| ☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| Delaware | 81-4282653 | |
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |
| 3050 Science Park Road | ||
| San Diego, California | 92121 | |
| (Address of principal executive offices) | (Zip Code) | |
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||
| Common Stock, par value $0.0001 per share | RXDX | Nasdaq Global Select Market |
| Large accelerated filer | ☒ | Accelerated filer | ☐ | |||
| Non-accelerated filer | ☐ | Smaller reporting company | ☐ | |||
| Emerging growth company | ☐ | |||||
| Auditor Name: | Auditor Location: | PCAOB ID# : | ||
| BDO USA, LLP | San Diego, CA | 243 |
| Page | ||||||
| Explanatory Note | i | |||||
| 1 | ||||||
| Item 10. | Directors, Executive Officers and Corporate Governance | 1 | ||||
| Item 11. | Executive Compensation | 10 | ||||
| Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholders Matters | 29 | ||||
| Item 13. | Certain Relationships and Related Transactions, and Director Independence | 31 | ||||
| Item 14. | Principal Accounting Fees and Services | 34 | ||||
| 35 | ||||||
| Item 15. | Exhibits and Financial Statement Schedules | 35 | ||||
| Signatures | ||||||
PART III
ITEM 10. DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
Board of Directors
Our Board of Directors (the Board) is currently composed of eight members. Information with respect to our directors is set forth below:
Name | Age* | Position | Independent | Committee Membership | ||||
| CLASS I DIRECTORS | ||||||||
| Joseph C. Papa | 67 | Lead Independent Director | X | Compensation (Chair) | ||||
| Mary Szela | 59 | Director | X | Audit; Compensation | ||||
| CLASS II DIRECTORS | ||||||||
| Helen C. Adams, CPA | 64 | Director | X | Audit (Chair) | ||||
| Martin Hendrix, Ph.D. | 55 | Director | X | Audit; Nominating and Corporate Governance | ||||
| Judith L. Swain, M.D. | 74 | Director | X | Nominating and Corporate Governance | ||||
| CLASS III DIRECTORS | ||||||||
| Mark C. McKenna | 43 | Chairman, President and Chief Executive Officer | ||||||
| Fred Hassan | 77 | Director | X | Nominating and Corporate Governance (Chair); Compensation | ||||
| James Laur | 62 | Director | ||||||
* As of April 21, 2023
Class I Directors
Joseph C. Papa has served on our board of directors since August 2020 and as our Lead Independent Director since August 2021. Mr. Papa has been Chief Executive Officer of Bausch and Lomb since May 2022 and prior to that Chairman of the board of directors and Chief Executive Officer of Bausch Health Companies Inc. since May 2016. Mr. Papa has more than 35 years of experience in the pharmaceutical, healthcare and specialty pharmaceutical industries, including 20 years of branded prescription drug experience. He served as the Chief Executive Officer of Perrigo Company plc from 2006 to April 2016, where he also served as Chairman from 2007 to April 2016. Prior to joining Perrigo, Mr. Papa served from 2004 to 2006 as Chairman and Chief Executive Officer of the Pharmaceutical and Technologies Services segment of Cardinal Health, Inc. From 2001 to 2004, he served as President and Chief Operating Officer of Watson Pharmaceuticals, Inc. Prior to joining Watson, Mr. Papa held management positions at DuPont Pharmaceuticals, Pharmacia/Searle and Novartis AG. Mr. Papa served as a director of Smith & Nephew plc, a developer of advanced medical devices, from 2008 to April 2018. Mr. Papa’s extensive leadership experience in the pharmaceutical industry contributed to our board of directors’ conclusion that he should serve as a director of our company.
Mary Szela has served as a member of our board of directors since February 2021. Ms. Szela also currently serves as the Chief Executive Officer and President of TriSalus Life Sciences, Inc., a privately held oncology company. From January 2016 to November 2016, Ms. Szela served as Chief Executive Officer and a director of Aegerion Pharmaceuticals, Inc. In November 2016, Aegerion Pharmaceuticals, Inc. merged with QLT Inc. to form Novelion Therapeutics Inc. where Ms. Szela served as Chief Executive Officer and as a member of its board of directors until November 2017. Ms. Szela served as the Chief Executive Officer and a member of the board of directors of Melinta Therapeutics, Inc., an antibiotic development company, from April 2013 to August 2015. Ms. Szela held ascending management positions at Abbott Laboratories from 1987 to 2012, including President of the company’s U.S. pharmaceutical business from January 2008 to December 2010. Ms. Szela has served as a member of the boards of directors of Kura Oncology, Inc. since November 2018, Omega Therapeutics since June 2019, Senda Biosciences since July 2019 and TriSalus Life Sciences, Inc. since January 2018. She also previously served as a member of the board of directors of Coherus Biosciences from 2014 to August 2021, Alimera Sciences Inc. from June 2018 to March 2021, Receptos, Inc. from June 2014 to July 2015, Novo Nordisk from March 2014 to March 2017, and Macrolide Pharmaceuticals, from March 2018 to July 2019. She earned an M.B.A. in Business and a B.S. in nursing, both from the University of Illinois. Ms. Szela’s extensive leadership experience in the pharmaceutical industry contributed to our board of directors’ conclusion that she should serve as a director of our company.
1
Class II Directors
Helen C. Adams, CPA has been a member of our board of directors since February 2021. From January 2013 to March 2018, Ms. Adams was the San Diego Area Managing Partner for Haskell & White LLP, a regional certified public accounting firm. Previously, Ms. Adams was a certified public accountant at Deloitte & Touche LLP from 1982 to 2009, serving most recently as a Partner in the Life Sciences and Technology Group. Since June 2021, Ms. Adams has served as a member of the board and the Audit Committee Chair of Immix Biopharma, Inc. From 2010 to 2013, Ms. Adams was a member of the board of directors of Genasys Inc., serving as the Audit Committee Chair and member of the Compensation Committee. In addition to her public company board service, Ms. Adams has served on the boards of directors of several organizations, including Athena San Diego, the Athena Foundation, Make A Wish San Diego and the California State University at San Marcos Foundation. Ms. Adams’ experience with accounting, auditing and financial oversight of public and private companies in the life sciences industry contributed to our board of directors’ conclusion that she should serve as a director of our company.
Martin Hendrix, Ph.D. has served as a member of our board of directors since October 2020. He joined Nestlé Health Science in April 2012, and currently serves as its Head of Global Business Development and M&A. In this position, Dr. Hendrix oversees all deal flow of Nestlé Health Science and is also responsible for its venture capital partnerships and direct equity investments. Dr. Hendrix has represented Nestlé on the boards of Enterome, Microbiome Diagnostic Partners, Procise Dx, Bodymed AG, as well as board observer roles for Evelo, Kaleido and Senda. Prior to joining Nestlé, from January 1998 to March 2012, Dr. Hendrix was a research chemist and subsequently a member of the Strategic Planning Group, as well as a Senior Director of Business Strategy at Bayer AG. Dr. Hendrix currently serves on the board of directors of Axcella Health Inc., Procise Dx, Bodymed AG, Senda (observer) and Prometheus Laboratories, Inc. Dr. Hendrix holds a Ph.D. in Chemistry from The Scripps Research Institute, an M.S. in Chemistry from the Georgia Institute of Technology. Dr. Hendrix’s extensive management and governance experience in the biotechnology industry contributed to our board of directors’ conclusion that Dr. Hendrix should serve as a director of our company.
Judith L. Swain, M.D. has served as a member of our board of directors since February 2021, and is the Chief Medical Officer of Physiowave, Inc., a medical device company, a role she has held since July 2016. She has also served as a Visiting Professor in the Department of Medicine at the National University of Singapore since July 2017. Prior to these roles, Dr. Swain was Executive Director of the Singapore Institute for Clinical Sciences, a research institute of the Agency for Science, Technology and Research (A*STAR), Singapore, and Professor, National University of Singapore from 2006 to 2017. Dr. Swain served as Dean for Translational Medicine at the University of California, San Diego from 2005 to 2006 and as Chair and Professor of Medicine at Stanford University from 1996 to 2005. Dr. Swain is a co-founder of Synecor, LLC and currently serves on the board of directors of Lexicon Pharmaceuticals, Inc. and the Institute for Life Changing Medicines. She previously served on the boards of directors of Upstream Medical Technologies, Ltd, the National Healthcare Group (Singapore) and the Lee Kong Chian School of Medicine of Nanyang Technological University. Dr. Swain earned an M.D. from the University of California, San Diego, and a B.S. in Chemistry from the University of California, Los Angeles. Dr. Swain’s extensive medical and scientific research experience with a variety of prominent research and academic institutions contributed to our board of directors’ conclusion that she should serve as a director of our company.
2
Class III Directors
Mark C. McKenna has served as our President and Chief Executive Officer and as a member of our board of directors since September 2019 and as our Chairman since August 2021. Prior to joining us, he served as President of Salix Pharmaceuticals, Inc. (Salix), a wholly-owned subsidiary of Bausch Health Companies, Inc. (Bausch), from March 2016 through August 2019. Prior to Salix, Mr. McKenna spent more than a decade in various roles with Bausch + Lomb, also a division of Bausch, most recently as Senior Vice President and General Manager of its U.S. Vision Care business. Before joining Bausch + Lomb, he held several positions with Johnson & Johnson. Mr. McKenna has served as a member of the board of Rebalance Health, Inc. since December 2021. Mr. McKenna holds a B.S. in Marketing from Arizona State University and an M.B.A. from Azusa Pacific University. Mr. McKenna’s knowledge of our business, as well as his significant development, commercial and executive management experience, contributed to our board of directors’ conclusion that he should serve as a director of the company.
Fred Hassan has served on our board of directors since May 2021. Mr. Hassan currently serves as Director of Warburg Pincus LLC, a global private equity investment institution which he joined in 2009. Previously, Mr. Hassan served as Chairman and Chief Executive Officer of Schering-Plough from 2003 to 2009. Before assuming these roles, from 2001 to 2003, Mr. Hassan was Chairman and Chief Executive Officer of Pharmacia Corporation, a company formed through the merger of Monsanto Company and Pharmacia & Upjohn, Inc. He joined Pharmacia & Upjohn, Inc. as Chief Executive Officer in 1997. Mr. Hassan has served as a director of BridgeBio Pharma, Inc. since 2021 and Precigen, Inc. since 2016. Previously, Mr. Hassan served as a director of Amgen, Inc. from July 2015 to May 2021, Time Warner Inc. (now Warner Media, LLC) from October 2009 to June 2018 and as a director of Avon Products, Inc. from 1999 until 2013. He was Chairman of the Board of Bausch & Lomb from 2010 until its acquisition by Valeant Pharmaceuticals International, Inc. in 2013 and served on the board of Valeant Pharmaceuticals from 2013 to 2014. Mr. Hassan received a B.S. in Chemical Engineering from the Imperial College of Science and Technology at the University of London and an M.B.A. from Harvard Business School. Mr. Hassan’s extensive executive leadership and corporate governance experience in the biotechnology industry contributed to our board of directors’ conclusion that Mr. Hassan should serve as a director of our company.
James Laur has served on our board of directors since April 2020, and through his work leading Cedars-Sinai Medical Center’s Technology Transfer Office, Mr. Laur played a role in our founding. Mr. Laur currently serves as Vice President, Intellectual Property for Cedars-Sinai, and, in addition to his role with Cedars-Sinai’s Technology Transfer Office, Mr. Laur is responsible for the Cedars-Sinai Accelerator program and is the Managing Director of Cedars-Sinai Health Ventures, Cedars-Sinai’s venture fund. Mr. Laur has served in a variety of business roles at Cedars-Sinai since originally joining its Legal Affairs Department in 1991. Mr. Laur is a member of the California State Bar and holds a dual degree B.A. in Political Science and Philosophy from the University of California, Los Angeles, as well as a J.D. from Boston College Law School. Mr. Laur’s extensive experience with early-stage life science companies, the healthcare sector, as well as his knowledge of intellectual property issues, technology ventures, innovation development and commercialization strategies contributed to our board of directors’ conclusion that he should serve as a director of our company.
Executive Officers
The following sets forth information about our executive officers. Biographical information for Mr. McKenna is included in the section above titled “Board of Directors.”
| Name | Age* | Position | ||
Mark C. McKenna | 43 | Chairman, President and Chief Executive Officer | ||
Keith W. Marshall, Ph.D. | 55 | Chief Financial Officer and Treasurer | ||
Mark Stenhouse | 56 | Chief Operating Officer |
*As of April 21, 2023.
3
Keith W. Marshall, Ph.D. has served as our Chief Financial Officer and Treasurer served since August 2020. Previously, he served as Executive Vice President, Chief Operating Officer, and Chief Financial Officer of Conatus Pharmaceuticals Inc., now Histogen, Inc., from August 2017 to May 2020. Dr. Marshall served as Chief Financial Officer and Head of Corporate Development at Torque Therapeutics Inc. from 2015 to 2017, where his responsibilities included finance, operations, human resources, corporate strategy and business development. He served as Managing Director and Advisor in Healthcare Investment Banking from 2012 to 2014 at GCA Savvian Advisors, where he provided strategic counsel to healthcare companies, and continued from 2014 to 2015 at TAG Healthcare Advisors under an alliance with GCA Savvian. Previously, Dr. Marshall was Managing Director from 2011 to 2012 at Sagent Advisors and Managing Director, Co-founder, and Chief Financial Officer from 2008 to 2011 at Montgomery, Marshall Healthcare Partners. Dr. Marshall began his banking career at JPMorgan. He holds an A.B. in Biology from Washington University in St. Louis, a Ph.D. in Pharmaceutical Chemistry from the University of California, San Francisco, and an M.B.A. with concentrations in Finance, Strategy, and Entrepreneurship from the University of Chicago Booth School of Business.
