73 | Director | ||
| Robert Blinstrub | 66 | Director | ||
| Jacqueline M. Lemke | 57 | Chief Financial Officer | ||
| Bamidele Ali | 43 | Chief Commercial Officer | ||
| Scott Murray | 64 | Vice President, Operations | ||
| Leroy Magwood | 38 | Chief Technologist of Research & Development |
Biographies of Officers and Directors
Philip L. Rose, Ph.D.
Dr. Rose joined the Company in January 2014 and currently serves as our Chief Executive Officer, President, Secretary, Treasurer and is a Director of the Company. Dr. Rose has extensive international business management experience in the electronic and specialty materials markets. Prior to joining XG Sciences, he spent 4 years as President of SAFC Hitech, a $100 million division of Sigma-Aldrich that makes precursors and performance materials for the LED, energy and display, and semiconductor markets. He also served concurrently for a period of time as CEO of Soulbrain-Sigma Aldrich based in South Korea and as an independent Director for Pixtronix, a company based in the Boston area. Before joining Sigma Aldrich, Dr. Rose spent almost 20 years with Rohm and Haas in various leadership positions in the semiconductor and flat panel display industries that include general management, mergers and acquisitions, business development and marketing. Dr. Rose has 7 years of experience living in Japan and South Korea and has travelled extensively in Asia over the past 15 years. Dr. Rose earned his Ph.D. in Physical Chemistry from Duke University and holds a B.S. in Chemistry from the University of Southern California and a certificate in Business Management from The Wharton School at the University of Pennsylvania. In light of the aforementioned experience qualifications, attributes and skills, we believe Dr. Rose is qualified to serve as a director.
63
Arnold A. Allemang
Mr. Allemang has served as a Director of XG Sciences since March 2010 and Chairman of the Board since January 2016. Mr. Allemang was employed by Dow, headquartered in Midland, Michigan for 43 years. Mr. Allemang joined Dow in 1965 in Freeport, Texas. After five years in the Solvents Development Lab, he transferred to Stade, Germany. In 1972, he returned to the United States for various technical and managerial assignments. Mr. Allemang was named unit manager for Freeport’s Chlorinated Ethanes in 1981, and two years later assumed the same role for Light Hydrocarbons and Acetylene. He transferred to Terneuzen, The Netherlands, in 1984 to become production manager for Light Hydrocarbons I, and in 1986 became responsible for process control and engineering functions. In 1988, he returned to Freeport to manage the site’s hydrocarbons production and moved to Midland in 1989 as Director of Technology Centers. Mr. Allemang was named Manufacturing General Manager for Dow Benelux in 1992, and in early 1993 was named regional vice president, Manufacturing and Administration, Dow Benelux. He was named vice president, Manufacturing Operations Dow Europe in late 1993. In August 1995, Mr. Allemang was named vice president, Operations, which included global manufacturing and engineering activities. He then became executive vice president of Dow in 2000 and was named senior advisor in 2004. In March 2008, he retired as a Dow employee Mr. Allemang was elected to the Dow Board of Directors in July 1996 and served until May 2015. Mr. Allemang received a bachelor’s degree in chemistry from Sam Houston State University in Huntsville, Texas. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Allemang is qualified to serve as a director.
Steven C. Jones
Mr. Jones has served as a Director of XG Sciences since March 2014. Mr. Jones is the Chairman of Aspen Capital Group, LLC, a private equity firm headquartered in Naples, FL, which manages private equity funds. Mr. Jones also serves as the President of Aspen Capital Advisors, LLC, which is Aspen Capital Group’s investment management subsidiary. Aspen believes its highest value is to be a “hands on” partner with the management teams of portfolio investments to help them achieve their growth objectives by bringing capital, strategic partners, customers, additional management and other business advice. Mr. Jones has substantial expertise in developing and financing emerging growth companies. In addition to his involvement with XG Sciences, Mr. Jones serves on the Boards of NeoGenomics, Inc. (NASDAQ: NEO) (“NEO”), and ERP Maestro, Inc, a software as a service company, and served on the board of T3 Communications, Inc., a business communications company, until May of 2018. Mr. Jones has also served as an Executive Vice President and consultant of NEO since November 2009 and previously served as Chief Financial Officer from October 2003 until November 2009. In December 2014, he was appointed to serve as a Director of XG Sciences by the Aspen Advanced Opportunity Fund, LP in connection with their investment in XGS. Prior to his career in structured private equity, among other positions, Mr. Jones was a Vice President in the Telecommunications, Media and Technology Investment Banking Group of Merrill Lynch & Co in New York and was the chief executive officer or chief financial officer of various public and private companies. Mr. Jones has a BS degree in Computer Engineering from the University of Michigan and an MBA from the Wharton School of the University of Pennsylvania. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Jones is qualified to serve as a director.
Molly P. Zhang
Molly P. Zhang (aka Peifang Zhang) has served as a director of the Company, since May 2017. From 2011 until retiring in October 2016, Dr. Zhang served as a Vice President of Orica Ltd., USA in a number of departments, including General Management, Global Manufacturing and Asset Management. Prior to joining Orica, Dr. Zhang spent 22 years with Dow, most recently as Managing Director of the SCG-Dow Group and Country General Manager of Dow Thailand. Prior to her roles with Dow in Thailand, Dr. Zhang served as Global Business Vice President of Dow Technology Licensing and Catalyst, and as Manufacturing Leader of Dow Asia Pacific. She also served as Manufacturing Director, Global Technology Director and as a board member of the SCG-Dow Group. Dr. Zhang currently serves as a member of the Supervisory Board for GEA Group, a publicly traded, global engineering, process technology solutions and specialty equipment company based in Dusseldorf, Germany, serving the food, chemical, and energy industries. She serves on the board of directors of Cooper-Standard Holdings, Inc., a publicly traded, global, tier 1 automotive components supplier based in Novi, MI. She also serves on the board of directors for Newmont Mining, a leading global mining company based in Greenwood Village, CO. Dr. Zhang has a Ph.D. in Chemical Engineering from the Technical University of Clausthal, Germany and a Diploma in Chemistry (MS degree), Technical University of Clausthal, Germany. Dr. Zhang is fluent in English, Chinese and German. In light of the aforementioned experience qualifications, attributes and skills, we believe Ms. Zhang is qualified to serve as a director.
64
Dave Pendell
David Pendell is a principal of the Aspen Advanced Opportunity Fund. From June 2009, Mr. Pendell has served as General Partner and Officer of Advanced Stage Capital LLC, which he owns and operates. Since January 2011 Mr. Pendell has served as General Partner and Officer of ASC Lease Income LLC. From May of 2013 until February 2016, Mr. Pendell served as a Board Observer of the Company until he was appointed to the Board in February 2016. In addition, Mr. Pendell has served and continues to serve on the board of directors of ERP Maestro (since June of 2013) and the board of directors of Strategic Health Services Inc. (since April of 2015) and has worked with Aspen Advanced Opportunity Fund and Veterans Capital Fund. Mr. Pendell was responsible for the successful launch and subsequent profitable sale of five start-up entities in which he was the lead entrepreneur and numerous other investments where he served as a lead investor/mentor/coach. Predominantly, Mr. Pendell led the growth of Pendell Printing, Inc. from a small print firm of less than $1MM in sales in 1971 to over $85MM in 1998 when the sale of the business was completed. Past management roles include Chairman of the Board/President of Pendell Printing Inc.; co-founder of Envision Inc., a graphic solutions enterprise; President and CEO of Baustert Engineering, Inc., a software business; co-founder of Ecoland, a timber/real estate development company; founder of The Earth Generation, an environmental education publisher; and co- Founder of Fuel Oil News, a publication for the home heating industry. Mr. Pendell also actively participates in the Michigan Angel Fund and Tamiami Angel Fund I and II. He received his BS in Psychology from Central Michigan University in 1969. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Pendell is qualified to serve as a director.