Mark Stenhouse has served as our Chief Operating Officer since March 2021. Previously, Mr. Stenhouse served as a member of our board of directors from April 2018 to October 2020 and then as an advisor to our company through February 2021. Mr. Stenhouse served as General Manager, Screening at Exact Sciences Corporation from November 2019 to January 2021 and as an advisor to the company through February 2021. Mr. Stenhouse served as President, Cologuard at Exact Sciences Corporation from April 2018 until November 2019. From October 2016 until March 2018, Mr. Stenhouse served as Vice President, U.S. Immunology of AbbVie, Inc., where he oversaw U.S. expansion into the immunology marketplace. From April 2010 until September 2016, Mr. Stenhouse served as Vice President and Vice President/General Manager, U.S. Immunology-Gastroenterology Franchise at AbbVie. From September 2006 through March 2010, Mr. Stenhouse held various senior management, marketing and sales positions within Abbott Laboratories’ U.S. Immunology division. Mr. Stenhouse has served as a member of the Board of Directors of Phathom Pharmaceuticals, Inc. since March 2020. Mr. Stenhouse holds a B.B.A. from the College of Charleston.
Corporate Governance
General
The Board has adopted Corporate Governance Guidelines, a Code of Conduct and Ethics and charters for our Nominating and Corporate Governance Committee, Audit Committee and Compensation Committee to assist the Board in the exercise of its responsibilities and to serve as a framework for the effective governance of the Company. You can access our current committee charters, and our Code of Conduct and Ethics in the “Governance” section of the “Investors” page of our website located at www.prometheusbiosciences.com, or by writing to our Secretary at our offices at 3050 Science Park Road, San Diego, California 92121. Please note, however, that the information contained on the website is not incorporated by reference in, or considered part of, this Amendment.
Board Diversity and Director Nomination Process
Our Nominating and Corporate Governance Committee is responsible for reviewing with the Board, on an annual basis, the appropriate characteristics, skills and experience required for the Board as a whole and its individual members. In evaluating the suitability of individual candidates (both new candidates and current members) for election or appointment, the Nominating and Corporate Governance Committee, in recommending candidates for election, and the Board will take into account many factors, including the following:
| • | personal and professional integrity, ethics and values; |
| • | experience in corporate management, such as serving as an officer or former officer of a publicly-held company; |
| • | experience as a board member or executive officer of another publicly-held company; |
| • | strong finance experience; |
| • | diversity of expertise and experience in substantive matters pertaining to our business relative to other board members; |
4
| • | diversity of background and perspective, including, but not limited to, with respect to age, gender, race, place of residence and specialized experience; |
| • | experience relevant to our business industry and with relevant social policy concerns; and |
| • | relevant academic expertise or other proficiency in an area of our business operations. |
Currently, the Board evaluates each individual in the context of the Board as a whole, with the objective of assembling a group that can best maximize the success of the business and represent stockholder interests through the exercise of sound judgment using its diversity of experience in these various areas.
Other than the foregoing, there are no stated minimum criteria for director nominees, although the Nominating and Corporate Governance Committee may also consider such other factors as it may deem to be in the best interests of our company and our stockholders. The Nominating and Corporate Governance Committee does, however, believe it is appropriate for at least one member of our Board to meet the criteria for an “audit committee financial expert” as defined by SEC rules, and that a majority of the members of our Board meet the definition of “independent director” under the Nasdaq Global Select Market (Nasdaq) qualification standards. The Nominating and Corporate Governance Committee also believes it is appropriate for our President and Chief Executive Officer to serve as the Chairman of our Board.
The following Board Diversity Matrix presents our Board diversity statistics in accordance with Nasdaq Rule 5606, as self-disclosed by our directors.
Board Diversity Matrix (As of April 28, 2023) | ||||||||||||
Board Size | ||||||||||||
Total Number of Directors | 8 | |||||||||||
| Female | Male | |||||||||||
Part I: Gender Identity | ||||||||||||
Directors | 3 | 5 | ||||||||||
Part II: Demographic Background | ||||||||||||
Asian | 0 | 1 | ||||||||||
White | 3 | 4 | ||||||||||
LGBTQ+ | 1 | |||||||||||
Identification and Evaluation of Nominees for Directors
The Nominating and Corporate Governance Committee identifies nominees for director by first evaluating the current members of our Board willing to continue in service. Current members with qualifications and skills that are consistent with the Nominating and Corporate Governance Committee’s criteria for board of director service and who are willing to continue in service are considered for re-nomination, balancing the value of continuity of service by existing members of our Board with that of obtaining a new perspective or expertise. If any member of our Board does not wish to continue in service or if our Board decides not to re-nominate a member for re-election or if the Board decides to expand the size of the board, the Nominating and Corporate Governance Committee identifies the desired skills and experience of a new nominee in light of the criteria above. The Nominating and Corporate Governance Committee generally polls our Board and members of management for their recommendations. The Nominating and Corporate Governance Committee may also review the composition and qualification of the boards of directors of our competitors and may seek input from industry experts or analysts. The Nominating and Corporate Governance Committee reviews the qualifications, experience and background of the candidates. Final candidates are interviewed by the members of the Nominating and Corporate Governance Committee and by certain of our other independent directors and executive management. In making its determinations, the Nominating and Corporate Governance Committee evaluates each individual in the context of our Board as a whole, with the objective of assembling a group that can best contribute to the success of our company and represent stockholder interests through the exercise of sound business judgment. After review and deliberation of all feedback and data, the Nominating and Corporate Governance Committee makes its recommendation to our Board.
5
The Nominating and Corporate Governance Committee evaluates nominees recommended by stockholders in the same manner as it evaluates other nominees. We have not received director candidate recommendations from our stockholders, and we do not have a formal policy regarding consideration of such recommendations. However, any recommendations received from stockholders will be evaluated in the same manner that potential nominees suggested by members of our Board, management or other parties are evaluated.
Under our amended and restated bylaws (Bylaws), a stockholder wishing to suggest a candidate for director should write to our Secretary and provide such information about the stockholder and the proposed candidate as is set forth in our Bylaws and as would be required by SEC rules to be included in a proxy statement. In addition, the stockholder must include the consent of the candidate and describe any arrangements or undertakings between the stockholder and the candidate regarding the nomination. In order to give the Nominating and Corporate Governance Committee sufficient time to evaluate a recommended candidate and include the candidate in our proxy statement for the annual meeting, the recommendation should be received by our Corporate Secretary at our principal executive offices not earlier than the close of business on the 120th day and not later than the close of business on the 90th day prior to the anniversary of the preceding year’s annual meeting of stockholders.
Communication from Stockholders
The Board will give appropriate attention to written communications that are submitted by stockholders, and will respond if and as appropriate. Our Secretary is primarily responsible for monitoring communications from stockholders and for providing copies or summaries to the directors as he considers appropriate.
Communications are forwarded to all directors if they relate to important substantive matters and include suggestions or comments that our Secretary and Chairman of the Board consider to be important for the directors to know. In general, communications relating to corporate governance and long-term corporate strategy are more likely to be forwarded than communications relating to ordinary business affairs, personal grievances and matters as to which we tend to receive repetitive or duplicative communications. Stockholders who wish to send communications on any topic to the Board should address such communications to the Board in writing: c/o Secretary, Prometheus Biosciences, Inc., 3050 Science Park Road, San Diego, California 92121.
Director Attendance at Annual Meetings
Although we do not have a formal policy regarding attendance by members of our Board at our annual meetings of stockholders, we encourage all of our directors to attend.
Board Leadership Structure – Chairman and Lead Independent Director
The Board is currently led by our Chairman, President and Chief Executive Officer, Mark C. McKenna, and our Lead Independent Director, Joseph C. Papa. The Board recognizes that it is important to determine an optimal board leadership structure to ensure the independent oversight of management as we continue to grow. We separate the roles of Chairman of the Board and Lead Independent Director in recognition of the differences between the two roles. The Chairman of the Board is responsible for setting the strategic direction for our Company, takes a lead role in managing the Board and facilitating effective communication among directors and presides over meetings of the full Board. The Lead Independent Director provides leadership to the independent directors, liaises on behalf of the independent directors and ensures board effectiveness to maintain high-quality governance of our Company and the effective functioning of the Board. We believe that this separation of responsibilities provides a balanced approach to managing the Board and overseeing our Company.
The Board has concluded that our current leadership structure is appropriate at this time. However, the Board will continue to periodically review our leadership structure and may make such changes in the future as it deems appropriate.
6
Role of Board in Risk Oversight Process
The Board has responsibility for the oversight of our risk management processes and, either as a whole or through its committees, regularly discusses with management our major risk exposures, their potential impact on our business and the steps we take to manage them. The risk oversight process includes receiving regular reports from Board committees and members of senior management to enable the Board to understand our risk identification, risk management and risk mitigation strategies with respect to areas of potential material risk, including operations, finance, legal, regulatory, strategic and reputational risk.
The Audit Committee reviews information regarding liquidity and operations, and oversees our management of financial risks. Periodically, the Audit Committee reviews our policies with respect to risk assessment, risk management, loss prevention and regulatory compliance. Oversight by the Audit Committee includes direct communication with our external auditors, and discussions with management regarding significant risk exposures and the actions management has taken to limit, monitor or control such exposures. The Compensation Committee is responsible for assessing whether any of our compensation policies or programs has the potential to encourage excessive risk-taking. The Nominating and Corporate Governance Committee manages risks associated with the independence of the Board, corporate disclosure practices, and potential conflicts of interest. While each committee is responsible for evaluating certain risks and overseeing the management of such risks, the entire Board is regularly informed through committee reports about such risks. Matters of significant strategic risk are considered by the Board as a whole.
Annual Board Evaluation
Our Corporate Governance Guidelines require the Nominating and Corporate Governance Committee to oversee an annual assessment by the Board of the Board’s performance. As provided in our Corporate Governance Guidelines, the Nominating and Corporate Governance Committee is responsible for establishing the evaluation criteria and implementing the process for such evaluation.
Code of Ethics
We have a written Code of Conduct and Ethics that applies to our directors, officers and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. We have posted a current copy of the code in the “Governance” section of the “Investors” page of our website located at www.prometheusbiosciences.com.
In addition, we intend to post on our website all disclosures that are required by law or the listing standards of Nasdaq concerning any amendments to, or waivers from, any provision of the code.
Policy on Speculative Trading Activities—Anti-Hedging and Pledging Policy
Our insider trading policy prohibits officers, directors and employees, and entities controlled by such individuals and members of their households, from making short sales in our equity securities, transacting in puts, calls or other derivative securities involving our equity securities, on an exchange or in any other organized market, engaging in hedging transactions, purchasing our securities on margin or pledging our securities as collateral for a loan.
Attendance by Members of the Board of Directors at Meetings
There were six meetings of the Board during the fiscal year ended December 31, 2022. During the fiscal year ended December 31, 2022, each director attended at least 75% of the aggregate of all meetings of the Board, and each director attended as least 75% of meetings of the committees on which such director served during the period in which he or she served as a director.
Committees of the Board
The Board has established three standing committees—Audit, Compensation and Nominating and Corporate Governance—each of which operates under a charter that has been approved by the Board. All of the members of each of the Board’s three standing committees are independent as defined under the Nasdaq rules. In addition, all members of the Audit Committee meet the independence requirements for Audit Committee members under Rule 10A-3 under the Exchange Act.
7
The current members of each of the Board committees and committee Chairs are set forth in the following chart.
| Name of Director | Audit | Compensation | Nominating and Corporate Governance | |||
Helen C. Adams, CPA† | C† | — | — | |||
Fred Hassan | — | X | C | |||
Martin Hendrix, Ph.D. | X | — | X | |||
Joseph C. Papa | — | C | — | |||
Judith L. Swain, M.D. | — | — | X | |||
Mary Szela | X | X | — |
| C | Committee Chairperson |
| † | Financial Expert |
Audit Committee
The Audit Committee’s main function is to oversee our accounting and financial reporting processes and the audits of our financial statements. This committee’s responsibilities include, among other things:
| • | appointing our independent registered public accounting firm; |
| • | evaluating the qualifications, independence and performance of our independent registered public accounting firm; |
| • | approving the audit and non-audit services to be performed by our independent registered public accounting firm; |
| • | reviewing the design, implementation, adequacy and effectiveness of our internal accounting controls and our critical accounting policies; |
| • | discussing with management and the independent registered public accounting firm the results of our annual audit and the review of our quarterly unaudited financial statements; |
| • | reviewing, overseeing and monitoring the integrity of our financial statements and our compliance with legal and regulatory requirements as they relate to financial statements or accounting matters; |
| • | reviewing on a periodic basis, or as appropriate, any investment policy and recommending to the Board any changes to such investment policy; |
| • | reviewing with management and our auditors any earnings announcements and other public announcements regarding our results of operations; |
| • | preparing the report that the SEC requires in our annual proxy statement; |
| • | reviewing and approving any related party transactions and reviewing and monitoring compliance with our code of conduct and ethics; and |
| • | reviewing and evaluating, at least annually, the performance of the audit committee and its members including compliance of the audit committee with its charter. |
The members of our Audit Committee are Ms. Adams, Dr. Hendrix and Ms. Szela. Ms. Adams serves as the Chairperson of the Audit Committee. All members of our Audit Committee meet the requirements for financial literacy under the applicable rules and regulations of the SEC and Nasdaq. The Board has determined that Ms. Adams is an “audit committee financial expert” as defined by applicable SEC rules and has the requisite financial sophistication as defined under the applicable Nasdaq rules and regulations. The Board has determined each of Ms. Adams, Dr. Hendrix and Ms. Szela are independent under the applicable rules of the SEC and Nasdaq. All members of our audit committee are independent under Nasdaq rules and Rule 10A-3. The audit committee operates under a written charter that satisfies the applicable standards of the SEC and Nasdaq, which the Audit Committee will evaluate and review at least annually. The Audit Committee met four times during 2022. Both our external auditor and internal financial personnel meet privately with the Audit Committee and have unrestricted access to this committee.
8
Compensation Committee
Our Compensation Committee reviews, approves and recommends to the Board policies relating to compensation and benefits of our officers, employees and directors. The Compensation Committee approves corporate goals and objectives relevant to the compensation of our chief executive officer and other executive officers, evaluates the performance of these officers in light of those goals and objectives and approves the compensation of these officers based on such evaluations. The Compensation Committee also reviews and approves or makes recommendations to the Board regarding the issuance of stock options and other awards under our equity plan. In addition, the Compensation Committee periodically reviews and recommends to the Board compensation for service on the Board and any committees of the Board. The Compensation Committee may delegate its authority under its charter to one or more subcommittees as it deems appropriate from time to time as further described in its charter. The Compensation Committee will review and evaluate, at least annually, its charter, as well as review and evaluate, at least annually, the performance of the Compensation Committee and its members, including compliance by the compensation committee with its charter.
The Compensation Committee has retained Radford, a division of Aon Consulting, Inc. (Radford), as its independent compensation consultant to advise the Compensation Committee on matters pertaining to director and executive compensation, including advising as to market levels and practices, plan design and implementation, comparable company data, consulting best practices and governance principles, as well as on matters related to employee equity compensation. Radford does not provide any other services to the Company. The Compensation Committee has determined, and Radford has affirmed, that Radford’s work does not present any conflicts of interest and that Radford is independent. In reaching these conclusions, the Compensation Committee considered the factors set forth in Exchange Act Rule 10C-1 and Nasdaq listing standards.
The members of our compensation committee are Mr. Papa, Mr. Hassan and Ms. Szela. Mr. Papa serves as the Chairperson of the committee. Our Board has determined that each member of this committee is independent under the applicable rules and regulations of Nasdaq and is a “non-employee director” as defined in Rule 16b-3 promulgated under the Exchange Act. The Compensation Committee met five times during 2022.
None of the members of our compensation committee is currently, or has at any time been, one of our officers or employees. None of our executive officers currently serves, or has served during the past fiscal year, as a member of the board of directors or compensation committee (or other board committee performing equivalent functions or, in the absence of any such committee, the entire board of directors) of any entity that has one or more executive officers serving as a member of our board of directors or compensation committee.
Nominating and Corporate Governance Committee
The Nominating and Corporate Governance Committee is responsible for assisting the Board in discharging the Board’s responsibilities regarding the identification of qualified candidates to become board members, the selection of nominees for election as directors at our annual meetings of stockholders (or special meetings of stockholders at which directors are to be elected), and the selection of candidates to fill any vacancies on the Board and any committees thereof. In addition, the Nominating and Corporate Governance Committee is responsible for overseeing our corporate governance policies, reporting and making recommendations to the Board concerning governance matters and oversight of the evaluation of the Board. The members of our Nominating and Corporate Governance Committee are Mr. Hassan, Dr. Hendrix and Dr. Swain. Mr. Hassan serves as the Chairperson of the committee. The Board has determined that each member of this committee is independent under the applicable rules and regulations of Nasdaq relating to Nominating and Corporate Governance Committee independence. The Nominating and Corporate Governance Committee operates under a written charter, which the Nominating and Corporate Governance Committee will review and evaluate at least annually. The Nominating and Corporate Governance Committee met two times during 2022.
9
ITEM 11. EXECUTIVE COMPENSATION
This compensation discussion and analysis discusses the material components of the executive compensation program for our executive officers who are named in the “Summary Compensation Table” below, whom we refer to as our “named executive officers.” For 2022, our named executive officers and their positions were as follows:
| • | Mark C. McKenna, Chairman, President and Chief Executive Officer |
| • | Keith W. Marshall, Ph.D., Chief Financial Officer and Treasurer |
| • | Mark Stenhouse, Chief Operating Officer |
This compensation discussion and analysis provides an overview of our executive compensation philosophy, the overall objectives of our executive compensation program, and each compensation component that we provide. In addition, we explain how and why the compensation committee and our board of directors arrived at specific compensation policies and decisions involving our executive officers during the fiscal year ended December 31,
2022.
2022 Business Highlights
2022 was a transformational year for our company. Since completing our initial public offering (IPO) in March 2021, we have been focused, among other things, on rapidly advancing our lead product candidate PRA023 through global Phase 2 clinical trials for the treatment of ulcerative colitis (UC) and Crohn’s disease (CD), pursuing indication expansion opportunities, advancing our other pipeline candidates into the clinic, and ensuring we are financed to execute against our strategic plan.
During 2022, we announced the following significant corporate accomplishments:
| • | Positive results for PRA023 in Phase 2 studies for UC and CD. We reported results from the initial cohort of our ARTEMIS-UC Phase 2 clinical trial and from our APOLLO-CD Phase 2a clinical trial of PRA023, demonstrating strong efficacy and favorable safety results in both studies. We believe these results position PRA023 to be a potential first-in-class and best-in-class treatment for patients suffering from UC and CD. |
| • | Advanced PRA023 into Phase 2 study for SSc-ILD. We initiated our third Phase 2 study of PRA023, for the treatment of systemic sclerosis-associated interstitial lung disease (SSc-ILD) and received Fast Track Designation from the FDA. |
| • | Advanced PRA052 into Phase 1 clinical trial. We initiated a Phase 1 clinical trial in normal healthy volunteers for our second product candidate, PRA052, an antibody blocking CD30 ligand (CD30L), and are also developing a companion diagnostic candidate designed to select potential responders for this program. |
| • | Completed upsized $500 million public offering of common stock. We completed an upsized public offering of $500 million of common stock following the release of our Phase 2 data on PRA023 in December 2022. |
Our remarkable corporate process in 2022 resulted in a meaningful increase in our stock price and the market value of our company. At the end of 2022, our market capitalization was over $5 billion dollars and our stock price had increased to $110.00 per share from an IPO price of $19.00 per share.
From our IPO through the end of 2022, our total shareholder return was 335% relative to the Nasdaq Biotechnology Index total shareholder return (TSR) over the same period of -13%. The following stock performance graph illustrates a comparison from March 12, 2021 (the date of our IPO) through December 31, 2022, of the total cumulative stockholder return on our common stock, the Nasdaq Composite Index and the Nasdaq Biotechnology Index.
10
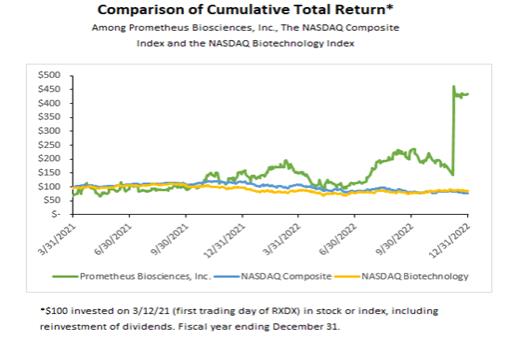
The graph assumes an initial investment of $100 on March 12, 2021 at the opening trading price of $19.00 per share, and that all dividends were reinvested, although dividends have not been declared on our common stock. The comparisons in the graph are required by the SEC and are not intended to forecast or be indicative of possible future performance of our common stock.
Overview of 2022 Executive Compensation Decisions
In general, our named executive officers’ total compensation is tied directly to corporate and individual performance with the exception of the CEO, whose total compensation is tied entirely to corporate performance. Specific elements of our executive compensation program that demonstrate our pay-for-performance philosophy include:
| • | The performance measures in our short-term cash incentive program are linked to key corporate objectives; |
| • | Corporate achievement represents 100% of the annual bonus opportunity for the CEO and 80% of the annual bonus opportunity for each of the other named executive officers; and |
| • | Our long-term equity incentives are provided in the form of options and restricted stock units, which vest over multi-year periods. |
The primary elements of our total direct compensation program for the named executive officers and a summary of the actions taken by the compensation committee during 2022 are set forth below.
11
| Market-Based Base Salary Increases for Named Executive Officers | • Salary adjustments for our named executive officers are based on performance and market-based salary recommendations. Our named executive officers received base salary increases for 2022 consistent with our pay positioning philosophy of targeting target total cash compensation above the market median, placing their salary levels at approximately the 50th percentile of similarly-situated executives at comparable companies based on our peer group. | |
| Annual Cash Incentives Paid Based on Corporate Performance | • Based on our transformational achievement of our corporate performance goals during 2022 despite the difficult biotech and investor environments, our named executive officers’ annual incentives tied to corporate performance were paid out at 200% of target. | |
| Long-Term Incentive Compensation in Form of Stock Options and RSUs Vesting Over Multi-Year Periods | • During 2022, our named executive officers received stock options and restricted stock units (RSUs), which vest over three or four years. Stock options are an important vehicle for tying executive pay to performance, because they deliver future value only if the value of our common stock increases above the exercise price. Both stock options and RSUs provide strong incentives for our executive officers to increase the value of our common stock over the long term, and they tightly align the interests of our executives with those of our stockholders. | |
Our Executive Compensation Practices
We endeavor to maintain sound executive compensation policies and practices consistent with our executive compensation philosophy. The following table highlights some of our executive compensation policies and practices, which are structured to drive performance and align our executives’ interests with our stockholders’ long-term interests:
| ✓ | Pay for Performance. We design our executive compensation program to align pay with company performance. |
| ✓ | Significant Portion of Compensation is at Risk. Under our executive compensation program, a significant portion of compensation is “at risk” based on our performance, including short-term cash incentives and long-term cash and equity incentives, to align the interests of our executive officers and stockholders. |
| ✓ | Independent Compensation Committee. The compensation committee is comprised solely of independent directors. |
| ✓ | Independent Compensation Advisor Reports Directly to the Compensation Committee. The compensation committee engages its own compensation consultant to assist with making compensation decisions. |
| ✓ | Annual Market Review of Executive Compensation. The compensation committee and its compensation consultant annually assess competitiveness and market alignment of our compensation plans and practices. |
| ✓ | Multi-Year Vesting Requirements. The equity awards granted to our executive officers vest over multi-year periods, consistent with current market practice and our retention objectives. |
| ✓ | Minimize Inappropriate Risk Taking. Our compensation program is weighted toward long-term incentive compensation to discourage short-term risk taking, and it includes goals that are quantifiable with objective criteria, multiple performance measures and caps on short-term incentive compensation. |
| ✓ | Competitive Peer Group. Our compensation committee selects our peers from biotechnology and pharmaceutical companies that are similar to us with respect to market capitalization, revenue, headcount and development stage, while also taking into account a number of qualitative criteria. |
12
| ✓ | No Excessive Perquisites. Our named executive officers generally participate in the same welfare and retirement plans as our other employees, and we do not provide our named executive officers with excessive perquisites or other personal benefits. |
| ✓ | Prohibition on Hedging and Pledging. Our insider trading policy prohibits our employees (including executive officers) and directors from engaging in hedging or short-term speculative transactions involving our securities. |
Compensation Philosophy and Objectives
We recognize that the ability to excel depends on the integrity, knowledge, imagination, skill, diversity and teamwork of our employees. To this end, the key objectives of our executive compensation program are:
| • | To attract, engage and retain an executive team who will provide leadership for our future success by providing competitive total pay opportunities; |
| • | To establish a direct link between our business results, individual executive performance and total executive compensation; and |
| • | To align the interests of our executive officers with those of our stockholders. |
Compensation Determination Process
Role of the Compensation Committee
The compensation committee of our board of directors develops, reviews and approves each of the elements of our executive compensation program. The compensation committee also regularly assesses the effectiveness and competitiveness of our compensation programs.
In the first quarter of each year, the compensation committee reviews the performance of each of our named executive officers during the previous year. At this time the compensation committee also reviews our performance relative to the corporate performance objectives set by the board of directors for the year under review and makes the final bonus payment determinations based on our overall corporate performance and the compensation committee’s evaluation of each named executive officer’s performance for the year under review. In connection with this review, the compensation committee also reviews and adjusts, as appropriate, annual base salaries for our named executive officers and grants, as appropriate, additional equity awards to our named executive officers and certain other eligible employees. The compensation committee also reviews and approves the corporate performance objectives for the upcoming fiscal year.
Role of Our Executive Officers
Our Chief Executive Officer, with the assistance and support of our Chief People Officer, aids the compensation committee by providing annual recommendations regarding the compensation of our named executive officers. The compensation committee also, on occasion, meets with our Chief Executive Officer to obtain recommendations with respect to our compensation programs and practices generally. The compensation committee considers, but is not bound to accept, the Chief Executive Officer’s recommendations with respect to named executive officer compensation.
Our Chief Executive Officer generally attends all of the compensation committee meetings, but the compensation committee also holds executive sessions that are not attended by any members of management or non-independent directors, as needed from time to time. Any deliberations or decisions regarding the compensation of our Chief Executive Officer are made without him present.
13
Role of Compensation Consultant and Comparable Company Information
The compensation committee is authorized to retain the services of third-party compensation consultants and other outside advisors, from time to time, to assist in its evaluation of executive compensation, including the authority to approve the consultant’s reasonable fees and other retention terms.