Robert Blinstrub
Mr. Blinstrub was appointed to serve as a director on March 19, 2019. He was a founder and President and CEO of AGM Automotive (“AGM”), a global designer, innovator and producer of engineered solutions for light vehicles, including lighting, overhead consoles, electronics and textiles headquartered in Troy, Michigan with 9 locations globally including the United States, Costa Rica, Mexico, Austria, and China, from 2001 to 2017. AGM leveraged a global footprint underpinned by world-class engineering, production, assembly and supply chain management functions. As an original founder of AGM, Mr. Blinstrub oversaw its development into a global automotive supplier with OEM customers representing the largest American, European, and Asian OEM’s. AGM historically doubled its revenue base every 18 months (profitably). During the latter part of his tenure, AGM acquired and successfully integrated two competitors in the North American and European markets. AGM accumulated countless Supplier awards from its customers for World Class Quality, Product Design, Engineering, Innovations, and Service. Prior to AGM, Mr. Blinstrub led multiple startups and operational turnarounds in his career. Mr. Blinstrub studied Finance and Marketing at the Broad College of Business at Michigan State University. In light of the aforementioned experience qualifications, attributes and skills, we believe Mr. Blinstrub is qualified to serve as a director.
Jacqueline M. Lemke
Ms. Lemke joined the Company in November 2018 and currently serves as our Chief Financial Officer. Ms. Lemke has served on the Board of Directors of the American Brain Tumor Association since January of 2017. From 2012 to 2016, she served as President and CEO of Bioanalytical Systems, Inc., a Nasdaq listed contract biopharmaceutical research organization where she successfully refinanced and modernized the company’s capabilities while positioning the business for sustainable value creation. Previously, Ms. Lemke was Vice President of Finance and Global CFO of Remy, Inc., a billion-dollar division of Remy International, from 2007 to 2010, where she built a global finance team and created a financial system to support rapid decision making and clear lines of management accountability. From 2004 to 2005, she served as Vice President of Finance and Global CFO Connected Home Solutions at Motorola, Inc., and, prior to that, was Global Strategic Planning Director of the multi-billion-dollar revenue Invista division at the DuPont Company. Ms. Lemke’s experience includes managing cyclical, global businesses, negotiating and implementing mergers, acquisitions and joint ventures and building an infrastructure to execute a restructured refinancing. She began her career as a tax consultant at Deloitte & Touche and is a Certified Public Accountant (CPA). Ms. Lemke earned her bachelor’s degree in finance and accounting from Drexel University and her master’s degree in management from Northwestern University.
Bamidele Ali
Bamidele Ali joined the Company in March 2017 as the Chief Commercial Officer. Prior to joining XG Sciences Mr. Ali served as Vice President of Business Development for Architected Materials, a hybrid-materials and manufacturing company. In that role, he was responsible for sales, global marketing and branding, joint developments, and product pricing strategies. Before joining Architected Materials, he spent 9 years as the president of Ali Technologies Inc. a nanotechnology focused product development and consulting firm. Mr. Ali also served as Director, Continuous Improvement & Program Management and Director, Additive Manufacturing Business Development, Mergers & Acquisition for DSM, an $8 billion life sciences and materials company. In these roles, he developed the company’s global additive manufacturing strategy as well as established a program management office and continuous improvement organization. Mr. Ali also spent several years leading strategic initiatives and developing medical equipment for General Electric Healthcare. He has extensive experience evaluating and integrating foreign companies and technology portfolios. He is also the founder of “ManufactureThis.Build”, an online algorithmic company specializing in financial modeling of production technologies and their applications. Mr. Ali is a Six Sigma Black Belt. He earned his B.S. in Electrical Engineering from the University of Kentucky and holds a certificate in chemistry from Virginia Tech University. Mr. Ali left the employ of XG Sciences, Inc. on March 13, 2020.
65
Scott Murray
Scott Murray has extensive experience as an operation executive and is skilled in business management, product and process development, and strategic leadership of growth of businesses. These experiences ranged from small, privately owned companies to Fortune 500 Companies. Mr. Murray has been the Vice President of Operations of the Company since October 2007. Prior to his tenure with the Company, Mr. Murray was with Motor Wheel Corporation for 17 years having held positions in engineering, marketing, and plant management. He served in the positions of Chief Engineer and Director of Manufacturing prior to the leaving to form a new company. Mr. Murray founded and was CEO of Uretech International Inc. from 1995 through 2004. He directed all process, product, marketing, and commercial activities for that company. As a manufacturer of specialty chemicals and urethane products, Uretech International supplied products to many markets including the automotive, medical, and office furniture industries as well as a variety of industrial applications. Most recently, as Director of Development for McKechnie Automotive, Mr. Murray was responsible for the technical and market introduction of a new product line and the startup of a manufacturing operation for that product line in Kentucky. Mr. Murray is a graduate of Michigan Tech University, where he earned a degree in Metallurgical Engineering. As a member of the Society of Automotive Engineers and the American Iron and Steel Institute served on numerous task forces and technical committees for professional organizations in the automotive industry. He is a past Committee Chairman for the American Society of Non-Destructive Testing. Mr. Murray is a registered Professional Engineer and holds a Six Sigma Black Belt certification.
Leroy Magwood, Ph.D.
Dr. Magwood joined the Company in January 2019 and currently serves as Chief Technology Officer. Prior to joining XG Sciences, Dr. Magwood served as Principal Engineer at Savannah River National Laboratory where he was involved in the development of organic dichromic compounds for use in sensors as well as energetic abatement of noxious gases from nuclear waste tanks. Before joining Savannah River National Laboratory, he spent 5 years in various technical roles at PPG Fiberglass Industries, Surmasis Pharmaceuticals and DSM Functional Materials contributing in such diverse areas as fiber glass sizing, drug delivery, UV Coatings and 3D Printing materials and processes. He earned is Ph.D. in Chemical and Biochemical Engineering from the University of Iowa and holds a B.S. in Physics from Benedict College.
Legal Proceedings
To our knowledge, neither we nor any of our officers or directors is a party to any material legal proceeding or litigation and such persons know of no material legal proceeding or contemplated or threatened litigation. There are no judgments against us or our officers or directors. None of our officers or directors, promoters or control persons has been convicted of a felony or misdemeanor relating to securities or performance in corporate office.
66
Corporate Governance
Audit Committee
Our Board has established an Audit Committee, which is composed of Arnold A. Allemang, and Molly P. Zhang and chaired by Steven C. Jones. Pursuant to our Audit Committee charter, the Audit Committee was established for the primary purpose of assisting the Board in its oversight of the Company’s tax, legal regulatory and ethical compliance. The Audit Committee assists the Board in certain areas, including, but not limited to:
| ● | Oversight and monitoring of the Company’s financial statements, accounting and financial reporting processes, financial statement audits, and other financial information provided by the Company to its shareholders and others; | |
| ● | Overseeing the Company’s compliance with legal, regulatory, and public disclosure requirements; | |
| ● | Oversight of the Company’s registered public accounting firm’s (“independent auditor”) qualifications and independence; | |
| ● | Overseeing the performance of the Company’s independent auditor and the internal audit function; | |
| ● | Overseeing the Company’s systems of disclosure controls and procedures, internal controls over financial reporting, and compliance with ethical standards adopted by the Company; | |
| ● | Oversight of treasury and finance matters; | |
| ● | Oversight and monitoring of enterprise risk management, privacy, and data security; | |
| ● | Oversight of the auditing, accounting, and financial reporting process generally; | |
| ● | Preparation of a report of the Committee to be included in the Company’s annual proxy statement in accordance with any applicable rules of the SEC; and | |
| ● | Review and approval of related-party transactions (as defined by any applicable rules of the SEC and any applicable listing standards of the NASDAQ). |
The members of the Committee are appointed by the Board at its annual meeting from among the Company’s directors. Members are appointed to serve until their successors are duly elected and qualified by the Board, or until their resignation or removal. The Board determines the number of members on the Committee from time to time, but in any event the Committee is to be composed of at least three Board members or any greater minimum number as required by applicable law, the Company’s Bylaws, or the Company’s contractual obligations. The Board may appoint a chairperson and secretary for the Committee. If the Board does not appoint a chairperson or a secretary, the members of the Committee may elect a chairperson or secretary, respectively, by majority vote. Steven Jones currently serves as Chairman of the Audit Committee.