Beginning in 2021, the compensation committee retained Radford, an independent third-party compensation consulting firm for guidance in making compensation decisions. Specifically, for 2022, the compensation committee requested Radford to advise it on a variety of compensation-related issues, including:
| • | conducting an analysis of current practices of comparable public companies to assist the compensation committee in developing director and executive compensation levels; |
| • | reviewing our peer group to determine whether additional or different peer companies or groups are necessary to provide appropriate information on market practices and compensation levels; and |
| • | providing general information concerning director and executive compensation trends and developments. |
Radford did not provide any other services to us in 2022 beyond its engagement as an advisor to the compensation committee on director and executive compensation matters. The compensation committee assessed the independence of Radford pursuant to SEC and Nasdaq rules and concluded that no conflict of interest existed that would have prevented Radford from serving as an independent consultant to the compensation committee currently or during 2022.
Competitive Positioning
The compensation committee reviews our peer group annually to reflect changes in market capitalization and other factors, including acquisitions, and revises the companies included in the peer group accordingly. For 2022, Radford assisted the compensation committee in identifying an appropriate peer group of companies for use as a reference when determining 2022 director and executive compensation. The peer group identified below was selected in September 2021 for purposes of making annual grants to our named executive officers in the fourth quarter of 2021 and for setting 2022 compensation and the selection criteria identified below were measured at such date.
The identified peer group consisted of 21 life sciences companies in similar phases of development as we are with the following characteristics and was selected based on the following parameters and not on the basis of executive compensation levels:
| MARKET CAPITALIZATION | • Generally between $500 million to $3 billion, representing a range of 0.3x to 3x our market capitalization at the time the peer group was established (~$870 million).
• Our market capitalization was positioned at approximately the median of the peers at the time the peer group was selected. | |
| SECTOR AND STAGE | • Public U.S. pre-commercial biotech companies, with a focus on gastroenterology, immunology and precision medicine, when possible.
• Emphasis on Phase 2 companies to reflect our Phase 2 stage of development at the time the peer group was selected. | |
| HEADCOUNT | • Companies with generally fewer than 250 employees based on our projected headcount of approximately 75 employees by the end of 2022. | |
| GEOGRAPHIC LOCATION | • Focused on U.S. companies, with a focus on biotech hubs, when possible. | |
14
This peer group consisted of the following companies:
Aclaris Therapeutics | Poseida Therapeutics | |
Agenus | Precision BioSciences | |
Applied Molecular Transport | Protagonist Therapeutics | |
Arena Pharmaceuticals | RAPT Therapeutics | |
Crinetics Pharmaceuticals | Relay Therapeutics | |
Enanta Pharmaceuticals | Replimune Group | |
Gossamer Bio | Revolution Medicines | |
IDEAYA Biosciences | Rubius Therapeutics | |
Kymera Therapeutics | Seres Therapeutics | |
Morphic | Zentalis Pharmaceuticals | |
Phathom Pharmaceuticals |
Our compensation committee reviewed the foregoing comparable company data in connection with its determinations of the 2022 base salaries, total target cash and equity awards for our named executive officers granted during the fourth quarter of 2021 and the retention equity awards granted in May 2022. The compensation committee generally attempts to set total target compensation opportunities between the 50th and 75th percentiles based on experience and performance.
In August 2022, the compensation committee approved the updated peer group of 20 companies identified below for purposes of making annual grants in the fourth quarter of 2022 and for setting 2023 compensation and the selection criteria identified below were measured at such date. The updated peer group included life sciences companies in similar phases of development as we are with the following characteristics and was selected based on the following parameters and not on the basis of executive compensation levels:
| MARKET CAPITALIZATION | • Generally between $500 million to $4 billion, representing a range of 0.3x to 3x our market capitalization at the time the peer group was established (~$1.1 billion).
• Our market capitalization was positioned at approximately the median of the peers at the time the peer group was selected. | |
| SECTOR AND STAGE | • Public U.S. pre-commercial biotech companies, with a focus on gastroenterology, immunology and precision medicine, when possible.
• Emphasis on Phase 2/ Phase 3 companies to reflect our Phase 2 stage of development at the time the peer group was selected. | |
| HEADCOUNT | • Companies with generally fewer than 250 employees based on our headcount approaching 100 employees. | |
| GEOGRAPHIC LOCATION | • Focused on U.S. companies, with a focus on biotech hubs, when possible. | |
Our updated peer group consisted of the following companies:
Aclaris Therapeutics | Intellia Therapeutics | |
Allogene Therapeutics | Kura Oncology | |
Arvinas | Kymera Therapeutics | |
Celldex Therapeutics | Madrigal Pharmaceuticals | |
Cerevel Pharmaceuticals | Mirati Therapeutics | |
Cogent Therapeutics | Morphic | |
Crinetics Pharmaceuticals | Relay Therapeutics | |
| Denali Therapeutics | Replimune Group | |
| Enanta Pharmaceuticals | Revolution Medicines | |
| Geron | Zentalis Pharmaceuticals |
15
Our compensation committee reviewed the foregoing comparable company data in connection with its determinations of the 2023 base salaries, total target cash and equity awards for our named executive officers granted during the fourth quarter of 2022. The compensation committee generally attempts to set total target compensation opportunities between the 50th and 75th percentiles based on experience and performance.
The compensation committee does not, however, rely entirely on that data to determine named executive officer compensation. Instead, as described above and consistent with past practice, the compensation committee members rely on their judgment and experience in setting those compensation levels and making those awards. As a result, variations on this pay positioning occur from year to year. We expect that the compensation committee will continue to review comparable company data in connection with setting the compensation we offer our named executive officers to help ensure that our compensation programs are competitive and fair.
The compensation levels of the named executive officers reflect to a significant degree the varying roles and responsibilities of such executives. As a result of the compensation committee’s and the board of director’s assessment of our Chief Executive Officer’s roles and responsibilities within our company, there are significant compensation differentials between this named executive officer and our other named executive officers.
Executive Compensation Components
The following describes each component of our executive compensation program, the rationale for each, and how compensation amounts are determined.
Base Salaries
In general, base salaries for our named executive officers are initially established through arm’s length negotiation at the time the executive is hired, taking into account such executive’s qualifications, experience and prior salary. Base salaries of our named executive officers are approved and reviewed annually by our compensation committee and adjustments to base salaries are based on the scope of an executive’s responsibilities, individual contribution, prior experience and sustained performance. This strategy is consistent with our intent of offering compensation that is cost-effective, competitive and contingent on the achievement of performance objectives.
In November 2021, the compensation committee reviewed the base salaries of our named executive officers. The compensation committee, in consultation with our Chief Executive Officer (with respect to the salaries of our other named executive officers) and its independent compensation consultant, determined that the base salaries of our named executive officers would be as follows, which increases were effective January 1, 2022:
Named Executive Officer | 2022 Base Salary | Percentage Increase from 2021 | ||||||
Mark C. McKenna | $ | 592,300 | 3.0 | % | ||||
Keith W. Marshall, Ph.D. | $ | 450,000 | 6.0 | % | ||||
Mark Stenhouse | $ | 437,750 | 3.0 | % | ||||
The foregoing increases placed our named executive officers’ salaries at approximately the 50th percentile of similarly-situated executives, in line with our pay positioning philosophy of setting base salaries at such level. The actual base salaries paid to all of our named executive officers for 2022 are set forth in the “Summary Compensation Table” below.
16
Performance Bonuses
Each named executive officer is also eligible for a performance bonus based upon the achievement of pre-determined corporate performance goals approved by our compensation committee. Bonus targets are established based on percentages of the executives’ respective base salaries for the relevant bonus year. The target levels for executive bonuses in 2022 were as follows: 60% of base salary for Mr. McKenna and 40% of base salary for each of Dr. Marshall and Mr. Stenhouse. The bonus target for Mr. McKenna was increased from the target level of 55% of base salary in effect for 2021. The target bonus levels of our other named executive officers as a percentage of base salary remained unchanged from 2021. All of our named executive officers’ annual bonus payouts are tied to performance relative to the corporate goals set by the compensation committee, with a maximum bonus payout of 200% of target.
At the beginning of each year, the compensation committee sets the corporate goals and milestones for that year, after considering recommendations and input from management and the company’s strategic objectives. For 2022, each named executive officer was eligible for a performance bonus based upon the achievement of these corporate performance goals and objectives, and in the case of Dr. Marshall and Mr. Stenhouse, also based on achievement of individual functional goals. These performance goals and objectives were used as a guide by our compensation committee in determining overall corporate performance for these executives as they represented those areas in which they were expected to focus their efforts during the year.
All final bonus payments to our named executive officers are determined by our compensation committee. The actual bonuses, awarded in any year, if any, may be more or less than the target, depending on the achievement of corporate objectives and may also vary based on other factors at the discretion of the compensation committee.
The compensation committee approved the 2022 corporate performance goals for annual bonus plan purposes. The 2022 performance goals were set at levels such that the attainment of executive target annual cash incentive award opportunities was not assured at the time they were established and would require a high level of effort and execution on the part of the executive officers and others in order to achieve the goals.
To determine the actual attainment of the corporate objectives established for 2022, the compensation committee reviewed the extent to which the corporate performance goals had been achieved or exceeded as well as the relative difficulty of achieving the goals. In light of our strong performance against the objectives set by the compensation committee, all of which were achieved at the maximum level possible, as well as the contributions of Dr. Marshall and Mr. Stenhouse based on individual functional goals, the compensation committee determined to award bonuses to our named executive officers at 200% of the target performance level for 2022.
The table below provides additional details about the 2022 corporate performance goals and specific achievements against our 2022 corporate performance goals that the compensation committee considered in its assessment of corporate and individual performance for 2022 annual bonus plan purposes:
2022 Corporate Goal | Weighting | 2022 Achievements | ||
| Drive PRA023 Development | 50% | • Completed PRA023 Phase 2 clinical trials in UC and CD and reported results positioning PRA023 to be a potential best-in-class and first-in-class therapy
• Initiated PRA023 Phase 2 clinical trial in SSc-ILD program and met 2022 enrollment targets
• Prioritized additional indications for PRA023 clinical development | ||
| Advance Pipeline | 20% | • Filed IND for PRA052 and initiated Phase 1 clinical trial
• Selected additional pipeline targets to advance
• Establish strategy to deliver a third IND in 2023 | ||
17
| Increase Platform Value | 15% | • Expanded Prometheus360TM capabilities to enable enhanced target prioritization
• Developed industrialization plan for PRA023’s genetic-based diagnostic test
• Implemented intellectual property strategies to protect key programs and assets | ||
| Enhance Corporate Capabilities | 15% | • Executed $500 million equity capital raise in 2022
• Outperformed major biotechnology indices and added additional research analysts
• Recognized as “Great Place To Work” by Biospace®, Great Place To Work® and others | ||
| Total | 100% | |||
In determining the annual bonus payouts to Dr. Marshall and Mr. Stenhouse, the compensation committee considered the fact that each of these named executive officers made significant contributions to obtaining the corporate goals outlined above as well as goals for their respective departments. Their performance against these corporate and functional goals were instrumental in achieving the Company’s outstanding results in 2022 and setting the Company up for continued future performance.
The annual bonuses paid to our named executive officers for 2022 are set forth in the Summary Compensation Table below.
Equity-Based Incentive Awards
The goals of our long-term, equity-based incentive awards are to align the interests of our named executive officers and other employees, non-employee directors and consultants with the interests of our stockholders. Because vesting is based on continued employment over multiple years, our equity-based incentives also encourage the retention of our named executive officers through the vesting period of the awards. In determining the size of the long-term equity incentives to be awarded to our named executive officers, we take into account a number of internal factors, such as the relative job scope, the value of existing long-term incentive awards, individual performance history, prior contributions to us, the size of prior grants as well as comparable company information, as described below in addition to market data on equity grant valuations within our peer group and industry. We have no program, plan or practice pertaining to the timing of stock option grants to named executive officers coinciding with the release of material non-public information.
We use equity awards to compensate our named executive officers both in the form of initial grants in connection with the commencement of employment and annual refresher grants. Our compensation committee typically approves annual equity awards during the fourth quarter of each year. While we intend that the majority of stock awards to our employees be made pursuant to initial grants or our annual grant program, the compensation committee retains discretion to make equity awards to employees at other times, including in connection with the promotion of an employee, to reward an employee, for retention purposes or for other circumstances recommended by management or the compensation committee.
Equity Vehicles
Annual equity awards are granted under the Prometheus Biosciences, Inc. 2021 Incentive Award Plan (the 2021 Plan), using a mix of different equity instruments to further its goal of attracting and retaining top performers and to balance the relative advantages of different instruments. During 2022, we granted both stock options and RSUs to our named executive officers.
| • | Stock options are an important vehicle for tying executive pay to performance, because they deliver future value only if the value of our common stock increases above the exercise price. As a result, they provide strong incentives for our executive officers to increase the value of our common stock over the long term, and they tightly align the interests of our executives with those of our stockholders. |
18
| • | RSU awards are granted because they are less dilutive to our stockholders, as fewer shares of our common stock are granted to achieve an equivalent value relative to stock options, and because RSU awards are an effective retention tool that maintain value even in cases where the share price is trading lower than the initial grant price. Our compensation committee determined to include RSUs in the equity award mix for our named executive officers based, in part, on the input of the committee’s independent compensation consultant, who advised that the use of RSU awards along with stock options is increasingly common in our peer group. |
The exercise price of each stock option grant is the fair market value of our common stock on the grant date, as determined by our board of directors from time to time. Option awards generally vest over four years, with 25% vesting on the first anniversary of the applicable vesting commencement date and then in equal monthly installments over a four-year period. RSU awards generally vest in four equal annual installments. From time to time, our compensation committee may, however, determine that a different vesting schedule is appropriate, as was the case with the special retention awards granted in May 2022, as described below.