Each member of the Committee is and must be “independent” in accordance with the Company’s contractual obligations and any applicable SEC and NASDAQ rules. The Board determines the standards that are currently applicable to determining whether a member is “independent” and whether each member or nominee member of the Committee satisfies those standards.
The members of the Committee must also satisfy other applicable qualification rules of NASDAQ and the SEC. Generally, each member of the Committee must have a strong level of accounting or financial acumen and must be able to read and understand fundamental financial statements. A member of the Committee may not have participated in the preparation of financial statements of the Company or any current subsidiary of it at any time during the past three years. To the extent required by applicable rules of the SEC, at least one member of the Committee must be a “financial expert” as defined by the applicable rules of the SEC. In general, to be considered a “financial expert,” an audit committee member must have the following attributes:
| ● | An understanding of generally accepted accounting principles and financial statements. |
| ● | The ability to assess the general application of generally accepted accounting principles in connection with the accounting for estimates, accruals, and reserves. |
| ● | Experience preparing, auditing, analyzing, or evaluating financial statements that present a breadth and level of complexity of accounting issues that are generally comparable to the breadth and complexity of issues that can reasonably be expected to be raised by the Company’s financial statements, or experience actively supervising one or more persons engaged in such activities. |
| ● | An understanding of internal controls and procedures for financial reporting. |
| ● | An understanding of audit committee functions. |
Steven Jones and Arnold Allemang qualify as “audit committee financial experts” as the term is defined under the SEC rules.
67
The Audit Committee has established its pre-approval policies and procedures, pursuant to which the Audit Committee approved the foregoing audit services, provided by Frazier & Deeter in 2017 and 2018 and by RSM for the year ended December 31, 2018 (See Item 9). Consistent with the Audit Committee’s responsibility for engaging the Company’s independent auditors, all audit and permitted non-audit services require pre-approval by the Audit Committee. The Audit Committee approves proposed services and fee estimates for these services. The Audit Committee chairperson or his designee has been designated by the Audit Committee to approve any services arising during the year that were not pre-approved by the Audit Committee.
The Audit Committee assists the Board in its general oversight of our financial reporting, internal controls, and audit functions, and is directly responsible for the appointment, compensation and oversight of the work of our independent registered public accounting firm. The Audit Committee reviews and discusses with management and our independent accountants the annual audited and quarterly financial statements (including the disclosures under “Management’s Discussion and Analysis of Financial Condition and Results of Operations”), reviews the integrity of the financial reporting processes, both internal and external, reviews the qualifications, performance and independence of our independent accountants, and prepares the Audit Committee Report included in this Annual Report on Form 10-K in accordance with rules and regulations of the Securities and Exchange Commission. The Audit Committee has the power to investigate any matter brought to its attention within the scope of its duties. It also has the authority to retain counsel and advisors to fulfill its responsibilities and duties.
Nominating and Corporate Governance Committee
Our Board has established and adopted a charter for a Nominating and Corporate Governance Committee (“NGC”). The Committee is composed of Arnold A. Allemang, Steven C. Jones, Molly P. Zhang and Robert Blinstrub and chaired by Steven C. Jones. As provided in its charter, the Nomination and Corporate Governance Committee was established for the primary purposes of considering and reporting to the Board on matters relating to the identification, selection, and qualification of Board members and candidates nominated to the Board as well as assisting the Board with respect to corporate governance matters. The NGC is responsible for providing support to the Board in certain areas, including:
| ● | Assisting the Board by identifying individuals qualified to become Board members. |
| ● | Recommending to the Board the director nominees for the next annual meeting of shareholders. |
| ● | Leading the Board in an annual review of the Board’s performance. |
| ● | Recommending to the Board director nominees for each committee. |
| ● | Developing, maintaining, and overseeing the Company’s corporate governance guidelines. |
| ● | Making recommendations to the Board with respect to corporate governance matters. |
The members of the NGC are appointed by the Board at its annual meeting from among the Company directors and are appointed to serve until their successors are duly elected and qualified by the Board, or until their resignation or removal. The Board determines the number of members on the NGC from time to time, but in any event the NGC must be composed of at least three Board members or any greater minimum number as required by applicable law, the Company’s Bylaws, or the Company’s contractual obligations. The Board may appoint a chairperson and secretary for the NGC. If the Board does not appoint a chairperson or a secretary, the members of the NGC may elect a chairperson or secretary, respectively, by majority vote. Steven Jones currently serves as the Chairperson of the NGC.
Each member of the NGC is and must be “independent” in accordance with the Company’s contractual obligations and any applicable SEC and NASDAQ rules. The Board determines the standards that are currently applicable to determining whether a member is “independent” and whether each member or nominee member of the NGC satisfies those standards.
Compensation Committee
Our Board has established a Compensation Committee, which is composed of Steven C. Jones and Robert Blinstrub and chaired by Arnold A. Allemang. The Compensation Committee was established for the primary purpose of assisting the Board with the review and determination of executive compensation and the oversight, review, and approval of significant employee benefits programs, policies, and plans. The Board has adopted a Compensation Committee charter.
68
The members of the Compensation Committee are appointed by the Board at its annual meeting from among the Company’s directors and are appointed to serve until their successors are duly elected and qualified by the Board, or until their resignation or removal. The Board will determine the number of members on the Compensation Committee from time to time, but in any event the Compensation Committee must be composed of at least three Board members or any greater minimum number as required by applicable law, the Company’s Bylaws, or the Company’s contractual obligations.
Each member of the Compensation Committee must be “independent” in accordance with the Company’s contractual obligations and any applicable SEC and NASDAQ rules. Each member of the Compensation Committee must also qualify as an “outside director” for purposes of 162(m) of the Internal Revenue Code of 1986, as amended. The Board shall determine the standards that are currently applicable to determining whether a member is “independent” and whether each member or nominee member of the Compensation Committee satisfies those standards.
Executive Committee
Our Board has established an Executive Committee, which is composed of Arnold A. Allemang and Steven C. Jones. Pursuant to our Executive Committee charter, the Executive Committee was established for the primary purpose of exercising the powers and duties of the Board between Board Meetings and while the Board is not in session and to implement policy decisions of the Board.
During the intervals between meetings of the Board, the Executive Committee may exercise all of the powers and authority of the Board of Directors for the purpose of acting upon matters that should not be postponed until the next scheduled meeting of the Board. The members of the Executive Committee are to exercise their business judgment to act in what they reasonably believe to be in the best interests of the Corporation and its shareholders. The Executive Committee will have all powers and authority of the Board enumerated in the Bylaws of the Corporation, except to:
| 1. | Take action specifically reserved for another committee of the Board; |
| 2. | Amend the Corporation’s Articles of Incorporation or Bylaws; |
| 3. | Adopt an agreement of merger, conversion, or share exchange; |
| 4. | Recommend to shareholders the sale, lease, or exchange of all or substantially all of the Corporation’s property and assets; |
| 5. | Recommend to shareholders a dissolution of the Corporation or a revocation of a dissolution; |
| 6. | Fill vacancies in the Board; |
| 7. | Declare a distribution or dividend or to authorize the issuance of shares; or |
| 8 | Take action with respect to any other matter that the Board may not delegate to the Committee under the Michigan Business Corporation Act, the Corporation’s Articles of Incorporation, the Corporation’s Bylaws, or the Corporation’s contractual commitments. |
Notwithstanding the foregoing, the Board may, by resolution or an amendment to this Charter, restrict the powers and authority of the Executive Committee, in its sole discretion. In addition, the Executive Committee shall comply with all directions of the Board and shall discharge all duties and responsibilities expressly delegated by the Board to the Executive Committee.
The Executive Committee may delegate any of its responsibilities, along with the authority to take action in relation to such responsibilities, to one or more subcommittees as the Committee may determine to be appropriate. The Board may appoint a chairperson and secretary for the Compensation Committee. If the Board does not appoint a chairperson or a secretary, the members of the Compensation Committee may elect a chairperson or secretary, respectively, by majority vote. Arnold Allemang currently serves as the Chairperson of the Compensation Committee.
69
Conflicts of Interest
Certain potential conflicts of interest are inherent in the relationships between our officers and directors and us.
From time to time, one or more of our affiliates may form or hold an ownership interest in and/or manage other businesses both related and unrelated to the type of business that we own and operate. These persons expect to continue to form or hold ownership interests in and/or manage additional businesses which may compete with our business with respect to operations, including financing and marketing, management time and services and potential customers. These activities may give rise to conflicts between or among the interests of us and other businesses with which our affiliates are associated. Our affiliates are in no way prohibited from undertaking such activities, and neither we nor our stockholders will have any right to require participation in such other activities.
Further, because we have transacted business and intend to continue to do so with some of our officers, directors and affiliates, as well as with firms in which some of our officers, directors or affiliates have a material interest, including, without limitation, Samsung Ventures, ASC XGS, LLC, XGS II, LLC and AAOF, LP, potential conflicts may arise between the respective interests of us and these related persons or entities. We believe that such transactions will be affected on terms at least as favorable to us as those available from unrelated third-parties.
With respect to transactions involving real or apparent conflicts of interest, we have adopted policies and procedures which require that: (i) the fact of the relationship or interest giving rise to the potential conflict be disclosed or known to the directors who authorize or approve the transaction prior to such authorization or approval; and (ii) the transaction be fair and reasonable to us at the time it is authorized or approved by our directors.
Risk Oversight
The Board of Directors is actively involved in the oversight of risks, including strategic, operational and other risks, which could affect our business. The Board of Directors does not have a standing risk management committee, but administers this oversight function directly through the Audit Committee and the Board of Directors as a whole. The Board of Directors considers strategic risks and opportunities and administers its respective risk oversight function by evaluating management’s monitoring, assessment and management of risks, including steps taken to limit our exposure to known risks, through regular interaction with our senior management and in Board and committee deliberations that are closed to members of management. The interaction with management occurs not only at formal Board and committee meetings but also through periodic and other written and oral communications.
Meetings of the Board and Committees
The Board met 4 times in 2019. The Board of Directors also acted at times by unanimous written consent, as authorized by our Bylaws and the Michigan Business Corporation Act. The Audit Committee met 4 times in 2019. The Compensation Committee met 2 times in 2019. The Nominating and Governance Committee did not meet in 2019.
Director Independence
Our Board of Directors has determined that we currently have five independent directors on our Board of Directors: Arnold A. Allemang, Molly P. Zhang, Steven C. Jones, Dave Pendell and Robert Blinstrub. Philip L. Rose is not considered independent. Because our common stock is not currently listed on a national securities exchange, we have used the definition of “independence” of the NASDAQ Stock Market to make this determination.
NASDAQ Listing Rule 5605(a)(2) provides that an “independent director” is a person other than an officer or employee of the Company or any other individual having a relationship that, in the opinion of the Company’s Board, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. The NASDAQ listing rules provide that a director cannot be considered independent if:
| ● | the director is, or at any time during the past three years was, an employee of the Company; |
| ● | the director or a family member of the director accepted any compensation from the Company in excess of $120,000 during any period of 12 consecutive months within the three years preceding the independence determination (subject to certain exclusions, including, among other things, compensation for Board or Board committee service); |
| ● | a family member of the director is, or at any time during the past three years was, an executive officer of the Company; |
70
| ● | the director or a family member of the director is a partner in, controlling stockholder of, or an executive officer of an entity to which the Company made, or from which the Company received, payments in the current or any of the past three fiscal years that exceed 5% of the recipient’s consolidated gross revenue for that year or $200,000, whichever is greater (subject to certain exclusions); |
| ● | the director or a family member of the director is employed as an executive officer of an entity where, at any time during the past three years, any of the executive officers of the Company served on the compensation committee of such other entity; or |
| ● | the director or a family member of the director is a current partner of the Company’s outside auditor, or at any time during the past three years was a partner or employee of the Company’s outside auditor, and who worked on the Company’s audit. |
The following is a summary of certain of the experience, qualifications, attributes and skills that led the Company’s Board of Directors to conclude that such person should serve as a director or officer. This information supplements the biographical information provided above.
Philip L. Rose, Ph.D., Chief Executive Officer, President, Treasurer & Director. Dr. Rose has extensive international business management experience, having previously served as the chief executive officer, president and director of several companies based both in the United States and abroad, and significant experience in the electronic and specialty materials markets. Based on his practical leadership and industry experience, Dr. Rose provides valuable experience and knowledge.
Arnold A. Allemang, Chairman of the Board. Mr. Allemang’s previous board service for several corporations and substantial managerial and operational history at The Dow Chemical Company will prove valuable to the Board as it seeks to implement and maintain growth strategies that will enable the Company to succeed.
Steven C. Jones, Director. Mr. Jones’ background in investment banking and in investing, as well as his prior experience serving as a member of several boards and a senior executive in several companies, enables him to provide the Board with valuable insight and expertise.
Dave Pendell, Director. Mr. Pendell’s experience in successfully building businesses in a range of end-use markets will prove very useful to the Board.
Molly P. Zhang, Director. Ms. Zhang’s experience supporting and advising companies with respect to their growth and business development will enable her to assist the Board and management as they endeavor to expand the Company.
Robert Blinstrub, Director. Mr. Blinstrub’s experience founding, running and selling a successful global supply company of innovative engineered products for light vehicles will enable him to assist the Board and management as they endeavor to expand the Company globally and on matters of supplier relations in the key field of automotives.
Code of Ethics
We have a Code of Ethics applicable to our principal executive, financial and accounting officers, a copy of which is referenced as Exhibit 14 to this report.