2022 Equity Awards to Our Named Executive Officers
Generally, the compensation committee determines the value of each executive officer’s annual equity grant using competitive market analysis prepared by its independent compensation consultant with market data for each role, the recommendations of our Chief Executive Officer based on his evaluation of their individual performance (except with respect to his own performance), the extent to which the executive officer is currently vested in his or her stock awards, scope and criticality of the executive’s role and parity in targets among executives in roles of a given level.
In May 2022, the compensation committee approved special equity awards for our named executive officers in the form of stock options which vest in full on the third anniversary of the grant date. These special equity awards were intended by the compensation committee to serve as special retention and incentive awards, given that the retention of our named executive officers was critical during 2022 as the company executed its global Phase 2 clinical trials which were on track to read out results in the fourth quarter of 2022, as well as the tumultuous market conditions since our IPO which had resulted in post-IPO equity awards having considerably reduced retention value.
In order to address these retention concerns and align executive incentives with long-term value creation, the compensation committee determined to grant our named executive officers stock options which would vest in full on the third anniversary of the grant date with a value at the time of grant equal to the annual grant values for their positions as recommended by Radford and based on the 50th percentile our peer group. The corresponding values were then converted into a number of options using the Black-Scholes option pricing model (utilizing the same assumptions that the company utilizes in preparation of its financial statements and the average closing price per share of the company’s common stock for the 30 trading days preceding the grant date).
In December 2022, the compensation committee approved annual equity awards for our named executive officers in the form of stock options and RSUs which vest in accordance with the standard vesting schedules described above. The annual awards were granted 50% in the form of stock options and 50% in the form of RSUs. The compensation committee determined to grant our named executive officers stock options and RSUs with an aggregate value at the time of grant equal to the annual grant values for their positions as recommended by Radford and based on the 50th percentile of our peer group established in August 2022 for Dr. Marshall and Mr. Stenhouse, and the 75th percentile of our peer group established in August 2022 for Mr. McKenna. The corresponding values were then converted into a number of options and RSUs.
The compensation committee determined that this positioning was appropriate for these annual awards given we had recently announced positive results from our PRA023 Phase 2 clinical trials in UC and CD which positioned PRA023 to be a potential best-in-class and first-in-class therapy and resulted in a significant increase in our stock price and TSR relative to the Nasdaq Biotechnology Index since our IPO. As a result, the compensation committee felt that, while they had competitive compensation information for the peer group identified in August 2022, at the time these annual awards were granted, our market capitalization was above the top end of the comparable company range at the time the peer group was selected.
19
Also in December 2022, the compensation committee approved an additional equity award for Dr. Marshall in the form of additional stock options in recognition of his efforts to complete our $500 million equity financing in December 2022. The stock options granted to Dr. Marshall vest in accordance with the standard four-year vesting schedule described above.
The number of options granted to our named executive officers during 2022 was calculated using the Black-Scholes option pricing model (utilizing the same assumptions that the company utilizes in preparation of its financial statements and the average closing price per share of the company’s common stock for the 30 trading days preceding the grant date) and the number of RSUs was calculated using the closing price per share of the company’s common stock on the date prior to the grant date. As a result, the actual aggregate grant-date fair value of these awards, computed in accordance with Accounting Standards Codification (ASC) Topic 718 promulgated by the Financial Accounting Standards Board (FASB), as described below in the footnotes to the Summary Compensation Table, and reflected in the Summary Compensation Table, may differ from the targeted values approved by the compensation committee.
The equity awards granted to our named executive officers during 2022 are set forth in the “Grants of Plan-Based Awards Table” below. For a description of certain accelerated vesting provisions applicable to the stock awards granted to our named executive officers, see “— Employment Letter Agreements with Our Named Executive Officers” below.
Health, Welfare, and Retirement Benefits
Health and Welfare Benefits
Our named executive officers are eligible to participate in our employee benefit plans, including our medical, dental, vision, group life, disability and accidental death and dismemberment insurance plans, in each case on generally the same basis as all of our other employees. We do, however, pay the premiums for enhanced life insurance coverage and pay for an annual comprehensive medical exam for our named executive officers.
Retirement Savings
We maintain a defined contribution employee retirement plan, or 401(k) plan, for our employees. Our named executive officers are eligible to participate in the 401(k) plan on the same basis as our other employees. The 401(k) plan is intended to qualify as a tax-qualified plan under Section 401(k) of the Internal Revenue Code. The 401(k) plan provides that each participant may make pre-tax deferrals from his or her compensation up to the statutory limit, which is $22,500 for calendar year 2023. Participants that are 50 years or older can also make “catch-up” contributions which, in calendar year 2023, may be up to an additional $7,500 above the statutory limit. Participant contributions are held and invested, pursuant to the participant’s instructions, by the plan’s trustee. In 2022, we matched 100% of employee deferrals under the 401(k) plan up to the first 6% of eligible compensation during 2022.
Tax Gross-Up
Pursuant to Mr. McKenna’s employment letter agreement, in the event a change in control occurs and an excise tax is imposed by reason of the application of Sections 280G and 4999 of the Internal Revenue Code as a result of any compensatory payments made to him under such arrangements in connection with such change in control, he will be entitled to an additional “gross-up” payment in an amount equal to any such excise tax plus any taxes resulting from such payments.
Post-Termination and Change in Control Benefits
Our named executive officers may become entitled to certain benefits or enhanced benefits in connection with a change in control of our company. The employment agreements with each of our named executive officers provide for accelerated vesting of all outstanding equity awards, as well as certain other benefits upon a qualifying termination in connection with a change in control of our company. For additional discussion, please see “—Employment Letter Agreements with Our Named Executive Officers” below.
20
Clawbacks
While our Chief Executive Officer and Chief Financial Officer are subject to any recovery rights that are provided under applicable laws, including SOX, we have not yet adopted a compensation recovery policy as required under the Dodd-Frank Act. We are reviewing the final clawback rule adopted by the SEC that implements the applicable provisions of the Dodd-Frank Wall Street Reform and Consumer Protection Act and Nasdaq’s related proposed listing standard, in each case relating to recoupment of incentive-based compensation. We will implement its clawback policy in accordance with the new listing standard when the new listing standard becomes final.
Tax and Accounting Considerations
Deductibility of Executive Compensation
The compensation committee and our board of directors have considered the potential future effects of Section 162(m) of the Internal Revenue Code on the compensation paid to our executive officers. Section 162(m) disallows a tax deduction for any publicly held corporation for individual compensation exceeding $1.0 million in any taxable year for “covered employees.” While we consider the tax deductibility of each element of executive compensation as a factor in our overall compensation program, the compensation committee, however, retains the discretion to approve compensation that may not qualify for the compensation deduction if, considering all applicable circumstances, it would be in our best interest for such compensation to be paid without regard to whether it may be tax deductible.
Accounting for Stock-Based Compensation
Under FASB ASC 718, we are required to estimate the grant date “fair value” for each grant of equity award using various assumptions. This calculation is performed for accounting purposes and reported in the compensation tables below, even though recipients may never realize any value from their awards. ASC 718 also requires us to recognize the compensation cost of stock-based awards in our income statements over the period that an employee is required to render service in exchange for the award.
Risk Assessment of Compensation Program
In March 2023, management assessed our compensation program for the purpose of reviewing and considering any risks presented by our compensation policies and practices that are reasonably likely to have a material adverse effect on us. As part of that assessment, management reviewed the primary elements of our compensation program, including base salary, short-term incentive compensation and long-term incentive compensation. Management’s risk assessment included a review of the overall design of each primary element of our compensation program, and an analysis of the various design features, controls and approval rights in place with respect to compensation paid to management and other employees that mitigate potential risks to us that could arise from our compensation program. Following the assessment, management determined that our compensation policies and practices did not create risks that were reasonably likely to have a material adverse effect on us and reported the results of the assessment to our compensation committee.
21
REPORT OF COMPENSATION COMMITTEE OF THE BOARD OF DIRECTORS
The compensation committee of our board of directors has submitted the following report for inclusion in this Amendment:
The compensation committee has reviewed and discussed with management the Compensation Discussion and Analysis set forth above. Based on such review and discussions, the compensation committee has recommended to the board of directors that the Compensation Discussion and Analysis be included in this Amendment to the Annual Report on Form 10-K for the year ended December 31, 2022, filed by us with the SEC.
This report of the compensation committee is not “soliciting material,” shall not be deemed “filed” with the SEC and shall not be incorporated by reference by any general statement incorporating by reference this Amendment or the Original Form 10-K into any filing under the Securities Act of 1933, as amended, or the Securities Exchange Act of 1934, as amended, whether made before or after the date hereof and irrespective of any general incorporation language in any such filing, except to the extent that we specifically incorporate this information by reference, and shall not otherwise be deemed filed under such acts.
The foregoing report has been furnished by the compensation committee.
Respectfully submitted,
Compensation Committee
Joseph C. Papa (Chairperson)
Mary Szela
Fred Hassan
22
COMPENSATION TABLES
2022 Summary Compensation Table
The following table presents summary information regarding the total compensation that was awarded to, earned by or paid to our named executive officers for services rendered for the years ended December 31, 2022, 2021, and 2020.
Name and Principal Position | Year | Salary ($) | Bonus ($)(1) | Stock Awards ($)(2) | Option Awards ($)(2) | Non-equity Incentive Plan Compensation ($)(3) | All Other Compensation ($)(4) | Total ($) | ||||||||||||||||||||||||
Mark C. McKenna Chairman, President and Chief Executive Officer | 2022 | 592,300 | — | 4,089,853 | 14,520,628 | 710,760 | 22,547 | 19,936,088 | ||||||||||||||||||||||||
| 2021 | 565,522 | 1,000,000 | — | 9,813,366 | 632,500 | 17,645 | 12,029,033 | |||||||||||||||||||||||||
| 2020 | 500,000 | 750,000 | — | 1,393,712 | — | 262,250 | 2,905,962 | |||||||||||||||||||||||||
Keith W. Marshall, Ph.D. Chief Financial Officer and Treasurer | 2022 | 450,000 | — | 1,081,172 | 5,211,847 | 360,000 | 19,452 | 7,122,471 | ||||||||||||||||||||||||
| 2021 | 418,681 | 200,000 | — | 3,132,618 | 340,000 | 12,986 | 4,104,285 | |||||||||||||||||||||||||
| 2020 | 157,212 | 73,000 | — | 531,894 | — | 210 | 762,316 | |||||||||||||||||||||||||
Mark Stenhouse(5) Chief Operating Officer | 2022 | 437,750 | — | 1,081,172 | 4,272,563 | 350,200 | 19,452 | 6,161,137 | ||||||||||||||||||||||||
| 2021 | 359,615 | 25,000 | — | 2,909,459 | 212,500 | 10,674 | 3,512,248 | |||||||||||||||||||||||||
| (1) | For 2021, represents a bonus paid to Mr. McKenna and Dr. Marshall in connection with the consummation of our initial public offering pursuant to their employment agreements, and for Mr. Stenhouse, represents a $25,000 signing bonus in connection with his commencement of employment pursuant to his employment agreement. For 2020, the amounts represent the discretionary annual bonuses paid to our named executive officers for 2020 service and performance. For 2020, the total for Mr. McKenna also includes a transaction bonus of $500,000 paid in August 2020 in consideration of the company’s execution of the co-development and manufacturing agreement with Dr. Falk Pharma GmbH for our PRA052 program (the Falk Agreement). |
| (2) | Represents the grant date fair value of RSUs and option awards granted in the applicable fiscal year. In accordance with SEC rules, this column reflects the aggregate fair value of the awards granted to the named executive officers computed as of the applicable grant date in accordance with FASB ASC Topic 718 for stock-based compensation transactions (ASC 718). Assumptions used in the calculation of these amounts are included in Note 9 to our consolidated financial statements included in on the Original Form 10-K for the year ended December 31, 2022, which was filed with the SEC on February 28, 2023. This amount does not reflect the actual economic value that will be realized by the named executive officers upon the vesting or exercise of the awards or the sale of the common stock underlying such awards. |
| (3) | Represents annual bonuses paid to the named executive officers for performance during the applicable year. |
| (4) | Includes matching contributions made by us on behalf of the named executive officers under our 401(k) plan (in 2022, $18,300 for Mr. McKenna, $18,300 for Dr. Marshall and $18,300 for Mr. Stenhouse; in 2021, $11,600 for Mr. McKenna and Dr. Marshall and $9,854 for Mr. Stenhouse; and, in 2020, $4,615 for Mr. McKenna), life insurance premiums paid by us on behalf of the named executive officers and a medical exam for Mr. McKenna. In 2020, Mr. McKenna also received $255,273 in relocation assistance provided by the company, of which $88,124 was a tax gross up on such portion of the relocation benefits that were taxable to him. |
| (5) | Mr. Stenhouse commenced employment with us on March 1, 2021. The total for Mr. Stenhouse in the “Option Awards” column for 2021 includes an incremental $5,000 in stock-based compensation expense under FASB ASC 718 incurred due to the acceleration during 2021 of vesting of his outstanding equity award granted on May 23, 2018 and the extension of the exercise period of such award by one year in connection with being a former member of our board. |
23
2022 Grants of Plan-Based Awards
The following table sets forth summary information regarding grants of plan-based awards made to our named executive officers during the year ended December 31, 2022.