71
| ITEM 11. | EXECUTIVE COMPENSATION |
Summary Compensation
The following table sets forth all compensation earned and accrued, in all capacities, during the fiscal years ended December 31, 2019 and 2018 by our named executive officers:
| Option | |||||||||||||||||||||||
| Name and Position | Year | Salary | Bonus | Expense(1) | Other | Total | |||||||||||||||||
| Philip L. Rose, | 2019 | 287,404 | 35,000 | 132,760 | 455,164 | ||||||||||||||||||
| Chief Executive Officer | 2018 | 275,000 | 30,000 | 132,760 | 437,760 | ||||||||||||||||||
| Jacqueline M. Lemke, | 2019 | 235,000 | - | 88,772 | 323,772 | ||||||||||||||||||
| Chief Financial Officer | 2018 | 18,077 | - | - | - | 18,077 | |||||||||||||||||
| Bamidele Ali, | 2019 | 215,000 | 13,000 | 52,329 | 280,329 | ||||||||||||||||||
| Chief Commercial Officer | 2018 | 215,000 | 20,000 | 52,329 | - | 287,329 | |||||||||||||||||
| Scott Murray, | 2019 | 160,000 | 15,000 | - | 175,000 | ||||||||||||||||||
| Vice President of Operations | 2018 | 151,861 | 15,000 | - | 880 | 167,741 | |||||||||||||||||
| Leroy Magwood, | 2019 | 140,192 | - | 4,327 | 144,519 | ||||||||||||||||||
| Chief Technology Officer | |||||||||||||||||||||||
| (1) | Option expense is calculated using the fair value of options that vested during the period. See Note 11 to our financial statements included in this report. |
Outstanding Equity Awards
The Board of Directors of the Company occasionally awards stock options and restricted common stock under the Company’s 2017 Plan. Additionally, stock warrants have been issued to certain officers and directors of the Company in conjunction with financing agreements. The following table sets forth information regarding outstanding stock option and stock warrant awards as of December 31, 2019:
| Name and Position(s) | Number of Securities Underlying Unexercised Options and Warrants that are currently Exercisable | Number of Securities Underlying Unexercised Options and Warrants that are currently Unexercisable | Exercise Price | Expiration Date | ||||||||||
| Philip L. Rose,Chief Executive Officer, Secretary, Treasurer | 250,830 | 79,170 | $ | 8.00 | 7/24/2024 | |||||||||
| Jacqueline M. Lemke,Chief Financial Officer | 30,000 | 90,000 | $ | 8.00 | 12/31/2025 | |||||||||
| Bamidele Ali,Chief Commercial Officer * | 40,000 | 40,000 | $ | 8.00 | 7/24/2024 | |||||||||
| Scott Murray,Vice President of Operations | 40,000 | - | $ | 8.00 | 7/24/2024 | |||||||||
| Leroy Magwood,Chief Technologist of Research & Development | - | 37,500 | $ | 8.00 | 12/31/2026 | |||||||||
| * | Mr. Ali left the employment of XG Sciences Inc. effective March 13, 2020. If unexercised within 90 days of March 13, 2020, the 40,000 warrants listed above as currently exercisable will expire. The 40,000 warrants listed above as currently unexercisable will not vest during this 90-day period and expired as of March 13, 2020. |
72
Director Compensation
Each of our non-employee Directors who was not appointed as a representative of a corporate investor in XG Sciences is entitled to receive compensation in accordance with director compensation plans as amended by the full Board of Directors from time to time. The following table sets forth information concerning the compensation of eligible Directors for the years ended December 31, 2019 and 2018:
| Name | Period | Cash Compensation | Restricted Stock | Option Expense | Total | ||||||||||||||
| Arnold Allemang | 2019 | $ | 12,000 | $ | 20,000 | $ | 11,553 | $ | 43,553 | ||||||||||
| 2018 | $ | 12,000 | $ | 20,000 | $ | 9,689 | $ | 41,689 | |||||||||||
| Steven Jones | 2019 | $ | 60,000 | $ | 20,000 | $ | 10,733 | $ | 90,733 | ||||||||||
| 2018 | $ | 56,000 | $ | 20,000 | $ | 8,869 | $ | 84,869 | |||||||||||
| David Pendell | 2019 | $ | 12,000 | $ | 20,000 | $ | 13,801 | $ | 45,801 | ||||||||||
| 2018 | $ | 12,000 | $ | 20,000 | $ | 11,937 | $ | 43,937 | |||||||||||
| Molly P. Zhang | 2019 | $ | 12,000 | $ | 20,000 | $ | 3,961 | $ | 35,961 | ||||||||||
| 2018 | $ | 12,000 | $ | 20,000 | $ | 2,097 | $ | 34,097 | |||||||||||
| Robert Blinstrub | 2019 | $ | 10,000 | $ | 10,000 | $ | 482 | $ | 20,482 | ||||||||||
| (1) | We granted stock options and restricted stock to each of our Board members as part of their compensation package. On September 30, 2019, September 30, 2018, and August 10, 2017, each of the independent Board members received 2,500 stock options and 2,500 shares of restricted stock for their Board services. The options were granted at a price of $8.00 per share and vest ratably over a four-year period beginning on the one-year anniversary. The stock options had an aggregate grant date fair value of $38,295, $29,580 and $26,120 on September 30, 2019, September 30, 2018, and August 10, 2017, respectively. Option expense is calculated using the fair value of options that vested during the period. See Note 11 to the December 31, 2019 financial statements included in this report. The restricted stock issued to the Board members has an aggregate fair value of $260,000 and vest ratably in arrears over four quarters on the last day of each fiscal quarter following the grant date. As of December 31, 2019, 2018, and 2017, 26,250, 15,000 and 5,000 of the 32,500 shares of restricted stock issued had vested, resulting in compensation expense of $90,000, $80,000 and $40,000 in for the years ended December 31, 2019, 2018, and 2017 respectively. |
73
2017 Stock Option Plan
The previously established 2007 Plan, which was scheduled to expire on October 30, 2017 and under which we granted key employees and directors options to purchase shares of our common stock at not less than fair market value as of the grant date. On May 4, 2017, the Board approved the 2017 Plan to replace the 2007 Plan, which became effective upon the approval of the stockholders holding a majority of the voting power in the Company on July 18, 2017. The 2017 Plan replaces the 2007 Plan and authorizes us to issue awards (stock options and restricted stock) with respect of a maximum of 1,200,000 shares of our common stock, which equals the number of shares authorized under the 2007 Plan.
The fair value of the options granted was estimated on the date of grant using the Black Scholes option-pricing model. As of December 31, 2019, and 2018, respectively, 839,625 and 797,875 options were outstanding. Vesting of the option shares with the employees range from immediately to 25% per year. Rights to exercise the options vest immediately upon a change in control of the Company or termination of the employee’s continuous service due to death or disability. The options expire at various dates through 2026.
Employment Agreements and Potential Payments Upon Termination
The Company is party to four employment agreements. The following descriptions summarize the commitments in these arrangements.
On December 16, 2013, the Company entered into an employment agreement with Philip L. Rose to serve as the Company’s Chief Executive Officer commencing on January 6, 2014 and continuing indefinitely, subject to termination by the Company for cause or without cause, or resignation by Dr. Rose with or without cause. If the Company terminates Dr. Rose without cause, or if Dr. Rose resigns with cause, the Company has agreed to pay Dr. Rose’s base salary for a period of six months, any permitted COBRA health insurance premiums for Dr. Rose and his family, and any pro-rata bonus amounts that are deemed to have been earned during Dr. Rose’s employment period prior to termination. In the event that the Company terminates Dr. Rose with cause, or if Dr. Rose resigns without cause, the Company has no further obligations beyond the severance date. The Agreement provides that Dr. Rose will be paid an initial base salary of $275,000 annually, with an annual Target Bonus opportunity of 30% of base salary, which may be earned up to a level of 150% of the Target Bonus under certain conditions. Additionally, Dr. Rose was awarded an option to purchase a total of 330,000 shares of common stock at $8.00 per share and with a life of seven years from the award date. These stock options have vested or will vest as follows: 171,658 on July 24, 2017, 39,586 on July 24, 2018, July 24, 2019, July 24, 2020 and 39,584 on July 24, 2021.
On March 22, 2017, the Company entered into an employment agreement with Bamidele Ali to serve as the Company’s Chief Commercial Officer. The employment agreement: (i) established an annual base salary of $215,000, (ii) granted the employee eligibility to participate in the Company’s Management Incentive Plan with a target annual bonus of 30% of the employee’s annual base salary, and (iii) provided for other fringe benefits, including a stock option grant to purchase 80,000 shares of the Company’s common stock at a strike price of $8.00 per share and with a life of seven years from the award date. These stock options vested ratably over a four-year period beginning on the one-year anniversary; 20,000 on July 24, 2018 and 20,000 each of the three years thereafter until 2021. The employment agreement created an “at will” employment relationship. However, upon termination of employment without cause (as defined in the employment agreement), then the Company was required pay as severance for a period of 3 months from the date of notice of termination 100% of the COBRA premiums for the employee’s family health insurance benefits and a pro-rata portion of any annual bonus that would be due for the year. Mr. Ali’s employment agreement terminated on March 13, 2020.