| Estimated Future Payouts Under Non-Equity Incentive Plan Awards(1) | All Other Stock Awards: Number of Shares of Stock or Units (#)(2) | All Other | ||||||||||||||||||||||||||||||||||
Name | Approval Date | Grant Date | Threshold ($) | Target ($) | Maximum ($) | Option Awards: Number of Securities Underlying Options (#)(3) | Exercise or Base Price of Option Awards ($/sh) | Grant Date Fair Value of Stock and Option Awards ($)(4) | ||||||||||||||||||||||||||||
Mark C. McKenna | — | — | 177,690 | 355,380 | 710,760 | — | — | — | — | |||||||||||||||||||||||||||
| 5/20/2022 | 5/20/2022 | — | — | — | — | 246,639 | 29.56 | 5,004,305 | ||||||||||||||||||||||||||||
| 12/08/2022 | 12/09/2022 | — | — | — | — | 128,930 | 110.20 | 9,516,323 | ||||||||||||||||||||||||||||
| 12/08/2022 | 12/09/2022 | — | — | — | 37,113 | — | — | 4,089,853 | ||||||||||||||||||||||||||||
Keith W. Marshall, Ph.D. | — | — | 90,000 | 180,000 | 360,000 | — | — | — | — | |||||||||||||||||||||||||||
| 5/20/2022 | 5/20/2022 | — | — | — | — | 86,582 | 29.56 | 1,756,749 | ||||||||||||||||||||||||||||
| 12/08/2022 | 12/09/2022 | — | — | — | — | 34,085 | 110.20 | 2,515,814 | ||||||||||||||||||||||||||||
| 12/08/2022 | 12/09/2022 | — | — | — | 9,811 | — | — | 1,081,172 | ||||||||||||||||||||||||||||
| 12/16/2022 | 12/16/2022 | 12,920 | 108.76 | 939,284 | ||||||||||||||||||||||||||||||||
Mark Stenhouse | — | — | 87,550 | 175,100 | 350,200 | — | — | — | — | |||||||||||||||||||||||||||
| 5/20/2022 | 5/20/2022 | — | — | — | — | 86,582 | 29.56 | 1,756,749 | ||||||||||||||||||||||||||||
| 12/08/2022 | 12/09/2022 | — | — | — | — | 34,085 | 110.20 | 2,515,814 | ||||||||||||||||||||||||||||
| 12/08/2022 | 12/09/2022 | — | — | — | 9,811 | — | — | 1,081,172 | ||||||||||||||||||||||||||||
| (1) | Represents the threshold, target and maximum annual bonus payouts pursuant to our annual bonus program, as described above. |
| (2) | Represents RSUs that vest as to 25% of such grant on each of the first four anniversaries of the grant date, subject to the recipient’s continued service through each vesting date. In addition, the employment agreements with our named executive officers provide for accelerated vesting under certain circumstances. For additional discussion, please see “— Employment Letter Agreements with our Named Executive Officers” below. |
| (3) | The stock options granted on May 20, 2022 vest in full and become exercisable on the third anniversary of the grant date, subject to the recipient’s continued employment or service through the applicable vesting date. The stock options granted on December 9, 2022 vest as to 25% of such grant on the one year anniversary of the vesting commencement date, and monthly thereafter in equal installments until fully vested at the fourth anniversary of the vesting commencement date, subject to the recipient’s continued service through each vesting date. The stock option award granted to Dr. Marshall on December 16, 2022 will vest to 25% of such grant on the one year anniversary of the grant date, and monthly thereafter in equal installments until fully vested at the fourth anniversary of the grant date, subject to the recipient’s continued service through each vesting date. In addition, the employment agreements with our named executive officers provide for accelerated vesting under certain circumstances. For additional discussion, please see “— Employment Letter Agreements with our Named Executive Officers” below. |
| (4) | Represents the grant date fair value of the RSUs and stock option awards granted in the applicable fiscal year. In accordance with SEC rules, this column reflects the aggregate fair value of the awards granted to the named executive officers computed as of the applicable grant date in accordance with ASC 718. Assumptions used in the calculation of these amounts are included in Note 9 to our consolidated financial statements included in on the Original Form 10-K for the year ended December 31, 2022, which was filed with the SEC on February 28, 2023. This amount does not reflect the actual economic value that will be realized by the named executive officers upon the vesting or exercise of the awards or the sale of the common stock underlying such awards. |
Option Exercises and Stock Vested
| None | of our named executive officers exercised any stock options or vested in any stock awards during 2022. |
Pension and Non-Qualified Deferred Compensation
| None | of our named executive officers participated in any pension or non-qualified deferred compensation plans during 2022. |
Employment Letter Agreements with our Named Executive Officers
Below are written descriptions of our employment agreements with each of our named executive officers. Each of our named executive officers’ employment is “at will” and may be terminated at any time.
24
Employment Letter Agreement with Mr. McKenna
We have entered into an employment letter agreement with Mr. McKenna, which governs the terms of his employment with us as our President and Chief Executive Officer. Regardless of the manner in which his service terminates, Mr. McKenna is entitled to receive amounts previously earned during his term of service, including unpaid salary and bonus. In addition, Mr. McKenna is entitled to certain severance benefits under his employment letter agreement in the event of his termination under certain circumstances, subject to his execution of a release of claims and compliance with post-termination obligations.
Upon a termination without cause or resignation for good reason (as such terms are defined below), Mr. McKenna is entitled to: (i) continuation of his base salary for 12 months (such applicable period, the severance period), (ii) his target bonus, payable in lump sum, (iii) payment of the COBRA premiums for him and his eligible dependents until the end of the severance period, and (iv) accelerated vesting of any unvested time-based vesting equity awards. In the event of Mr. McKenna’s termination without cause or resignation for good reason within 24 months following a change in control (as such term is defined in our 2021 Plan), the severance period will be increased to 18 months. Mr. McKenna is also eligible to receive a gross-up payment for any excise taxes payable under Section 280G and 4999 of the Internal Revenue Code as a result of a change in control transaction (plus an additional amount necessary to put him in the same after-tax position as if no excise tax had been imposed). For purposes of Mr. McKenna’s employment letter agreement:
| • | “cause” generally means his (i) conviction of a felony, plea of guilty or no contest to a felony, or confession of guilt to a felony; (ii) act or omission which constitutes willful misconduct or gross negligence that results in loss, damage or injury to us or our prospects, including, but not limited to (a) dishonesty or a breach of fiduciary duty to us or our stockholders, or (b) theft, fraud, embezzlement or other illegal conduct; (iii) continued failure, refusal or unwillingness to perform, to the reasonable satisfaction of our board of directors determined in good faith, any material duty or responsibility assigned to him, which failure of performance continues for a period of more than 30 days after written notice thereof has been provided by our board of directors, setting forth in reasonable detail the nature of such failure of performance; or (iv) the material breach of any of the provisions of his employment letter agreement or any other written agreement between him and us. |
| • | “good reason” generally means a resignation that occurs following the occurrence of any of the following without his written consent: (i) a material change in the geographic location at which he must perform his duties (with a relocation outside a 35-mile radius of his principal place of employment prior to such relocation considered material for this purpose); (ii) a material reduction of his base compensation, target bonus and/or benefits; (iii) any action or inaction that constitutes a material breach of his employment letter agreement by us; or (iv) a material reduction in his authority, duties or responsibilities (including a requirement to report to any person or entity other than our board of directors, or following a change in control, the board of directors (or similar governing body) of the ultimate parent company of the surviving entity in such change in control that has at least one class of publicly traded securities listed on a national stock exchange). |
Employment Letter Agreement with Dr. Marshall
We have entered into an employment letter agreement with Dr. Marshall, which governs the terms of his employment with us as our Chief Financial Officer. Regardless of the manner in which his service terminates, Dr. Marshall is entitled to receive amounts previously earned during his term of service, including unpaid salary and bonus. In addition, Dr. Marshall is entitled to certain severance benefits under his employment letter agreement in the event of his termination under certain circumstances, subject to his execution of a release of claims and compliance with post-termination obligations.
Upon a termination without cause or resignation for good reason, Dr. Marshall is entitled to: (i) continuation of his base salary for 12 months (such applicable period, the severance period), (ii) his target bonus, payable in lump sum, (iii) payment of the COBRA premiums for him and his eligible dependents until the end of the severance period, and (iv) accelerated vesting of any unvested time-based vesting equity awards. For purposes of Dr. Marshall’s employment letter agreement, the terms cause and good reason generally have the same meanings as given to such terms in Mr. McKenna’s employment agreement described above.
25
Employment Letter Agreement with Mr. Stenhouse
We have entered into an employment letter agreement with Mr. Stenhouse, which governs the terms of his employment with us as Chief Operating Officer. Regardless of the manner in which his service terminates, Mr. Stenhouse is entitled to receive amounts previously earned during his term of service, including unpaid salary and bonus. In addition, Mr. Stenhouse is entitled to certain severance benefits under his employment letter agreement in the event of his termination under certain circumstances, subject to his execution of a release of claims and compliance with post-termination obligations.
Upon a termination without cause or resignation for good reason, Mr. Stenhouse is entitled to: (i) continuation of his base salary for 12 months (such applicable period, the severance period), (ii) his target bonus, payable in lump sum, (iii) payment of the COBRA premiums for him and his eligible dependents until the end of the severance period, and (iv) accelerated vesting of any unvested time-based vesting equity awards. Pursuant to his employment letter, Mr. Stenhouse also received a $25,000 signing bonus to cover temporary housing costs in San Diego. For purposes of Mr. Stenhouse’s employment letter agreement, the terms cause and good reason generally have the same meanings as given to such terms in Mr. McKenna’s employment agreement described above.
Restrictive Covenant Obligations
Each of our named executive officers is subject to restrictive covenant obligations under our standard confidentiality and inventions assignment agreement.
Potential Payments Upon Termination or Change in Control
The following table summarizes the potential payments to our named executive officers in the scenarios listed in the table below. The table assumes that the termination of employment or change in control, as applicable, occurred on December 31, 2022. Except as described in the footnote below, the value of the accelerated vesting of stock and option awards was computed using $110.00, which was the closing price per share of our common stock on December 30, 2022, the last trading day of 2022 (less, in the case of option awards, the exercise price per share of such option awards).
Triggering Event | Cash Severance ($)(1) | Accelerated Options ($)(2) | Accelerated RSUs ($)(2) | Health Benefits ($)(3) | Tax Gross-Up ($)(4) | Total ($) | ||||||||||||||||||
Mark C. McKenna | ||||||||||||||||||||||||
Involuntary Termination Without Cause/Resignation for Good Reason Apart from a CIC | 947,680 | 106,995,928 | 4,082,430 | 38,226 | — | 112,064,264 | ||||||||||||||||||
Death/Disability | — | — | — | — | — | — | ||||||||||||||||||
Involuntary Termination Without Cause/Resignation for Good Reason in Connection with a CIC | 1,243,830 | 106,995,928 | 4,082,430 | 57,338 | 17,591,428 | 129,970,954 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||
Keith W. Marshall, Ph.D. | ||||||||||||||||||||||||
Involuntary Termination Without Cause/Resignation for Good Reason Apart from a CIC | 630,000 | 32,175,954 | 1,079,210 | 38,226 | — | 33,923,390 | ||||||||||||||||||
Death/Disability | — | — | — | — | — | — | ||||||||||||||||||
Involuntary Termination Without Cause/Resignation for Good Reason in Connection with a CIC | 630,000 | 32,175,954 | 1,079,210 | 38,226 | — | 33,923,390 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||
Mark Stenhouse | ||||||||||||||||||||||||
Involuntary Termination Without Cause/Resignation for Good Reason Apart from a CIC | 612,850 | 24,174,324 | 1,079,210 | 26,808 | — | 25,893,192 | ||||||||||||||||||
Death/Disability | — | — | — | — | — | — | ||||||||||||||||||
Involuntary Termination Without Cause/Resignation for Good Reason in Connection with a CIC | 612,850 | 24,174,324 | 1,079,210 | 26,808 | — | 25,893,192 | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
| |||||||||||||
26
| (1) | Represents cash severance payable in the form of base salary continuation and a lump sum target bonus upon a termination without cause or a resignation for good reason pursuant to the named executive officers’ employment letter agreements. For the named executive officers, represents 12 months’ base salary plus full target bonus (increased to 18 months’ base salary and full target bonus for Mr. McKenna in the event of such a termination within 24 months following a change in control). |
| (2) | The value attributable to the accelerated options represents the excess of the closing price per share of our common stock of $110.00 on December 30, 2022, the last trading day of 2022, over the exercise price of the unvested options the vesting of which accelerates in connection with the specified event. The value attributable to the accelerated RSUs represents the closing price per share of our common stock of $110.00 on December 30, 2022, the last trading day of 2022, multiplied by the number of RSUs the vesting of which accelerates in connection with the specified event. In the event of an involuntary termination without cause or resignation for good reason, all outstanding unvested stock and option awards held by the named executive officers will vest upon such termination. |
| (3) | Represents the value of the continuation of health benefits for the period corresponding to the period for which the named executive officer will receive cash severance benefits following the date of the named executive officer’s termination. |
| (4) | Mr. McKenna is also eligible to receive a gross-up payment for any excise taxes payable under Section 280G and 4999 of the Internal Revenue Code as a result of a change in control transaction (plus an additional amount necessary to put him you in the same after-tax position as if no excise tax had been imposed). This column includes the estimated amount of reimbursement for excise taxes payable pursuant to Sections 280G and 4999 of the Internal Revenue Code arising as a result of the receipt of benefits and payments as a result of the applicable triggering event, and such estimated amount is based on the assumptions noted above for the expected amounts payable upon the applicable triggering event. The estimates of “excess parachute payments” for purposes of these calculations do not take into account any mitigation for payments which could be shown (under the facts and circumstances) not to be contingent on a change in control or for any payments being made in consideration of noncompetition agreements or as reasonable compensation. |
DIRECTOR COMPENSATION
Director Compensation Table
The following table presents summary information regarding the total compensation that was awarded to, earned by or paid to our non-employee directors for services rendered during the year ended December 31, 2022.