On October 29, 2018, the Company entered into an employment agreement with Jacqueline Lemke to serve as the Company’s Chief Financial Officer. The employment agreement: (i) establishes an annual base salary of $235,000, (ii) grants the employee eligibility to participate in the Company’s Management Incentive Plan with a target annual bonus of 30% of the employee’s annual base salary and (iii) provides for other fringe benefits, including a stock option grant of 120,000 shares of the Company’s common stock at a strike price of $8.00 per share and with a life of seven years from the award date. These stock option vest ratably over a four-year period, beginning on the grant date (December 31, 2018); 30,000 on December 31, 2019 and 30,000 each of the three years after until 2022. The employment agreement creates an “at will” employment relationship. However, if the Company terminates the employee’s employment without cause (as defined in the employment agreement), then the Company must pay as severance for a period of 3 months from the date of notice of termination, base salary, 100% of COBRA premiums and a pro-rata portion of any annual bonus that would be due for the year in which termination occurs.
On June 30, 2019, the Company entered into an employment agreement with Leroy Magwood to serve as the Company’s Chief Technologist. The employment agreement: (i) establishes an annual base salary of $180,000, (ii) grants the employee eligibility to participate in the Company’s Management Incentive Plan with a target annual bonus of 20% of the employee’s annual base salary and (iii) provide for other fringe benefits, including a stock option grant of 10,000 shares of the Company’s common stock at a strike price of $8.00 per share and with a life of seven years from the award date. These stock option vest ratably over a four-year period, beginning on the grant date (June 30, 2019); 2,500 on June 30, 2020 and 2,500 each of the three years after until 2022. The employment agreement creates an “at will” employment relationship. However, if the Company terminates the employee’s employment without cause (as defined in the employment agreement), then the Company must pay as severance for a period of 3 months from the date of notice of termination 100% of COBRA premiums and a pro-rata portion of any annual bonus that would be due for the year in which termination occurs. Effective January 13, 2020, the Company appointed Dr. Magwood as the Company’s Chief Technology Officer and Dr. Magwood was granted an additional 20,000 shares of the Company’s common stock at a strike price of $8.00 per share and with a life of seven years from the award date. These stock option vest ratably over a four-year period, beginning on the grant date (December 31, 2019); 5,000 on December 31, 2020 and 5,000 each of the three years after until 2022.
74
| ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
As of March 31, 2020, (the “Ownership Date”), the following table sets forth certain information with respect to the beneficial ownership of our common stock, Series A Preferred Stock and Series B Preferred Stock by (i) by each of our directors and executive officers, (ii) by all of our director and executive officers as a group, and (iii) by each person or entity known by us to beneficially own more than 5% of any class of our outstanding shares. We have determined the number and percentage of shares beneficially owned by such person in accordance with Rule 13d-3 under the Exchange Act.
Name and Address of Beneficial Owner(1)(2) |
Shares of |
Percentage of |
Shares of | Percentage of | Total | |||||||||||||||
| Directors, Executive Officers, and Significant Employees | ||||||||||||||||||||
| Philip L. Rose(8) | 265,986 | 6.2 | % | — | — | 3.4 | % | |||||||||||||
| Arnold A. Allemang(9) | 460,032 | 11.3 | % | — | — | 6.0 | % | |||||||||||||
| Steven C. Jones(10) | 339,011 | 8.3 | % | — | — | 4.5 | % | |||||||||||||
| David G. Pendell(11) | 55,204 | 1.4 | % | 5,919 | * | * | ||||||||||||||
| Molly P. Zhang(12) | 9,375 | * | — | — | * | |||||||||||||||
| Jacqueline M. Lemke(13) | 35,000 | * | — | — | * | |||||||||||||||
| Bamidele Ali(14) | 40,000 | 1.0 | % | — | — | * | ||||||||||||||
| Scott Murray(15) | 40,100 | 1.0 | % | — | — | * | ||||||||||||||
| Robert Blinstrub(16) | 32,500 | * | — | — | * | |||||||||||||||
| Directors & Executive Officers as a Group (9 persons) | 1,277,208 | 28.4 | % | 5,919 | * | 15.9 | % | |||||||||||||
| Certain Other Beneficial Owners – Over 5% Ownership | ||||||||||||||||||||
| Aspen Advanced Opportunity Fund, LP(18) | 3,638,885 | 47.5 | % | 3,638,885 | 71.3 | % | 39.9 | % | ||||||||||||
| POSCO(19) | 481,250 | 11.2 | % | 281,250 | 7.7 | % | 6.3 | % | ||||||||||||
| ASC-XGS, LLC(20) | 311,293 | 7.2 | % | 311,293 | 8.8 | % | 4.1 | % | ||||||||||||
| XGS II, LLC(21) | 360,946 | 8.2 | % | 360,946 | 9.8 | % | 4.7 | % | ||||||||||||
| SVIC No. 15 New Technology Business Investment LLP (Samsung)(22) | 590,079 | 12.8 | % | 590,079 | 16.6 | % | 7.8 | % | ||||||||||||
| Michael R. Knox(23) | 267,470 | 6.4 | % | 135,423 | 3.8 | % | 3.5 | % | ||||||||||||
| * | Less than 1% |
| (1) | Beneficial ownership is determined in accordance with the rules of the SEC and generally includes voting or investment power with respect to securities. In accordance with SEC rules, shares of stock issuable upon the exercise of options, warrants and other convertible securities which are currently exercisable and convertible or which become exercisable or convertible within sixty (60) days following the date of the information in this table are deemed to be beneficially owned by, and outstanding with respect to, the holder of such option, warrant or other convertible security. Subject to community property laws where applicable, and unless otherwise specified, to our knowledge, each person listed is believed to have sole voting and investment power with respect to all shares owned by such person. |
| (2) | Unless otherwise specified, the address of the beneficial owner shall be the business address of the Company, c/o XG Sciences, Inc., 3101 Grand Oak Drive, Lansing, MI 48911. |
75
| (3) | Number of shares of common stock includes, as of the Ownership Date: (i) shares of common stock registered in the name of the respective stockholder; (ii) shares of common stock which are issuable upon the conversion of Series A Preferred Stock registered in the name of the respective stockholder that are immediately convertible at the current Series A Conversion Rate; (iii) shares of common stock underlying common stock options and/or common stock warrants of the respective stockholder and which are exercisable within sixty (60) days of the Ownership Date; and (iv) shares of common stock which are issuable upon the exercise of warrants to purchase Series A Preferred Stock currently exercisable within sixty (60) days of the Ownership Date that are registered in the name of the respective stockholder and the conversion of such Series A Preferred Stock into common stock at the current Series A Conversion Rate. |
| (4) | Applicable percentage of ownership of common stock for each respective stockholder is based on 4,024,443 shares of common stock issued and outstanding as of the Ownership Date, together with: (i) shares of common stock which are issuable upon the conversion of Series A Preferred Stock registered in the name of the respective stockholder that are immediately convertible at the current Series A Conversion Rate; (ii) shares of common stock underlying stock options and/or common stock warrants registered in the name of the respective stockholder and which are exercisable within sixty (60) days of the Ownership Date; and (iii) shares of common stock underlying warrants to purchase Series A Preferred Stock registered in the name of the respective stockholder which are exercisable and convertible into common stock, at the current Series A Conversion Rate, within sixty (60) days of the Ownership Date. |
| (5) | Number of shares of Series A Preferred Stock includes, as of the Ownership Date: (i) shares of common stock which are issuable upon the conversion of Series A Preferred Stock registered in the name of the respective stockholder that are immediately convertible at the current Series A Conversion Rate; and (ii) shares of common stock which are issuable upon the exercise of warrants to purchase Series A Preferred Stock currently exercisable within sixty (60) days of the Ownership Date that are registered in the name of the respective stockholder and the conversion of such Series A Preferred Stock into common stock at the current Series A Conversion Rate. |