Name | Fees Earned or Paid in Cash ($) | Stock Awards ($) | Option Awards ($)(1) | All Other Compensation ($) | Total ($) | |||||
Helen C. Adams, CPA | 40,000 | — | 404,043 | — | 444,043 | |||||
Fred Hassan | 47,500 | — | 394,414 | — | 441,914 | |||||
Martin Hendrix, Ph.D. | 55,000 | — | 384,785 | — | 439,785 | |||||
James D. Laur | 40,000 | — | 384,785 | — | 424,785 | |||||
Joseph C. Papa | 40,000 | — | 423,282 | — | 463,282 | |||||
Judith L. Swain, M.D. | 45,000 | — | 384,785 | — | 429,785 | |||||
Mary Szela | 57,500 | — | 384,785 | — | 442,285 |
| (1) | Represents the grant date fair value of option awards granted in during 2022. In accordance with SEC rules, this column reflects the aggregate fair value of the awards granted to the directors computed as of the applicable grant date in accordance with ASC 718. Assumptions used in the calculation of these amounts are included in Note 9 to our consolidated financial statements included in on the Original Form 10-K for the year ended December 31, 2022, which was filed with the SEC on February 28, 2023. This amount does not reflect the actual economic value that will be realized by the directors upon the vesting or exercise of the awards or the sale of the common stock underlying such awards. |
The aggregate number of shares subject to the stock options outstanding as of December 31, 2022 for the individuals who served as non-employee directors during 2022 was as follows:
Name | Number of Securities Underlying Options Outstanding at December 31, 2022 | |
Helen C. Adams, CPA | 52,609 | |
Fred Hassan | 61,554 | |
Martin Hendrix, Ph.D. | 60,500 | |
James D. Laur | 60,500 |
27
Name | Number of Securities Underlying Options Outstanding at December 31, 2022 | |||
Joseph C. Papa | 89,708 | |||
Judith L. Swain, M.D. | 50,500 | |||
Mary Szela | 60,500 | |||
Non-Employee Director Compensation Program
We maintain a non-employee director compensation program that provides for annual retainer fees and/or long- term equity awards for our non-employee directors. Each non-employee director receives an annual retainer of $40,000. In addition, a non-employee director serving as chairman of the board or lead independent director will receive equity awards with a value of $25,000. In addition, a non-employee director serving as the chair of the audit, compensation, or nominating and corporate governance committees will receive equity awards with a value of $20,000, $15,000 and $10,000, respectively. Prior to 2023, these awards were granted solely in the form of stock options. Commencing in 2023, these awards will be granted in the form of stock options and restricted stock units, weighted equally. The number of options granted will be calculated on the grant date in accordance with the Black-Scholes option pricing model (utilizing the same assumptions that the company utilizes in preparation of its financial statements and the average closing price per share of the company’s common stock for the 30 trading days preceding the grant date). The number of restricted stock units granted will be calculated on the grant date based on the closing price per share of the company’s common stock the trading day preceding the grant date. A non-employee director serving as a member (other than the chair) of the audit, compensation or nominating and corporate governance committee will receive additional annual cash retainers of $10,000, $7,500 and $5,000, respectively.
Non-employee directors will receive, upon their initial election or appointment to our board of directors, a number of equity awards having a value of $600,000. In addition, on the date of each annual meeting of our stockholders, non-employee directors will receive annual equity awards having a value of $400,000. Prior to 2023, these awards were granted solely in the form of stock options. Commencing in 2023, these awards will be granted in the form of stock options and restricted stock units, weighted equally. The number of options and restricted stock units to be granted are calculated in the same manner as described above. Initial option awards will vest monthly over three years and initial restricted stock units will vest in three annual installments, and annual awards will vest on the first to occur of (1) the first anniversary of the grant date or (2) the next occurring annual meeting of our stockholders, in each case subject to the non-employee director’s continued service through the applicable vesting date. In addition, all equity awards granted to our non-employee directors will vest upon a change in control of our company.
Except as noted above, the other terms of the non-employee director compensation policy adopted at the time of our initial public offering remain in effect.
Compensation under our non-employee director compensation policy is subject to the annual limits on non-employee director compensation set forth in the 2021 Plan. Our board of directors or its authorized committee may modify the non-employee director compensation program from time to time in the exercise of its business judgment, taking into account such factors, circumstances and considerations as it shall deem relevant from time to time, subject to the annual limit on non-employee director compensation set forth in the 2021 Plan. As provided in the 2021 Plan, our board of directors or its authorized committee may make exceptions to this limit for individual non-employee directors in extraordinary circumstances, as the board of directors or its authorized committee may determine in its discretion, provided that the non-employee director receiving such additional compensation may not participate in the decision to award such compensation or in other compensation decisions involving non-employee directors.
28
ITEM 12. SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS
The following table and accompanying footnotes set forth certain information with respect to the beneficial ownership of our common stock at April 21, 2023 for:
| • | each of our directors; |
| • | each of our Named Executive Officers; |
| • | all of our current directors and executive officers as a group; and |
| • | each period, or group of affiliated persons, who beneficially owned more than 5% of our outstanding common stock. |
We have determined beneficial ownership in accordance with the rules of the SEC, and the information is not necessarily indicative of beneficial ownership for any other purpose. Except as indicated by the footnotes below, we believe, based on information furnished to us, that the persons and entities named in the table below have sole voting and sole investment power with respect to all shares of common stock that they beneficially owned,
subject to applicable community property laws.
Applicable percentage ownership is based on 47,788,541 shares of common stock outstanding as of April 21, 2023. In computing the number of shares of common stock beneficially owned by a person and the percentage ownership of that person, we deemed to be outstanding all shares of common stock subject to options, warrants or other rights held by that person or entity that are currently exercisable within 60 days of April 21, 2023. We did not deem these shares outstanding, however, for the purpose of computing the percentage ownership of any other person. Unless otherwise indicated, the address of each beneficial owner listed in the table below is c/o Prometheus Biosciences, Inc., 3050 Science Park Road, San Diego, California 92121.
| Beneficial Ownership | ||||||||
| Number | Percentage | |||||||
Name of Beneficial Owner | ||||||||
5% or Greater Stockholders | ||||||||
Entities affiliated with Cedars Sinai Intellectual Property Company(1) | 4,001,132 | 8.4% | ||||||
Entities affiliated with Nestlé S.A.(2) | 3,730,841 | 7.8% | ||||||
Entities affiliated with RTW Investments(3) | 3,354,303 | 7.0% | ||||||
BlackRock, Inc.(4) | 2,807,704 | 5.9% | ||||||
Entities affiliated with Point72 Biotech Private Investments, LLC(5) | 3,114,107 | 6.5% | ||||||
Named Executive Officers and Directors | ||||||||
Mark C. McKenna(6) | 1,136,501 | 2.3% | ||||||
Keith W. Marshall, Ph.D.(7) | 261,973 | * | ||||||
Mark Stenhouse(8) | 224,486 | * | ||||||
Joseph C. Papa(9) | 80,898 | * | ||||||
Helen C. Adams, CPA(10) | 40,609 | * | ||||||
Fred Hassan(11) | 49,331 | * | ||||||
Martin Hendrix, Ph.D.(12) | 50,500 | * | ||||||
James Laur(13) | 50,500 | * | ||||||
Judith L. Swain, M.D.(14) | 38,000 | * | ||||||
Mary Szela(15) | 51,611 | * | ||||||
|
|
|
| |||||
All executive officers and directors as a group (10 persons) | 1,984,409 | 4.0% | ||||||
|
|
|
| |||||
| * | Less than 1%. |
| (1) | Based on the Amendment No. 2 to Schedule 13G filed with the SEC on March 8, 2023. Consists of 4,001,132 shares of common stock held by Cedars Sinai Intellectual Property Company (CSIPC), a wholly-owned subsidiary of Cedars-Sinai Medical Center (CSMC). CSMC is deemed to share voting and dispositive power with respect to the shares held by CSIPC. Thomas M. Priselac, the President and Chief Executive Officer of CSMC, and David M. Wrigley, the Executive Vice President and Chief Financial Officer of CSMC, are deemed to share voting and dispositive power with respect to the shares held by CSIPC. The address of CSIPC, CSMC, Mr. Priselac and Mr. Wrigley is c/o Cedars-Sinai Medical Center, 8700 Beverly Boulevard, Los Angeles, California 90048. |
29
| (2) | Based on the Schedule 13G filed with SEC on February 14, 2022. Consists of (i) 2,085,841 shares of common stock held by Nestlé Health Science US Holdings, Inc. (NHS) and (ii) 1,645,000 shares of common stock held by Société des Produits Nestlé S.A. (SPN). Each of (a) SPN, an indirect parent of NHS, (b) NIMCO US, Inc. (NIMCO), the direct parent of NHS, (c) Nestlé US Holdco, Inc. (Nestlé US Holdco), an indirect parent of NHS, and (d) Nestlé S.A., a publicly traded company and the ultimate parent of each of NHS, NIMCO, Nestlé US Holdco and SPN, has shared voting power and shared dispositive power with respect to the 2,085,841 shares held by NHS. Each of (x) SPN and (y) Nestlé S.A. has shared voting power and shared dispositive power with respect to the 1,645,000 shares held by SPN. The principal executive office of NHS, NIMCO and Nestlé US Holdco is 1812 North Moore Street, Arlington, VA 22209 and the principal executive office of SPN and Nestlé S.A. is Avenue Nestlé 55, CH-1800, Vevey Switzerland. |
| (3) | Based on the Amendment No. 1 to Schedule 13G filed with the SEC on February 14, 2023 by RTW Investments, LP (the Adviser) and Roderick Wong. Consists of 3,354,303 shares of common stock held in the aggregate by one or more private funds (together the RTW Funds) managed by the Adviser. The Adviser, in its capacity as the investment manager of the RTW Funds, has the power to vote and the power to direct the disposition of all shares held by the RTW Funds. Accordingly, the Adviser may be deemed to beneficially own an aggregate of 3,354,303 shares. Roderick Wong is the Managing Partner and Chief Investment Officer of the Adviser and as such shares voting and investment control over such shares. The address of The Adviser, RTW Funds and Mr. Wong is 40 10th Avenue, Floor 7, New York, New York 10014. |
| (4) | Based on the Schedule 13G filed with the SEC on February 3, 2023, BlackRock, Inc. has the sole power to vote or direct the vote of an aggregate of 2,774,595 shares of common stock, and has the sole power to dispose or direct the disposition of an aggregate of 2,807,704 shares of common stock. The address of BlackRock, Inc. is 55 East 52nd Street, New York, NY 10055. |
| (5) | Based on the Amendment No. 2 to Schedule 13G filed with SEC on February 14, 2023. Consists of (i) 1,848,024 shares of common stock held by Point72 Biotech Private Investments, LLC (Point72 Biotech) and (ii) 1,266,083 shares of common stock held an investment fund managed by Point72 Asset Management, L.P. (Point72 Asset Management). Differentiated Ventures Investments, LLC (DVI) is the managing member of Point72 Biotech and may be deemed to share beneficial ownership over the 1,848,024 shares held by Point72 Biotech. 72 Investment Holdings, LLC (72 IH) is the sole member of DVI and may be deemed to share beneficial ownership over the 1,848,024 shares of which DVI may be deemed the beneficial owner. Point72 Asset Management is the manager to a private investment fund and pursuant to an investment management agreement, Point72 Asset Management maintains investment and voting power with respect to securities held by such private investment fund and, as a result, may be deemed to share beneficial ownership over the 1,266,083 shares held by such private investment fund. Point72 Capital Advisors, Inc. (Point72 Capital Advisors) is the general partner of Point72 Asset Management and may be deemed to share beneficial ownership of the 1,266,083 shares of which Point72 Asset Management may be deemed the beneficial owner. Steven A. Cohen is the sole member of 72 IH and Point72 Capital Advisors and may be deemed to beneficially own both the 1,848,024 shares of which 72 IH may be deemed the beneficial owner and the 1,266,083 shares of which Point72 Capital Advisors may be deemed the beneficial owner. The address of Point72 Biotech, Point72 Asset Management, DVI, 72 IH, Point72 Capital Advisors and Mr. Cohen is c/o Point72, L.P., 72 Cummings Point Road, Stamford, CT 06902. |
| (6) | Consists of (i) 18,031 shares of common stock held by Mr. McKenna and (ii) 1,118,470 shares of common stock underlying options held by Mr. McKenna that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (7) | Consists of 261,973 shares of common stock underlying options held by Dr. Marshall that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (8) | Consists of (i) 16,631 shares of common stock held by Mr. Stenhouse and (ii) 207,855 shares of common stock underlying options held by Mr. Stenhouse that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (9) | Consists of (i) 15,000 shares of common stock held by Mr. Papa and (ii) 65,898 shares of common stock underlying options held by Mr. Papa that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (10) | Consists of (i) 500 shares of common stock held by Ms. Adams and (ii) 40,109 shares of common stock underlying options held by Ms. Adams that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (11) | Consists of 49,331 shares of common stock underlying options held by Mr. Hassan that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (12) | Consists of 50,500 shares of common stock underlying options held by Dr. Hendrix that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (13) | Consists of 50,500 shares of common stock underlying options held by Mr. Laur that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (14) | Consists of 38,000 shares of common stock underlying options held by Dr. Swain that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
| (15) | Consists of 51,611 shares of common stock underlying options held by Ms. Szela that are exercisable as of April 21, 2023 or that will become exercisable within 60 days after such date. |
30
Equity Compensation Plan Information
The following table sets forth information as of December 31, 2022 regarding common stock that may be issued under our equity compensation plans, consisting of the 2021 Plan, the Prometheus Biosciences, Inc. 2017 Equity Incentive Plan (the 2017 Plan) and the Prometheus Biosciences, Inc. 2021 Employee Stock Purchase Plan (the 2021 ESPP). We do not have any non-shareholder approved equity compensation plans.