| (6) | Applicable percentage of ownership of Series A Preferred Stock, as of the Ownership Date, is based on 3,544,408 shares of common stock which are issuable upon conversion of all 1,890,354 shares of Series A Preferred Stock that are immediately convertible, together with shares of common stock underlying warrants to purchase Series A Preferred Stock registered in the name of the respective stockholder which are exercisable and convertible into common stock, at the current Series A Conversion Rate, within sixty (60) days of the Ownership Date. |
| (7) | Applicable percentage of ownership of fully converted voting common stock, as of the Ownership Date, is based on: (i) 4,024,443 shares of common stock issued and outstanding, (ii) 3,544,408 shares of common stock which are issuable upon the conversion of 1,890,354 shares of Series A Preferred Stock that are immediately convertible, (iii) shares of common stock underlying options and/or common stock warrants of the respective stockholder which are exercisable within sixty (60) days of the Ownership Date, and (iv) shares of common stock which are issuable upon the exercise of warrants to purchase Series A Preferred Stock currently exercisable within sixty (60) days of the Ownership Date that are registered in the name of the respective stockholder and the conversion of such Series A Preferred Stock into common stock at the current Series A Conversion Rate. |
| (8) | Philip Rose figures include: (i) 12,500 shares of common stock; (ii) 250,830 shares of common stock underlying currently exercisable options; and (iii) 2,656 shares of common stock underlying currently exercisable warrants. |
| (9) | Arnold Allemang figures include: (i) 414,875 shares of common stock; (ii) 24,656 shares of common stock underlying currently exercisable warrants; and (iv) 18,501 shares of common stock underlying currently exercisable options. The shares of common stock and warrants are held in the name of the Arnold Avery Allemang Revocable Trust. |
| (10) | Steven Jones figures include: (i) 7,500 shares of common stock; (ii) 17,312 shares of common stock underlying currently exercisable options; (iii) 82,500 shares of common stock and 9,969 shares of common stock underlying currently exercisable warrants, held in the name Jones Network, LP, of which Mr. Jones is the General Partner; (iv) 6,250 shares of common stock and 2,656 shares of common stock underlying currently exercisable warrants, held in the name Jones Extended Family Trust, of which Mr. Jones is trustee; (v) 13,000 shares of common stock and 3,750 shares of common stock underlying currently exercisable warrants, held in the name of MadSavAsh Investments, LLC, of which Mr. Jones is managing member; (vi) 45,000 shares of common stock and 8,281 shares of common stock underlying currently exercisable warrants, held in the name Steven & Carisa Jones 401K Plan and Trust, of which Mr. Jones is trustee; (vii) 2,793 shares of common stock held jointly with his 3 children: 931 shares held in the name of Savannah W. Jones & Steven C. Jones JTWROS, 931 shares held in the name of Ashleigh C. Jones and Steven C. Jones JTWROS, and 931 shares held in the name of Madison A. Jones & Steven C. Jones JTWROS; (viii) 75,000 shares of common stock held in the name of Steven & Carisa Jones JTWROS; (ix) 50,000 shares of common stock held jointly in a spousal trust; (x) 12,000 shares of common stock in a Roth IRA held in the name of Steven C. Jones Roth IRA; and (xi) 3,000 shares of common stock in a Roth IRA in the name of Carisa A. Jones Roth IRA. Mr. Jones is an affiliate of Aspen Advanced Opportunity Fund, LP (“AAOF”), which owns 1,107,406 shares of Series A Preferred Stock which are currently convertible into 2,076,386 shares of common stock and currently exercisable warrants to purchase 833,333 shares of Series A Preferred Stock which are currently convertible into 1,562,499 shares of common Stock. Mr. Jones disclaims beneficial ownership of the shares and warrants owned by AAOF. |
| (11) | David Pendell figures include: (i) 7,500 shares of common stock; (ii) 12,375 shares of common stock underlying currently exercisable options; (iii) 2,875 shares of common stock underlying currently exercisable warrants; (iv) 5,919 shares of common stock issuable upon the conversion of 3,157 shares of Series A Preferred Stock held jointly with his wife Vicky Pendell; (v) 6,250 shares of common stock and 2,656 shares of common stock underlying currently exercisable warrants, held in the name David Pendell Revocable Trust; (vi) 15,629 shares of common stock held in the name of the Shirley G. Pendell Irrevocable Trust, of which Mr. Pendell is a trustee; and (vii) 2,000 shares of common stock held in the name of Pendell Irrevocable Trust U/A dated 12/9/98, of which Mrs. Vicky Pendell is a trustee. Mr. Pendell is an affiliate of AAOF, which owns 1,107,406 shares of Series A Preferred Stock which are currently convertible into 2,076,386 shares of common stock and currently exercisable warrants to purchase 833,333 shares of Series A Preferred Stock which are currently convertible into 1,562,499 shares of common Stock. Mr. Pendell is also an affiliate of XGS II, LLC, which owns 109,172 shares of Series A Preferred Stock which are currently convertible into 204,697 shares of common stock and currently exercisable warrants to purchase 83,333 shares of Series A Preferred Stock which are currently convertible into 156,249 shares of common Stock. Mr. Pendell is also an affiliate of ASC XGS, LLC, which owns 166,023 shares of Series A Preferred Stock which are currently convertible into 311,293 shares of common stock. Mr. Pendell disclaims beneficial ownership of any shares and warrants owned by AAOF, XGS II or ASC XGS. |
76
| (12) | Molly P. Zhang figures include: (i) 7,500 shares of common stock; and (ii) 1,875 shares of common stock underlying currently exercisable options. |
| (13) | Jacqueline M. Lemke figures include: (i) 5,000 shares of common stock; and (ii) 30,000 shares of common stock underlying currently exercisable options. |
| (14) | Bamidele Ali figures include: (i) 40,000 shares of common stock underlying currently exercisable options. |
| (15) | Scott Murray figures include (i) 100 shares of common stock; and (ii) 40,000 shares of common stock underlying currently exercisable options. |
| (16) | Robert Blinstrub figures include: (i) 32,500 shares of common stock. |
| (17) | Leroy Magwood does not currently own any common or other stock. |
| (18) | Aspen Advanced Opportunity Fund, LP figures include: (i) 2,076,386 shares of common stock underlying 1,107,406 shares of immediately convertible Series A Preferred Stock, and (ii) 1,562,499 shares of common stock issuable upon the exercise and conversion of currently exercisable warrants to purchase 833,333 shares of Series A Preferred Stock. |
| (19) | POSCO figures include: (i) 200,000 shares of common stock, (ii) 187,500 shares of common stock underlying 100,000 shares of immediately convertible Series A Preferred Stock, and (iii) 93,750 shares of common stock issuable upon the exercise and conversion of currently exercisable warrants to purchase 50,000 shares of Series A Preferred Stock. |
| (20) | ASC XGS, LLC figures include 311,293 shares of common stock underlying 166,023 shares of immediately convertible Series A Preferred Stock. |
| (21) | XGS II, LLC figures include: (i) 204,697 shares of common stock underlying 109,172 shares of immediately convertible Series A Preferred Stock, and (ii) 156,249 shares of common stock issuable upon the exercise and conversion of currently exercisable warrants to purchase 83,333 shares of Series A Preferred Stock. |
| (22) | SVIC No. 15 New Technology Business Investment LLP, an investment vehicle owned by Samsung Group, figures include 590,079 shares of common stock underlying 314,709 shares of immediately convertible Series A Preferred Stock. |
| (23) | Michal Knox figures include: (i) 121,667 shares of common stock; (ii) 5,000 shares of common stock underlying currently exercisable warrants; (iii) 135,423 shares of common stock underlying 72,226 shares of immediately convertible shares of Series A Preferred Stock; and (iii) 5,380 shares of common stock issued in the name of his wife, Linnea Van Dyne. Mr. Knox retired from the Company and resigned as a Director on February 24, 2016. |
Change in Control Arrangements
We are not aware of any arrangements that could result in a change of control.
77
| ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS, AND DIRECTOR INDEPENDENCE |
Related Party Transactions
During 2018, Mr. Allemang, Jones and Pendell and certain of their affiliates invested the following amounts into our Offering and purchased the number of shares indicated below.
| Purchase Dates (2018) | Amount Invested | Shares Purchased | ||||||||
| Arnold Allemang and affiliates | April 19 and June 27, 2018 | $ | 1,500,000 | 187,500 | ||||||
| Steven C. Jones and affiliates | June 27 – August 29, 2018 | $ | 1,065,944 | 133,243 | ||||||
| David G. Pendell and affiliates | September 30, 2018 | $ | 20,000 | 2,500 | ||||||
| Total | $ | 2,585,944 | 323,243 | |||||||
In conjunction with a financing with AAOF, we and our stockholders listed therein entered into a Shareholder Agreement on March 18, 2013 that contains a number of specific provisions pertaining to the Board of Directors as well as individual Directors. On February 26, 2016, we amended the Shareholder Agreement.
Among other things, the Shareholder Agreement provides for certain voting and nomination rights to be calculated on the basis of “Full Conversion” stock ownership (under which calculation, all convertible notes, preferred shares, or other convertible equity securities are deemed converted into common stock) as follows:
| ● | So long as AAOF or its affiliates own 10% of more of the aggregate outstanding Shareholder Stock (as defined in the Shareholder Agreement): |
| - | the size of the Board of Directors shall be set at seven individuals. |
| - | one person nominated by AAOF shall be elected to the Board of Directors. |
| - | two members of the Board of Directors, other than those nominated by AAOF, POSCO or Hanwha Chemical, shall qualify as independent Directors. |
| ● | So long as POSCO owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by POSCO shall be elected to the Board of Directors. POSCO does not currently own at least 10% of the aggregate outstanding Shareholder Stock and therefore, there is no POSCO representative on the Board of Directors. |
| ● | So long as Hanwha Chemical owns 10% of more of the aggregate outstanding Shareholder Stock, one person nominated by Hanwha Chemical shall be elected to the Board of Directors. Hanwha does not currently own at least 10% of the aggregate outstanding Shareholder Stock and therefore, there is no Hanwha representative on the Board of Directors. |
As of December 31, 2016, the ownership percentage of AAOF, as calculated for purposes of Director voting, required the shareholders bound by the Shareholder Agreement to vote for a Director nominated by AAOF. Mr. Jones is the AAOF representative to the Board pursuant to the terms of the Shareholder Agreement.
78
The Shareholder Agreement grants preemptive rights to shareholders and holders of convertible notes who are parties to the Shareholder Agreement. Pursuant to the terms therein, such shareholders and noteholders have the right to purchase their pro rata share of all shareholder stock that the Company may, from time to time, propose to sell, issue, or exchange after the date of the Shareholder Agreement, other than certain excluded stock which includes stock granted to employees or as merger consideration. Each shareholder’s pro rata shares shall be equal to the ratio of (i) the aggregate number of shares of the Company’s common stock on a fully diluted basis, owned by the such shareholder at the time of the delivery of a preemptive rights notice to (ii) the aggregate number of shares of Company’s common stock on a fully diluted basis owned by all of the Company’s shareholders at the time of the delivery of a preemptive rights notice.
The Shareholder Agreement may be amended or terminated by agreement (either generally or in a particular instance and either retroactively or prospectively), only with the written consent of (i) a majority of the Board, and (ii) persons holding, in the aggregate, shares of Shareholder Stock representing at least sixty percent (60%) of the voting power of all shares of Shareholder Stock then held by the parties thereto and their permitted assignees.
The February 26, 2016 amendment provides that holders of Excluded Stock are not subject to the terms of the Shareholder Agreement. Excluded Stock means shares of common stock that are subject to a registration statement that has been filed with the SEC and has been declared effective, and, for the avoidance of any doubt, includes the 3,000,000 shares being offered under the Registration Statement. This amendment took effect upon the effectiveness of our Registration Statement. The Amendment to the Shareholder Agreement further clarifies that preemptive rights shall not apply to Excluded Stock (including, without limitation, the 3,000,000 shares being offered under the Registration Statement), and amends the termination date of the Shareholder Agreement.
Per the amendment on May 30, 2018, the Shareholder Agreement will continue in effect, unless the Shareholder Agreement is earlier terminated in accordance with its terms until the date on which the Company’s common stock is listed on the NASDAQ Stock Market of the New York Stock Exchange. As a result, the Shareholder Agreement will continue to remain in effect and certain of our larger shareholders will be entitled to continue to exercise their rights under such Shareholder Agreement, but purchasers of shares of common stock under the registration statement filed in connection with our current offering are not required to adopt the Shareholder Agreement.
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The following table sets forth the aggregate fees billed to us for fiscal years ended December 31, 2019 by our current auditors, RSM US LLP and for the fiscal years ended December 31, 2019 and December 31, 2018 by our former accountants, Frazier & Deeter, LLC (in thousands):
| 2019 | 2018 | |||||||
| Audit fees - RSM (1) | $ | 138,870 | $ | 86,700 | ||||
| Audit fees – Frazier Deeter (1) | 15,000 | 39,500 | ||||||
| Non-Audit fees: | — | — | ||||||
| Audit related fees (2) | — | — | ||||||
| Tax fees | — | — | ||||||
| All other fees | — | — | ||||||
| Total fees billed | $ | 153,870 | $ | 126,200 | ||||
| (1) | Audit fees consist of fees billed for professional services rendered for the audit of the Company’s annual consolidated financial statements and reviews of its interim consolidated financial statements included in quarterly reports and other services related to statutory and regulatory filings or engagements. |
| (2) | Audit-related fees principally include assurance and related services by the independent auditors that are reasonably related to registration statement filings that are not captured under “Audit Fees.” |
79
| ITEM 15. | EXHIBITS AND FINANCIAL STATEMENT SCHEDULES |
80
81
Financial Statement Schedules
None.
82
In accordance with Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| XG SCIENCES, INC. | ||
| Dated: April 29, 2020 | By: | /s/ Philip L. Rose |
| Name: | Philip L. Rose | |
| Title: | Chief Executive Officer, President, Treasurer, Principal Executive Officer and Director | |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| /s/ Philip L. Rose | Chief Executive Officer, President, Treasurer, | April 29, 2020 | ||
| Philip L. Rose | Principal Executive Officer and Director | |||
| /s/ Jacqueline M. Lemke | Chief Financial Officer | April 29, 2020 | ||
| Jacqueline M. Lemke | ||||
| /s/ Arnold A. Allemang | Chairman of the Board | April 29, 2020 | ||
| Arnold A. Allemang | ||||
| /s/ Steven C. Jones | Director | April 29, 2020 | ||
| Steven C. Jones | ||||
| /s/ Molly P. Zhang | Director | April 29, 2020 | ||
| Molly P. Zhang | ||||
| /s/ Dave Pendell | Director | April 29, 2020 | ||
| Dave Pendell | ||||
| /s/ Robert Blinstrub | Director | April 29, 2020 | ||
| Robert Blinstrub |
SUPPLEMENTAL INFORMATION TO BE FURNISHED WITH REPORTS FILED PURSUANT TO SECTION 15(D) OF THE ACT BY REGISTRANTS WHICH HAVE NOT REGISTERED SECURITIES PURSUANT TO SECTION 12 OF THE ACT.
No such annual report, proxy statement, form of proxy or other soliciting material has been sent to its shareholders. The registrant will not be sending an annual report or proxy material to its shareholders subsequent to the filing of this form.
83