| Plan Category | (A) Number of Securities to be Issued Upon Exercise of Outstanding Options, Warrants and Rights | (B) Weighted Average Per Share Exercise Price of Outstanding Options, Warrants and Rights | (C) Number of Securities Remaining Available for Future Equity Compensation Plans (Excluding Securities Reflected in Column (A)) | |||||||||
Equity compensation plans approved by security holders | 7,670,698 | (1) | $ | 22.40 | (2) | 2,545,436 | (3) | |||||
Equity compensation plans not approved by security holders | — | — | — | |||||||||
Total | 7,670,698 | $ | 22.40 | 2,545,436 | ||||||||
| (1) | Includes 3,464,083 outstanding options to purchase common stock under the 2021 Plan, 251,908 shares of common stock underlying RSUs under the 2021 Plan and 3,954,707 outstanding options to purchase common stock under the 2017 Plan. |
| (2) | Represents the weighted-average exercise price of outstanding options. RSUs are not taken into account for purposes of determining the weighted average exercise price. |
| (3) | Includes 1,861,545 shares of common stock available for issuance under the 2021 Plan and 683,891 shares of common stock available for issuance under the 2021 ESPP (all of which were eligible for purchase pursuant to the offering period in effect on December 31, 2022). With respect to our 2021 Plan, this number includes future awards and shares remaining available for issuance under the 2017 Plan which became available for issuance under the 2021 Plan. This amount does not include any additional shares that may become available for future issuance under the 2021 Plan pursuant to the automatic increase to the share reserve on January 1 of each of our calendar years through 2031 by the number of shares equal to the lesser of (i) 5% of the total outstanding shares of our common stock as of the immediately preceding December 31 and (ii) such smaller number of shares as is determined by our board of directors. Additionally, this amount does not include any additional shares that may become available for future issuance under the 2021 ESPP pursuant to the automatic increase to the share reserve on January 1 of each of our calendar years through 2031 by the number of shares equal to the lesser of (i) 1% of the total outstanding shares of our common stock as of the immediately preceding December 31 and (ii) such smaller number of shares as determined by our board of directors. |
ITEM 13. CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS, AND DIRECTOR INDEPENDENCE
Certain Relationships and Related Party Transactions
The following is a summary of transactions entered into or existing since January 1, 2022 to which we have been a party in which the amount involved exceeded or will exceed $120,000, and in which any of our directors, executive officers or, to our knowledge, beneficial owners of more than 5% of our capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than equity and other compensation, termination, change in control and other arrangements, which are described under “Executive Compensation.” We also describe below certain other transactions with our directors, executive officers and stockholders.
Agreements with Cedars-Sinai
In September 2017, we entered into an exclusive license agreement with Cedars-Sinai that grants us an exclusive, worldwide license from Cedars-Sinai with respect to certain patent rights, information and materials related to therapeutic targets and companion diagnostic product candidates for the diagnosis and treatment of inflammatory bowel disease and other immune-mediated diseases, in each case to conduct research and development, as well as to commercialize diagnostic or therapeutic products (Cedars-Sinai Products) that are covered by the patents or that are developed through use of the licensed rights. As upfront consideration for the original license agreement, we issued to Cedars-Sinai 257,500 shares of fully vested common stock and 335,000 shares of restricted common stock, which shares fully vested in September 2020. We are obligated to pay Cedars-Sinai low-to mid-single digit percentage royalties on net sales of therapeutic and diagnostic Cedars-Sinai Products. In August 2021, we amended and restated the agreement to, among other things, add a joint steering committee and cover new intellectual property. During the year ended December 31, 2022, the Company incurred $0.8 million in costs under the research agreements.
31
Prometheus Laboratories, Inc.
PLI Acquisition and Spinoff
In June 2019, we, NHS and SPN entered into a stock purchase agreement whereby we acquired 100% of the issued and outstanding equity of Prometheus Laboratories, Inc. (PLI) from NHS, as well as certain intellectual property rights from SPN. In consideration for the acquisition, we issued to NHS and SPN an aggregate of 25,000,000 shares of Series C convertible preferred stock in connection with our Series C convertible preferred stock financing (or approximately $25 million of stock based upon a fair market value of $1.00 per share). Under the terms of the agreement, we are also required to pay NHS $15.0 million in deferred cash payments due as follows: $5.0 million due on June 30, 2020 and $10.0 million due on June 30, 2021, which were settled as follows: $5.0 million of deferred cash payments were converted to 5,000,000 Series C convertible preferred stock in June 2020; $3.8 million of deferred cash payments were converted to 5,088,851 shares of Series D-1 convertible preferred stock in October 2020; and $6.2 million of deferred cash payments were converted to 7,219,560 shares of Series D-2 convertible preferred stock in January 2021. Martin Hendrix, Ph.D., a member of our board of directors, is the Head of Global Business Development and M&A at NHS.
On December 31, 2020, we completed a spinoff of PLI, by means of a pro rata distribution of all its outstanding stock to our stockholders of record on December 30, 2020.
Transition Services Agreement
In connection with the spinoff, we and PLI entered into a transition services agreement. Pursuant to this agreement, we provided PLI certain transitional services, including general and administrative, finance and clinical operations support, and PLI provided us with certain transitional services, including providing for the use of our current facilities under a sublease, in each case for specified monthly service fees. The initial term of the agreement was for one year, subject to earlier termination and extension thereafter. The transition services agreement was extended through June 2022 during the year ended December 31, 2021 and provided for the use of facilities under a sublease. During the year ended December 31, 2022, the Company paid PLI $1.5 million for these services under the agreement.
Registration Rights Agreement
We entered into an amended and restated investors’ rights agreement in November 2018, as amended and restated in June 2019, and as amended and restated in October 2020, with the holders of our convertible preferred stock, including entities with which certain of our directors are affiliated. The registration rights agreement provides for certain rights relating to the registration of their shares of common stock issuable upon conversion of their convertible preferred stock and certain additional covenants made by us. Except for the registration rights (including the related provisions pursuant to which we have agreed to indemnify the parties to the investors’ rights agreement), all rights under this agreement terminated upon the closing of our initial public offering. The registration rights will terminate seven years after the closing of our initial public offering.
Voting Agreement
We entered into an amended and restated voting agreement in November 2018, as amended and restated in March 2020, as further amended in August 2020, and as amended and restated in October 2020, with the holders of our convertible preferred stock, including entities with which certain of our directors are affiliated, pursuant to which the following directors were each elected to serve as members on our board of directors and continue to so serve: Helen C. Adams, CPA, Martin Hendrix, Ph.D., James Laur, Mark C. McKenna, Joseph C. Papa, Judith Swain, M.D. and Mary Szela, M.D. Pursuant to the voting agreement, Mr. McKenna, as our Chief Executive Officer, serves on our board of directors as the CEO director. Dr. Hendrix and Mr. Laur, as designated by NHS and Cedars-Sinai, respectively, and Dr. Swain and Ms. Szela were initially selected to serve on our board of directors as representatives of the holders of our convertible preferred stock. Ms. Adams and Mr. Papa were initially selected to serve on our board of directors as representatives of the holders of our common stock and convertible preferred stock, as designated by a majority of our common and preferred stockholders, voting together as a single class.
32
The voting agreement terminated upon the closing of our initial public offering, and members previously elected to our board of directors pursuant to this agreement will continue to serve as directors until they resign, are removed or their successors are duly elected by holders of our common stock. The composition of our board of directors is described in more detail under “Board of Directors” and “Corporate Governance” above.
Employment Agreements
We have entered into employment agreements with each of our named executive officers. For more information regarding these agreements, see the section in this Amendment entitled “Executive Compensation—Narrative Disclosure to Summary Compensation Table—Employment Agreements with our Named Executive Officers.”
Indemnification Agreements
We have entered into indemnification agreements with each of our directors and executive officers. These agreements, among other things, require us or will require us to indemnify each director (and in certain cases their related venture capital funds) and executive officer to the fullest extent permitted by Delaware law, including indemnification of expenses such as attorneys’ fees, judgments, fines and settlement amounts incurred by the director or executive officer in any action or proceeding, including any action or proceeding by or in right of us, arising out of the person’s services as a director or executive officer.
Our amended and restated certificate of incorporation and Bylaws provide that we will indemnify each of our directors and officers to the fullest extent permitted by the Delaware General Corporation Law. Further, we have entered into indemnification agreements with each of our directors and officers, and we have purchased a policy of directors’ and officers’ liability insurance that insures our directors and officers against the cost of defense, settlement or payment of a judgment under certain circumstances.
Stock Option and Restricted Stock Unit Grants to Executive Officers and Stock Option Grants to Directors
We have granted stock options and restricted stock units to our executive officers and stock options to our directors as more fully described in the section in this Amendment entitled “Executive Compensation.”
Policies and Procedures for Related Person Transactions
The Board adopted a written related person transaction policy, setting forth the policies and procedures for the review and approval or ratification of related-person transactions. This policy covers, with certain exceptions set forth in Item 404 of Regulation S-K under the Securities Act of 1933, as amended (the Securities Act), any transaction, arrangement or relationship, or any series of similar transactions, arrangements or relationships in which we were or are to be a participant, where the amount involved exceeds $120,000 and a related person had or will have a direct or indirect material interest, including, without limitation, purchases of goods or services by or from the related person or entities in which the related person has a material interest, indebtedness, guarantees of indebtedness and employment by us of a related person. In reviewing and approving any such transactions, our Audit Committee is tasked to consider all relevant facts and circumstances, including, but not limited to, whether the transaction is on terms comparable to those that could be obtained in an arm’s length transaction and the extent of the related person’s interest in the transaction. All of the transactions described in this section occurred prior to the adoption of this policy.
33
Director Independence
The Board currently consists of eight members. The Board has determined that all of our directors, other than Mr. McKenna and Mr. Laur, are independent directors in accordance with the listing requirements of Nasdaq. The Nasdaq independence definition includes a series of objective tests, including that the director is not, and has not been for at least three years, one of our employees and that neither the director nor any of his family members has engaged in various types of business dealings with us. In addition, as required by Nasdaq rules, the Board has made a subjective determination as to each independent director that no relationships exist, which, in the opinion of the Board, would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. In making these determinations, the Board reviewed and discussed information provided by the directors and us with regard to each director’s business and personal activities and relationships as they may relate to us and our management. There are no family relationships among any of our current directors or executive officers.
| ITEM 14. | PRINCIPAL ACCOUNTING FEES AND SERVICES |
The following table summarizes the fees of BDO USA, LLP, our independent registered public accounting firm, billed to us for each of the last two fiscal years for audit services and billed to us in each of the last two fiscal years for other services:
| Fiscal Years Ended December 31, | ||||||||
| Fee Category | 2022 | 2021 | ||||||
Audit Fees(1) | $ | 632,294 | $ | 493,683 | ||||
Audit-Related Fee | — | — | ||||||
Tax Fees | — | — | ||||||
All Other Fees | — | — | ||||||
|
|
|
| |||||
Total Fees | $ | 632,294 | $ | 493,683 | ||||
|
|
|
| |||||
| (1) | Audit fees consist of fees for the audit of our financial statements, the review of the unaudited interim financial statements included in our quarterly reports on Form 10-Q and the issuance of consents and comfort letters in connection with registration statements, including the filing of our registration statement on Form S-1 for our initial public offering. |
Audit Committee Pre-Approval of Audit and Non-Audit Services
The Audit Committee has established a policy that all audit and permissible non-audit services provided by our independent registered public accounting firm will be pre-approved by the Audit Committee, and all such services were pre-approved in accordance with this policy during the fiscal year ended December 31, 2022. These services may include audit services, audit-related services, tax services and other services. The audit committee considers whether the provision of each non-audit service is compatible with maintaining the independence of our auditors. Pre-approval is detailed as to the particular service or category of services and is generally subject to a specific budget. Our independent registered public accounting firm and management are required to periodically report to the audit committee regarding the extent of services provided by the independent registered public accounting firm in accordance with this pre-approval, and the fees for the services performed to date.
34
PART IV
ITEM 15. EXHIBITS AND FINANCIAL STATEMENT SCHEDULES
| (a) | The following documents are filed as part of this report: |
(1) FINANCIAL STATEMENTS
The financial statements required by Item 15(a) were filed as part of the Original Form 10-K under Item 8 “Financial Statements and Supplementary Data.”
(2) FINANCIAL STATEMENT SCHEDULES
All schedules to the financial statements have been omitted as the required information is either inapplicable or presented in the financial statements.
(3) EXHIBITS
35
EXHIBIT INDEX
36
| # | Indicates management contract or compensatory plan. |
| † | Portions of this exhibit have been omitted for confidentiality purposes. |
| * | This certification is deemed not filed for purpose of Section 18 of the Exchange Act or otherwise subject to the liability of that section, nor shall it be deemed incorporated by reference into any filing under the Securities Act or the Exchange Act. |
| ** | Certain exhibits and schedules have been omitted pursuant to Item 601(a)(5) of Regulation S-K. The Company hereby undertakes to furnish supplemental copies of any of the omitted exhibits and schedules upon request by the SEC; provided, however, that the Company may request confidential treatment pursuant to Rule 24b-2 of the Exchange Act for any exhibits or schedules so furnished. |
37
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the Registrant has duly caused this Amendment No. 1 to the Annual Report on Form 10-K to be signed on its behalf by the undersigned thereunto duly authorized.
| Prometheus Biosciences, Inc. | ||||||
| Date: April 28, 2023 | By: | /s/ Mark C. McKenna | ||||
| Mark C. McKenna | ||||||
| Chairman of the Board, President and Chief Executive Officer (Principal Executive Officer) | ||||||